Unbiased Cell Identity: A Guide to High-Content Imaging in Complex Neural Cultures
This article explores the transformative role of high-content imaging (HCI) in identifying and characterizing diverse cell types within mixed neural cultures, a critical challenge in neuroscience research and drug development.
Unbiased Cell Identity: A Guide to High-Content Imaging in Complex Neural Cultures
Abstract
This article explores the transformative role of high-content imaging (HCI) in identifying and characterizing diverse cell types within mixed neural cultures, a critical challenge in neuroscience research and drug development. We cover the foundational principles of HCI and Cell Painting assays that generate unique morphological fingerprints for different neural cells. The piece delves into advanced methodologies, including the implementation of convolutional neural networks (CNNs) like CellSighter for automated classification, and their application in both 2D and 3D culture systems. We also address key troubleshooting and optimization strategies for overcoming hurdles such as high culture density and segmentation errors. Finally, the article provides a rigorous validation and comparative analysis, benchmarking HCI performance against traditional methods and highlighting its superior accuracy and throughput for quality control in iPSC-derived models and preclinical screening.
The Need for Unbiased Identification: From Cellular Heterogeneity to Morphological Fingerprints
The Challenge of Variability in iPSC-Derived Neural Models
The use of induced pluripotent stem cell (iPSC)-derived neural models has revolutionized the study of neurological disorders and drug development by providing unprecedented access to human cell types that are otherwise difficult to obtain [1] [2]. However, a significant challenge impedes the reliability and reproducibility of these models: inherent variability. This variability stems from multiple sources, including genetic background of donors, differences in reprogramming techniques, and inconsistencies in differentiation protocols [2]. For researchers using high-content imaging to identify cell types in mixed neural cultures, this heterogeneity can confound results, leading to misleading conclusions and failed drug screens. This application note details the sources of this variability, provides quantitative methods for its assessment, and outlines standardized protocols to mitigate its impact, ensuring more robust and reproducible research outcomes.
The variability in iPSC-derived neural models is not random but originates from specific, identifiable stages of the cell culture process. Recognizing these sources is the first step toward implementing effective quality control.
- Genetic Background: Inter-individual genetic differences are a major contributor, accounting for 5-46% of the variation in iPSC cellular phenotypes [2]. Lines derived from the same individual are consistently more similar to each other than lines from different donors.
- Reprogramming and Culture Artifacts: The processes of reprogramming somatic cells and subsequent cell culture can introduce somatic mutations and epigenetic variations. The retention of tissue-specific DNA methylation marks from the cell of origin can influence a line's differentiation propensity [2].
- Differentiation Protocol Inconsistencies: The efficiency and outcome of neural differentiation are highly sensitive to minor variations in protocol execution. The use of diverse differentiation protocols (e.g., small molecule-based vs. transcription factor-based) generates neural cells with differing identities, maturity, and purity [1] [2]. This is particularly critical for high-content imaging, as a mixed or impure cellular population can severely skew morphometric analyses.
Table 1: Key Sources of Variability in iPSC-Derived Neural Models
| Source Category | Specific Examples | Impact on Model |
|---|---|---|
| Genetic Background | Donor-specific genetic variation; Expression Quantitative Trait Loci (eQTLs) | Drives 5-46% of phenotypic variation; affects gene expression, differentiation potency [2] |
| Reprogramming & Culture | Somatic mutations; Epigenetic memory of cell of origin; Passage number | Alters genetic stability; influences lineage differentiation bias [2] |
| Differentiation Protocol | Protocol type (e.g., small molecules vs. NGN2 overexpression); Reagent batch variability | Impacts neuronal subtype identity, maturity, and culture purity; can mask disease phenotypes [1] [2] |
Quantitative Assessment of Variability
To control for variability, it must first be quantified. High-content imaging, combined with robust image analysis, provides unbiased metrics to assess the composition and morphology of neural cultures.
High-Content Imaging and Cell Type Identification
Traditional validation methods like flow cytometry are destructive and low-throughput. An advanced alternative employs high-content imaging based on the Cell Painting (CP) assay, which uses fluorescent dyes to label multiple cellular compartments [3]. When combined with Convolutional Neural Networks (CNNs), this approach can identify and classify different cell types (e.g., neural progenitors, postmitotic neurons, microglia) in dense, mixed cultures with an accuracy above 96% [3]. This method is non-destructive, scalable, and provides a powerful tool for quality control before proceeding to more specialized functional assays.
Morphometric Analysis for Neuronal Health and Phenotype
Quantifying neurite outgrowth and branching is a fundamental readout for neuronal development and health. Spatial Light Interference Microscopy (SLIM) is a label-free, quantitative phase imaging technique that allows for long-term, non-destructive measurement of neurite dynamics [4]. The resulting images can be analyzed with semi-automated tracing software like NeuronJ (an ImageJ plugin) to quantify parameters such as total neurite length, number of branches, and growth rates over time [4]. Studies using SLIM have demonstrated that neurite growth rates are highly dependent on cell confluence, with neurons in low-confluence conditions exhibiting significantly higher growth rates than those in medium- or high-confluence conditions [4].
Table 2: Quantitative Metrics for Assessing Neural Cultures
| Assessment Method | Key Measurable Parameters | Significance in Model Validation |
|---|---|---|
| Cell Painting + CNN Classification [3] | Cell type classification accuracy; Proportion of neurons vs. progenitors | Ensures culture composition and purity; critical for reproducible phenotyping |
| SLIM + NeuronJ Tracing [4] | Neurite length (μm); Number of branch points; Growth rate over time | Unbiased, label-free readout of neuronal health, maturation, and network formation |
| Network Science Analysis [5] | Degree centrality; Assortativity coefficient; Clustering coefficient | Reveals self-optimizing topology and information flow capacity of the neuronal network |
Protocols for Mitigating Variability
The following protocols are designed to standardize the generation and analysis of iPSC-derived neural cultures, thereby reducing unwanted variability.
Protocol: Quality Control via Cell Painting and CNN Classification
This protocol provides a workflow for non-destructively validating the composition of a mixed neural culture prior to a dedicated experiment [3].
Workflow Diagram: Cell Type Identification
Materials:
- Research Reagent Solutions:
- Cell Painting Dyes: Mixture of fluorescent dyes staining nuclei, nucleoli, cytoskeleton, Golgi, and endoplasmic reticulum [3].
- Imaging Medium: Phenol-red free culture medium to reduce background fluorescence.
- Fixed Cell Preparation: Cells are typically fixed for compatibility with subsequent assays.
Method:
- Cell Seeding: Plate dissociated neural cells at a defined density in a 384-well microplate suitable for high-content imaging.
- Staining: Follow the standard Cell Painting protocol to stain the cells with the panel of fluorescent dyes.
- Image Acquisition: Use an automated confocal screening microscope to acquire 4-channel images from multiple sites per well.
- Image Analysis:
- Use a deep learning-based algorithm (e.g., a ResNet CNN) for robust cell segmentation, even in dense cultures.
- Train the CNN classifier on a reference set of images with known cell identities.
- Apply the trained model to new images to classify each cell (e.g., neuron, progenitor, microglia).
- Quality Control Decision: Calculate the percentage of the desired cell type. Proceed with the experiment only if the purity meets a pre-defined threshold (e.g., >80%).
Protocol: Label-Free Neurite Outgrowth Quantification
This protocol uses SLIM to non-invasively track the development of neuronal processes over time, providing unbiased morphometric data [4].
Workflow Diagram: Neurite Outgrowth Analysis
Materials:
- Research Reagent Solutions:
- Primary Neurons: Cortical neurons from postnatal (P0-P1) mice [4].
- Coating Reagent: Poly-D-lysine to promote neuronal adhesion.
- Culture Media: Plating and maintenance media as specified in the method.
Method:
- Cell Culture: Plate primary cortical neurons (or iPSC-derived neurons) at defined, low-to-medium confluence on poly-D-lysine-coated glass-bottom dishes. High confluence leads to "shingling" (overlapping neurites), which complicates accurate tracing [4].
- SLIM Imaging: Place the culture dish on the SLIM microscope stage within a environmental chamber (37°C, 5% CO₂). Acquire quantitative phase images every few hours over several days.
- Neurite Tracing with NeuronJ:
- Import the SLIM image sequences into ImageJ with the NeuronJ plugin.
- Manually trace each neurite emerging from the soma, designating them as axons or dendrites based on morphology (axons are longer and of constant diameter; dendrites are thicker and taper).
- Use the batch processing function to extract length measurements for all traced neurites.
- Data Analysis: Calculate the average neurite length per neuron and per neurite over time. Compare growth rates under different experimental conditions. As a reference, neurons in low-confluence conditions show a steady and higher growth rate compared to those in medium-confluence conditions [4].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for iPSC Neural Differentiation and Imaging
| Reagent / Tool | Function | Application Note |
|---|---|---|
| LDN193189 & SB431542 | Small molecule inhibitors for dual SMAD inhibition; induces neural induction [1] | Foundational step in most small molecule-based differentiation protocols. |
| Retinoic Acid (RA) & SHH Agonists | Promotes caudal and ventral patterning toward motor neuron fate [1] | Critical for obtaining specific neuronal subtypes. |
| Cell Painting Dye Panel [3] | Fluorescent dyes for staining multiple organelles to create a morphological fingerprint | Enables high-content screening and unbiased cell classification via CNN. |
| MitoTracker Dyes [6] | Cell-permeant dyes that accumulate in mitochondria based on membrane potential | Allows live-cell imaging of mitochondrial health and dynamics, key in neurodegeneration models. |
| Adeno-Associated Viral (AAV) Vectors [6] | Efficient viral transduction for stable expression of fluorescent reporters (e.g., Mito-GFP) in neurons | Provides specific, high signal-to-noise labeling for long-term studies. |
Variability in iPSC-derived neural models is a formidable but surmountable challenge. By understanding its sources and implementing rigorous quantitative assessments—such as Cell Painting with CNN classification for purity and SLIM imaging for neurite outgrowth—researchers can significantly enhance the reliability of their models. The protocols and tools detailed herein provide a practical framework for standardizing cultures. Adopting these strategies is crucial for generating biologically relevant, reproducible data that can accelerate the discovery of therapeutics for neurological diseases.
Why Traditional Cell Validation Methods Fall Short
In the rapidly advancing field of mixed neural culture research, the limitations of traditional cell validation methods have become a critical bottleneck. Techniques such as sequencing, flow cytometry, and immunocytochemistry, while valuable, are often low in throughput, costly, and destructive, hindering their utility for comprehensive quality control in complex, heterogeneous cellular systems [3]. This application note details these shortcomings and presents high-content imaging (HCI) and morphological profiling as transformative solutions, providing researchers with robust, scalable, and information-rich alternatives for characterizing cell identity and state in physiologically relevant models.
The Critical Shortcomings of Traditional Validation Methods
Current standards for validating cell culture composition, particularly in induced pluripotent stem cell (iPSC)-derived neural models, rely on a combination of methods. However, these approaches present significant challenges for modern, dense, and mixed culture systems.
Table 1: Limitations of Traditional Cell Validation Methods
| Method | Key Limitations | Impact on Research and Development |
|---|---|---|
| Sequencing [3] | - Lacks spatial and morphological context- Destructive, preventing longitudinal studies- Moderate throughput | Incomplete picture of cellular state; unable to track temporal changes in the same culture. |
| Flow Cytometry [3] [7] | - Requires cell dissociation, losing 2D/3D architectural context- Limited number of simultaneous markers due to spectral overlap- Destructive | Loss of critical information on cell-cell interactions and spatial organization of cell types. |
| Immunocytochemistry (ICC) [7] | - Typically low-throughput and labor-intensive- Subjective or semi-quantitative analysis- Multiplexing capability is limited | Low scalability for screening applications; introduces user bias; difficult to profile many targets at once. |
These limitations are particularly problematic for iPSC-derived neural cultures, where genetic drift, clonal variation, and differentiation protocol inconsistencies lead to significant variability in the final cellular composition [3]. The inability to perform rapid, non-destructive quality control hinders experimental reproducibility and the reliable use of these models in systematic drug screening pipelines [3] [7].
High-Content Imaging as a Transformative Solution
High-content imaging (HCI) overcomes these barriers by combining automated microscopy with multi-parametric image analysis to quantify cellular and subcellular features in a high-throughput manner [8] [9]. Unlike traditional methods, HCI preserves the spatial context of cells and can be applied to the same sample over time for live-cell imaging.
Key Technological Advantages
- Multiplexed Morphological Profiling: Assays like Cell Painting use a panel of fluorescent dyes to label multiple organelles (e.g., nucleus, endoplasmic reticulum, mitochondria, Golgi apparatus, cytoskeleton), generating thousands of quantitative morphological features per cell [3] [10]. This creates a unique "morphotextural fingerprint" for different cell types and states.
- Single-Cell Resolution in Complex Cultures: Advanced image analysis software (e.g., CellProfiler) and machine learning models, particularly Convolutional Neural Networks (CNNs), can segment and classify individual cells even in dense, mixed cultures with high accuracy (exceeding 96% in benchmark studies) [3].
- Scalability and Automation: Fully integrated HCS platforms enable the automated acquisition and analysis of images from 384-well or 1536-well microplates, making it feasible to profile hundreds of conditions in a single experiment [11] [12].
Table 2: Quantitative Performance of HCI vs. Traditional Methods in Neural Cultures
| Application Context | HCI Approach | Reported Performance | Traditional Method Comparison |
|---|---|---|---|
| Cell Line Classification [3] | Cell Painting + Random Forest | F-score: 0.75 ± 0.01 | N/A - Baseline |
| Cell Line Classification [3] | Cell Painting + Convolutional Neural Network | Accuracy > 96% | Significant improvement over RF classifier |
| iPSC Neural Culture QC [3] | Regionally-restricted morphological profiling | 96% prediction accuracy | Outperformed population-level classification (86% accuracy) |
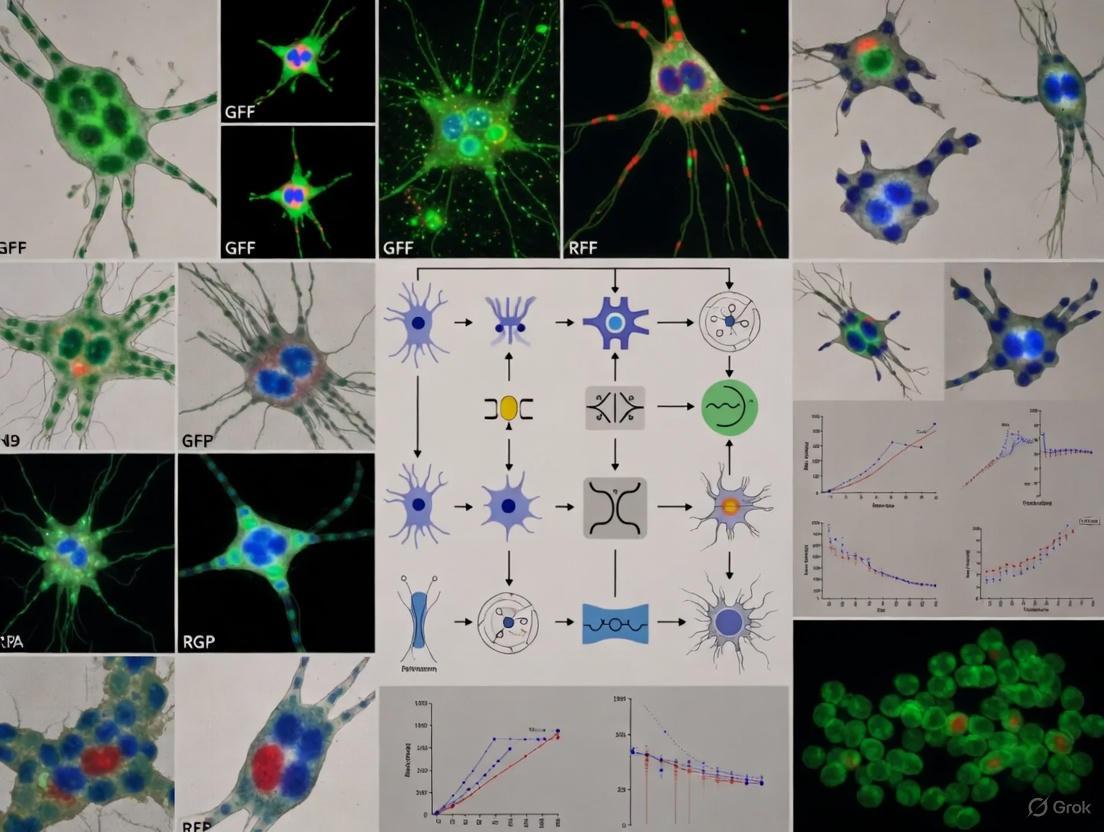
The following workflow diagram illustrates a typical high-content imaging and analysis pipeline for cell validation in mixed neural cultures:
Detailed Experimental Protocol: Cell Type Identification in Mixed Neural Cultures via NeuroPainting
This protocol, adapted from the NeuroPainting assay, is optimized for the morphological profiling of human iPSC-derived neural cell types, including neurons, progenitors, and astrocytes [10].
Research Reagent Solutions
Table 3: Essential Reagents and Materials for NeuroPainting
| Item | Function / Description | Example Catalog Numbers |
|---|---|---|
| CellCarrier-96 Ultra Microplates [7] | 96-well, low-skirted, SBS-footprint plates for imaging | PerkinElmer (6055300) |
| Hoechst 33342 [9] | Stains DNA; labels nuclei for segmentation and analysis. | Thermo Fisher Scientific (H3570) |
| Concanavalin A, Alexa Fluor 488 Conjugate [10] | Labels endoplasmic reticulum (ER). | Thermo Fisher Scientific (C11252) |
| Wheat Germ Agglutinin (WGA), Alexa Fluor 555 Conjugate [10] | Labels plasma membrane and Golgi apparatus. | Thermo Fisher Scientific (W32464) |
| Phalloidin, Alexa Fluor 568 Conjugate [10] | Labels F-actin in the cytoskeleton. | Thermo Fisher Scientific (A12380) |
| SYTO 14 Green Fluorescent Nucleic Acid Stain [10] | Labels nucleoli and cytoplasmic RNA. | Thermo Fisher Scientific (S7576) |
| MitoTracker Deep Red [10] | Labels mitochondria. | Thermo Fisher Scientific (M22426) |
| Automated Imaging System | Confocal, high-content microscope with environmental control. | PerkinElmer Opera Phenix [7] [10] |
| Image Analysis Software | Open-source software for creating custom analysis pipelines. | CellProfiler [6] [10] |
Step-by-Step Procedure
Part 1: Cell Seeding and Fixation
- Plate Preparation: Coat 96-well microplates with an appropriate extracellular matrix (e.g., Poly-L-ornithine/Laminin for neurons) [7].
- Cell Seeding: Seed dissociated iPSC-derived neural cells at optimized densities to ensure healthy morphology without overcrowding [10].
- Neurons: 2,500 cells/well, fix 25 days post-plating.
- Astrocytes: 3,000 cells/well, fix 48 hours post-plating.
- Neural Progenitor Cells (NPCs): 15,000 cells/well, fix 24 hours post-plating [10].
- Fixation: At the desired time point, aspirate the medium and add 4% formaldehyde in PBS for 20 minutes at room temperature.
- Permeabilization and Washing: Wash wells twice with PBS, then permeabilize cells with 0.1% Triton X-100 in PBS for 15 minutes. Wash twice more with PBS.
Part 2: NeuroPainting Staining
- Prepare the staining cocktail in PBS containing 1% BSA with the following dyes [10]:
- Hoechst 33342 (1:2000)
- Concanavalin A, Alexa Fluor 488 (1:200)
- Wheat Germ Agglutinin, Alexa Fluor 555 (1:200)
- Phalloidin, Alexa Fluor 568 (1:200)
- SYTO 14 (1:200)
- MitoTracker Deep Red (1:200)
- Add the staining cocktail to each well and incubate for 30 minutes at room temperature, protected from light.
- Aspirate the cocktail and wash the wells three times with PBS.
- Leave a small volume of PBS in the wells to prevent drying. Seal the plate and proceed to imaging or store at 4°C in the dark.
Part 3: Automated Image Acquisition
- Use a high-content confocal imaging system (e.g., PerkinElmer Opera Phenix) with a 20x or 40x objective [10].
- Define the imaging fields per well to capture a statistically significant number of cells (e.g., 10-20 fields/well for a 96-well plate).
- Acquire images in all fluorescent channels corresponding to the dyes used. Ensure consistent exposure times and laser powers across all plates in a study.
Part 4: Image Analysis and Feature Extraction
- Cell Segmentation: Use a customized CellProfiler pipeline to identify individual cells and subcellular compartments [10].
- Identify nuclei using the Hoechst channel.
- Identify whole-cell regions using the combined membrane (WGA) and cytoskeletal (Phalloidin) signals.
- Feature Extraction: For each segmented cell, extract ~4,000 morphological features describing the size, shape, intensity, and texture of each stained compartment [10].
- Data Preprocessing: Perform well-level averaging and preprocess the data (low-variance filtering, robust standardization, correlation-based feature selection) to yield a final set of ~700 informative morphological features [10].
The following diagram illustrates the logical relationship between the assay output and the analytical steps that lead to biological insight:
Concluding Remarks
Traditional cell validation methods, while foundational, are no longer sufficient for the demands of modern, complex neural culture systems. Their destructive nature, low throughput, and lack of spatial context impede progress in disease modeling and drug discovery. High-content imaging and morphological profiling, as exemplified by the NeuroPainting assay, provide a robust, scalable, and information-rich framework for quantitative cell validation. By adopting these advanced techniques, researchers can achieve unprecedented resolution in characterizing cellular identity and state, ultimately enhancing the reliability and translational potential of their iPSC-based neural models.
High-content imaging (HCI) is a powerful phenotypic screening method that uses automated microscopy to extract quantitative data from cellular images [13]. Unlike conventional assays that measure only one or two features, HCI captures vast amounts of morphological information, making it particularly valuable for detecting subtle phenotypes in complex systems like mixed neural cultures [13] [14].
Cell Painting is a specific, highly multiplexed morphological profiling assay that employs a suite of fluorescent dyes to visualize multiple cellular components simultaneously [13] [15]. By "painting" different organelles and structures, it generates a rich, high-dimensional readout of cellular state. When applied to mixed neural cultures derived from induced pluripotent stem cells (iPSCs), this approach provides a powerful tool for quantifying cell composition and identifying cell types based on their intrinsic "morphotextural fingerprint," even in dense co-cultures [14].
Core Principles of the Technologies
Fundamental Concepts of Morphological Profiling
Morphological profiling involves quantifying hundreds to thousands of morphological features from microscopy images to create a unique fingerprint for each sample or perturbation [13]. This approach is fundamentally unbiased, as it does not target specific pathways, allowing for discoveries unconstrained by prior biological assumptions [13]. The core principle is that biological perturbations—whether chemical, genetic, or disease-related—induce specific, detectable changes in cellular architecture.
- Feature Extraction: Automated image analysis software identifies individual cells and measures approximately 1,500 morphological features, including various measures of size, shape, texture, and intensity [13] [15].
- Profile Comparison: Profiles of cell populations treated with different experimental perturbations are compared to identify biologically relevant similarities and differences [13].
- Single-Cell Resolution: Unlike some profiling methods, morphological profiling with Cell Painting enables analysis at single-cell resolution, allowing detection of perturbations even in subsets of cells within a population [13].
The Cell Painting Assay Mechanism
Cell Painting uses six fluorescent stains imaged in five channels to reveal eight broadly relevant cellular components or organelles [13] [15]. This comprehensive labeling strategy provides a holistic view of cellular morphology.
Figure 1: Cell Painting staining targets and experimental workflow from sample preparation to data analysis.
Application in Mixed Neural Culture Research
Challenges in Neural Culture Characterization
iPSC technology has revolutionized neuroscience by enabling generation of human brain-resident cell types, including neurons, astrocytes, microglia, and oligodendrocytes [14]. However, genetic drift and batch-to-batch heterogeneity cause significant variability in reprogramming and differentiation efficiency, hindering the use of iPSC-derived systems in systematic drug screening or cell therapy pipelines [14]. Traditional validation methods like sequencing, flow cytometry, and immunocytochemistry are often low-throughput, costly, and/or destructive [14].
Cell Painting for Cell Type Identification
Research has demonstrated that Cell Painting can distinguish neural cell types with high accuracy based on their morphological profiles [14]. In one study, traditional morphotextural feature extraction from cells and their nuclei provided sufficient distinctive power to separate astrocyte-derived 1321N1 astrocytoma cells from neural crest-derived SH-SY5Y neuroblastoma cells without replicate bias [14]. The study found that both texture (e.g., energy, homogeneity) and shape (e.g., nuclear area, cellular area) metrics contributed to this separation [14].
Convolutional Neural Networks (CNNs) have proven particularly effective for this application, significantly outperforming random forest classification (96.0% accuracy vs. 71.0%) in cell type prediction [14]. This approach uses image crops centered around individual cells as input rather than relying on extraction of features from segmented cell objects [14]. Gradient-weighted Class Activation Mapping (Grad-CAM) revealed that cell borders, nuclear, and nucleolar signals were the most distinctive features for classification [14].
Robustness in Dense Cultures
A significant advantage for neural culture applications is that nucleocentric morphological profiling maintains accuracy even in very dense cultures [14]. While classification accuracy decreased slightly at 95-100% confluency (92.0% vs. >96% at lower densities), performance remained remarkably robust [14]. This is particularly valuable for iPSC-derived cultures that often reach high densities and form complex cellular networks.
Experimental Protocols
Cell Painting Protocol for Morphological Profiling
The following protocol outlines the standard Cell Painting procedure, with specific considerations for neural culture applications:
- Cell Plating: Plate cells in 96- or 384-well multi-well plates at the desired confluency. For iPSC-derived neural cultures, optimize plating density to account for differentiation time and expected proliferation rates [15].
- Treatment/Perturbation: Apply experimental perturbations via chemical treatments (small molecules at 1-100 μM final concentrations) or genetic modifications. For neural differentiation studies, treatment typically occurs after plating and may extend for 48 hours or longer depending on the biological question [15].
- Fixation and Staining:
- Fix cells with appropriate fixative (e.g., 4% formaldehyde)
- Permeabilize cells (e.g., with 0.1% Triton X-100)
- Stain with the Cell Painting dye cocktail [15]
- Image Acquisition: Acquire images on a high-content screening system. For 5-channel Cell Painting, image acquisition time varies based on samples per well, brightness, and z-dimension sampling [15]. Confocal capabilities may be necessary for thick samples or maximum sensitivity [15].
- Analysis: Use automated software to extract ~1,500 morphological features from each cell. For neural cultures, employ deep learning approaches (CNNs) for optimal cell classification accuracy [14].
Table 1: Cell Painting Staining Protocol Components and Specifications
| Dye Target | Specific Dye Examples | Cellular Compartment | Staining Purpose |
|---|---|---|---|
| Nuclei & Nucleoli | Hoechst 33342 | DNA in nucleus & nucleoli | Segmentation anchor; nuclear morphology & cell cycle [13] [15] |
| Endoplasmic Reticulum | Concanavalin A, Alexa Fluor conjugates | ER membrane | ER organization, distribution & structure [13] |
| Golgi Apparatus & Plasma Membrane | Wheat Germ Agglutinin, Alexa Fluor conjugates | Golgi complex & plasma membrane | Golgi integrity, cell surface features & shape [13] [15] |
| Actin Cytoskeleton | Phalloidin, Alexa Fluor conjugates | Filamentous actin | Cytoskeletal organization, cell shape & motility [15] |
| Mitochondria & RNA | SYTO 14 | Mitochondria & cytoplasmic RNA | Mitochondrial morphology, distribution & metabolic state [13] |
Image Analysis and Feature Extraction Workflow
The image analysis pipeline transforms raw images into quantitative morphological profiles suitable for cell type identification and characterization.
Figure 2: Image analysis workflow from raw data to biological insights in mixed neural cultures.
Quantitative Profiling and Machine Learning Classification
For mixed neural cultures, the analytical approach shifts from population-level profiling to single-cell classification. The process involves:
- Traditional Feature-Based Analysis: Extract standardized morphotextural features from cells and nuclei, then visualize using UMAP for population separation [14].
- Deep Learning Implementation: Use ResNet convolutional neural networks with image crops centered around individual cells as input [14].
- Model Training: Train CNNs with sufficient biological replicates to ensure generalizability. Models trained on multiple replicates outperform those trained on single replicates when predicting instances from a third unseen replicate [14].
- Nucleocentric Approach: For dense neural cultures, use the nuclear region of interest and its immediate environment as input, which maintains accuracy even when whole-cell segmentation becomes challenging [14].
Table 2: Performance Comparison of Cell Analysis Methods in Neural Cultures
| Methodological Aspect | Traditional Feature Extraction + Random Forest | CNN with Whole-Cell Input | Nucleocentric CNN |
|---|---|---|---|
| Overall Classification Accuracy | 71.0±1.0% [14] | 96.0±1.8% [14] | >96% at low-moderate density, 92.0±1.7% at 95-100% confluency [14] |
| Key Differentiating Features | Nuclear texture energy, cellular area, DAPI contrast [14] | Cell borders, nuclear & nucleolar signals [14] | Nuclear & perinuclear morphology |
| Performance in Dense Cultures | Poor (segmentation challenges) | Decreased accuracy | Maintains high accuracy [14] |
| Implementation Complexity | Moderate | High | High |
| Interpretability | High (direct feature analysis) | Low (requires Grad-CAM) | Low (requires Grad-CAM) |
Research Reagent Solutions
Essential materials and reagents for implementing Cell Painting in neural culture research:
Table 3: Essential Research Reagents for Cell Painting Applications
| Reagent Category | Specific Examples | Function in Assay |
|---|---|---|
| Cell Painting Kits | Image-iT Cell Painting Kit [15] | Pre-optimized reagent set containing all necessary fluorescent dyes for standardized implementation |
| Individual Fluorescent Dyes | Hoechst 33342, Concanavalin A Alexa Fluor conjugates, Phalloidin Alexa Fluor conjugates, Wheat Germ Agglutinin Alexa Fluor conjugates, SYTO 14 [13] [15] | Individual stains for specific cellular compartments; allow custom panel configuration |
| Cell Lines | 1321N1 astrocytoma cells, SH-SY5Y neuroblastoma cells [14] | Validation and optimization models for neural cell type identification |
| High-Content Imaging Systems | CellInsight CX7 LZR Pro system, Cellomics systems [15] | Automated microscopy platforms for high-throughput image acquisition of multi-well plates |
| Analysis Software | CellProfiler, Deep learning frameworks (ResNet, CNN) [14] | Open-source and commercial software for image analysis, feature extraction, and classification |
High-content imaging combined with Cell Painting provides a powerful framework for quantitative morphological analysis of complex cellular systems. When applied to mixed neural cultures, this approach enables robust, single-cell classification of neural cell types based on their intrinsic morphotextural fingerprints, even in dense co-cultures where traditional segmentation methods fail. The ability to perform non-destructive, high-throughput quality control of iPSC-derived neural cultures addresses a critical bottleneck in neuroscience research and drug discovery. As deep learning methodologies continue to advance alongside improved staining protocols, morphological profiling promises to become an increasingly valuable tool for characterizing cellular heterogeneity in complex neural systems.
Decoding the Morphotextural Fingerprint of Neural Cells
Application Note
High-content analysis (HCA) represents an advanced technological platform that combines automated microscopy with multi-parametric imaging and analysis to extract quantitative data from cell populations [16] [9]. In the context of neural research, the ability to accurately identify and characterize distinct cell types within mixed neural cultures is crucial for advancing our understanding of neural development, function, and disease mechanisms. Traditional methods for cell type validation, including sequencing, flow cytometry, and immunocytochemistry, are often low in throughput, costly, and destructive [3] [14]. This application note details a robust methodology using high-content image-based morphological profiling to quantitatively and systematically characterize induced pluripotent stem cell (iPSC)-derived mixed neural cultures, achieving exceptional classification accuracy above 96% [3].
The Morphotextural Fingerprint Concept
The term "morphotextural fingerprint" refers to the unique combination of morphological and textural features that can be quantitatively extracted from cellular images to define a specific cell type identity. In neural cell lines, including astrocyte-derived 1321N1 astrocytoma and neural crest-derived SH-SY5Y neuroblastoma cells, this fingerprint manifests as a distinct profile of shape, intensity, and texture metrics across different cellular regions [3]. Representation of standardized feature sets in UMAP space reveals clear separation of neural cell types without replicate bias, demonstrating that cell types can be distinguished across biological replicates based on their unique morphotextural signatures [3] [14]. These fingerprints remain sufficiently distinct even in dense, mixed cultures, enabling reliable cell identity discrimination.
Quantitative Performance Data
Table 1: Performance Comparison of Classification Methods for Neural Cell Identification
| Classification Method | Accuracy | Precision | Recall | Key Advantages | Limitations |
|---|---|---|---|---|---|
| Convolutional Neural Network (CNN) | 96.0 ± 1.8% [14] | High and balanced [14] | High and balanced [14] | Superior accuracy; handles raw image data directly; robust to density variations | "Black box" nature complicates model interpretation |
| Random Forest (RF) | 71.0 ± 1.0% [14] | Imbalanced [14] | Imbalanced (46% misclassification of 1321N1 cells) [14] | Allows feature importance analysis | Poor performance with high-dimensional data; biased feature selection |
Table 2: Culture Density Impact on CNN Classification Accuracy
| Culture Confluency Range | Classification Accuracy | Notes |
|---|---|---|
| 0-80% | No significant decrease [14] | Robust performance across low to high densities |
| 80-95% | Maintained high accuracy [14] | Nucleocentric approach preserves accuracy |
| 95-100% | 92.0 ± 1.7% [14] | Slight decrease due to segmentation challenges |
Table 3: Feature Contributions to Morphotextural Fingerprinting
| Feature Category | Examples | Contribution to Cell Type Separation |
|---|---|---|
| Texture Metrics | Nucleus Channel 3 Energy, Homogeneity [3] [14] | High contribution to UMAP separation |
| Shape Metrics | Cellular Area, Nuclear Area [3] [14] | High contribution to UMAP separation |
| Intensity-related Features | Channel 3 Intensity, Mean/Max/Min Intensity [3] [14] | Less pronounced; more correlated with biological replicate |
Experimental Workflow
Signaling Pathways in Neural Differentiation
Protocols
Protocol 1: Cell Painting and Staining for Mixed Neural Cultures
Purpose: To fluorescently label multiple cellular compartments for comprehensive morphotextural analysis.
Reagents and Materials:
- Cell painting dyes (various fluorophore-conjugated probes)
- Fixed mixed neural cultures on coverslips
- Permeabilization buffer (0.1% Triton X-100 in PBS)
- Blocking solution (1-5% BSA in PBS)
- Mounting medium with DAPI
- Wash buffer (PBS)
Procedure:
- Fixation and Permeabilization: Fix cultures with 4% paraformaldehyde for 15 minutes at room temperature. Permeabilize with 0.1% Triton X-100 in PBS for 10 minutes.
- Blocking: Incubate with blocking solution (1-5% BSA in PBS) for 30-60 minutes to reduce non-specific binding.
- Staining Cocktail Preparation: Prepare cell painting dye cocktail according to manufacturer's instructions. A typical 4-channel confocal imaging setup includes:
- Nuclear stain (e.g., DAPI)
- Cytoplasmic stain (e.g., Phalloidin for F-actin)
- Mitochondrial stain
- Golgi apparatus and endoplasmic reticulum stains
- Staining Incubation: Apply staining cocktail to fixed cultures and incubate overnight at 4°C or for 1-2 hours at room temperature protected from light.
- Washing: Perform three washes with PBS, 5 minutes each with gentle agitation.
- Mounting: Mount coverslips using anti-fade mounting medium. Seal edges with clear nail polish.
- Storage: Store slides at 4°C in the dark until imaging.
Technical Notes: Consistent staining conditions across all samples is critical for comparative analysis. Include appropriate controls for autofluorescence and staining specificity.
Protocol 2: High-content Imaging and Analysis Workflow
Purpose: To acquire and analyze high-content images for morphotextural fingerprint extraction and cell type classification.
Equipment and Software:
- High-content analysis platform (e.g., Thermo Scientific CellInsight CX7 HCA Platform or ArrayScan systems) [16]
- Confocal microscope with automated stage
- Image analysis software (e.g., HCS Studio Cell Analysis Software) [16]
- Computational resources for deep learning (GPU-enabled)
Procedure:
- Instrument Calibration: Calibrate the HCA instrument according to manufacturer specifications. Ensure consistent lighting and focus across all imaging sessions.
- Image Acquisition Setup:
- Configure automated acquisition settings including exposure times, z-stack parameters (if applicable), and field selection.
- Set up plate mapping for systematic sampling across culture conditions.
- Define imaging areas ensuring adequate cell numbers (minimum 500-1000 cells per condition recommended).
- Automated Image Acquisition: Run automated imaging protocol. The CellInsight CX7 platform allows interrogation of multiple sample types with techniques including laser autofocus and confocal acquisition [16].
- Image Pre-processing:
- Perform flat-field correction to account for illumination irregularities.
- Apply background subtraction to enhance signal-to-noise ratio.
- Cell Segmentation:
- Use nuclear markers (DAPI) for primary object identification.
- Apply cytoplasm-based segmentation for whole-cell identification.
- For dense cultures, implement specialized algorithms to separate touching cells.
- Feature Extraction: Extract morphotextural features for each cell across all channels. Key feature categories include:
- Shape features: Area, perimeter, eccentricity, form factor
- Intensity features: Mean, maximum, minimum, standard deviation of pixel intensities
- Texture features: Contrast, correlation, energy, homogeneity (calculated from gray-level co-occurrence matrices)
- Model Training and Classification:
- For CNN approach: Use image crops centered around individual cells as input to ResNet architecture.
- Train with equal sampling of cell numbers per class to avoid bias.
- For optimal performance, include at least 5000 training instances per class [14].
- Validate model performance on independent test sets.
Technical Notes: For dense cultures (>80% confluency), employ nucleocentric profiling by using nuclear ROI and immediate periphery as input to maintain classification accuracy [3] [14].
Protocol 3: Differentiation of hiPSCs into Mixed Neural Cultures
Purpose: To generate mixed cultures of neuronal and glial cells from human induced pluripotent stem cells for morphotextural analysis.
Reagents and Materials:
- hiPSCs (e.g., IMR90-derived hiPSCs)
- mTeSR1 medium with supplements
- hESC-qualified basement membrane matrix
- Neural induction medium (NRI)
- Neuronal differentiation medium (ND)
- Laminin for coating
- Accutase or collagenase for dissociation
Procedure:
- hiPSC Maintenance:
- Culture hiPSCs on qualified matrix-coated dishes in mTeSR1 medium.
- Passage colonies when they reach appropriate size (approximately 1 mm diameter) using manual cutting or enzymatic dissociation.
- Embryoid Body (EB) Formation (Days 0-1):
- Cut undifferentiated hiPSC colonies into fragments of approximately 200 × 200 µm using a syringe with 30G needle.
- Transfer fragments to low-attachment plates to allow EB formation in neural induction medium.
- Neuroectodermal Differentiation (Days 1-7):
- Plate EBs on laminin-coated dishes in neuroepithelial induction (NRI) medium.
- Culture for 7 days, monitoring rosette formation (nestin+, β-III-tubulin+ structures).
- Neural Stem Cell (NSC) Expansion (Days 7-14):
- Mechanically isolate rosettes and dissociate into single cells.
- Replate on laminin-coated dishes in neural induction medium for NSC expansion.
- Terminal Differentiation (Days 14-28):
- Switch to neuronal differentiation medium to promote maturation into mixed neuronal and glial cultures.
- Culture for additional 14 days, with medium changes every 2-3 days.
- Characterization:
- Validate culture composition using immunocytochemistry for neuronal (NF200) and glial (GFAP) markers.
- Perform functional characterization through calcium imaging or other functional assays.
Technical Notes: The entire differentiation process takes approximately 28 days to establish mature mixed neural cultures suitable for morphotextural fingerprint analysis [17].
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Reagents and Materials for Morphotextural Analysis
| Reagent/Material | Function | Example Applications |
|---|---|---|
| Cell Painting Dye Cocktail | Multi-compartment cellular staining | Simultaneous labeling of nucleus, cytoplasm, mitochondria, Golgi, and ER [3] |
| HCS NuclearMask Stains | Nuclear segmentation and identification | Primary object identification in high-content analysis [9] |
| CellROX Reagents | Oxidative stress measurement | Detection of reactive oxygen species in neural cells [16] |
| Click-iT EdU HCS Assays | Cell proliferation analysis | S-phase identification and cell cycle analysis [9] |
| Alexa Fluor Conjugates | High-quality fluorescence labeling | Immunofluorescence and specific protein detection [9] |
| LIVE/DEAD Staining Kits | Cell viability assessment | Viability quantification in high-content screens [9] |
| BacMam Gene Delivery | Targeted fluorescent protein expression | Organelle-specific labeling with Organelle Lights reagents [9] |
| FluxOR Assay Kits | Ion channel screening | Potassium ion channel function analysis [9] |
The application of morphotextural fingerprinting for neural cell identification represents a significant advancement in high-content analysis, enabling unbiased, quantitative classification of cell types in complex mixed cultures. The methodology outlined in this application note, combining cell painting with convolutional neural networks, achieves exceptional classification accuracy above 96% [3] [14], significantly outperforming traditional machine learning approaches. This approach maintains robust performance even in dense cultures through nucleocentric profiling and provides a cost-effective, scalable solution for quality control in iPSC-derived neural culture models. As the field progresses, the integration of these methodologies with advanced 3D culture systems and organoid models will further enhance their relevance for neurodevelopmental studies, disease modeling, and drug discovery applications.
From Pixels to Predictions: Implementing Deep Learning for Automated Cell Classification
High-content imaging represents a powerful paradigm for quantifying cell type and state in complex biological systems. For researchers working with dense, mixed neural cultures derived from induced pluripotent stem cells (iPSCs), this approach is particularly valuable for quality control, as traditional methods like flow cytometry and immunocytochemistry are often low-throughput, costly, and destructive [18]. The integration of specific staining protocols with multi-channel imaging and advanced computational analysis creates a robust framework for unbiased cell identification, achieving classification accuracies exceeding 96% in validation studies [18] [3]. This application note details the essential staining techniques, dye selection, and imaging workflows that underpin successful high-content imaging for neural cell identification.
Staining and Dye Selection for High-Content Imaging
The foundation of effective image-based profiling lies in the strategic selection of fluorescent stains that highlight distinct subcellular compartments. The table below summarizes key dyes and their applications.
Table 1: Essential Stains and Dyes for High-Content Imaging of Neural Cultures
| Reagent | Staining Target | Excitation/Emission (nm) | Key Applications | Notes and Considerations |
|---|---|---|---|---|
| Hoechst 33342 [19] | dsDNA (Nucleus) | ~350/461 | Nuclear counterstain; identification of apoptotic cells (condensed nuclei); cell cycle studies. | Cell-permeant. Known mutagen; handle with care. Fluorescence is quenched by BrdU. |
| Acridine Orange (AO) [20] | Nucleic acids (DNA/RNA) and acidic compartments | Varies by complex | Live-cell imaging; phenotypic profiling; visualization of nuclei and cytoplasmic organelles. | Metachromatic dye; offers a two-channel readout. Enables dynamic, real-time measurements. |
| Cell Painting Dyes [18] | Multiple compartments (e.g., nucleus, cytoplasm, mitochondria) | Multi-channel | Creating a morphological fingerprint for cell type/state identification. | Typically a 4-6 channel assay. Used to distinguish cell types with high fidelity. |
| GCaMP6f [21] | Intracellular Calcium (Ca²⁺) | ~488/510 (GFP-based) | Monitoring functional neuronal activity and maturation in live cells. | Genetically Encoded Calcium Indicator (GECI). Use with neuron-specific promoters (e.g., hSyn) for specificity. |
| LNA/DNA Imaging Probes [22] | Specific proteins (via antibody conjugation) | Varies by fluorophore | Highly multiplexed protein imaging (confocal and super-resolution). | Enables sequential multiplexing of dozens of targets (e.g., synaptic proteins) in the same sample. |
Detailed Experimental Protocols
Protocol 1: Nuclear Staining with Hoechst 33342 for Fixed Cells
This protocol is ideal for providing a fundamental nuclear counterstain in fixed-cell imaging workflows [19].
You will need:
- Cells cultured appropriately for microscopy
- Hoechst 33342, trihydrochloride, trihydrate
- Phosphate-buffered saline (PBS)
- Fluorescence microscope with DAPI filter set
Staining Procedure:
- Prepare Hoechst Stock Solution: Dissolve Hoechst 33342 in deionized water to a final concentration of 10 mg/mL (16.23 mM). Sonicate if necessary to dissolve completely. Aliquot and store at 2–6°C for up to 6 months or at ≤ –20°C for longer storage [19].
- Prepare Staining Solution: Dilute the Hoechst stock solution 1:2,000 in PBS. For example, add 5 µL of stock to 10 mL of PBS.
- Stain Cells: Remove culture medium from cells and add sufficient staining solution to cover them completely.
- Incubate: Protect from light and incubate for 5–10 minutes at room temperature.
- Wash and Image: Remove the staining solution. Wash the cells 3 times with PBS. Image the cells using a microscope equipped with a DAPI filter set.
Protocol Tips:
- Hoechst is a known mutagen and should be handled with appropriate care.
- Dissolving the dye directly in PBS for the stock solution is not recommended; use deionized water instead.
- Over-staining can result in a green haze (emission ~510-540 nm) from unbound dye; optimize concentration for your system [19].
Protocol 2: Live-Cell Morphological Profiling with Acridine Orange
This protocol enables image-based phenotypic profiling in live cells, allowing for the assessment of dynamic processes [20].
You will need:
- Live cells in an appropriate culture medium and vessel
- Acridine Orange (AO) stock solution
- Live-cell imaging compatible microscope with environmental control
Staining and Imaging Procedure:
- Prepare Staining Solution: Dilute Acridine Orange in pre-warmed culture medium or buffer to the working concentration (specific concentration should be optimized for the cell type, e.g., 1-5 µg/mL).
- Stain Cells: Replace the culture medium with the AO staining solution.
- Incubate: Incubate for 15-30 minutes at 37°C and 5% CO₂, protected from light.
- Wash (Optional): For reduced background, the staining solution can be replaced with fresh, pre-warmed medium. However, the dye can also be imaged directly in the staining solution.
- Image: Immediately image live cells using a fluorescence microscope. AO stains nuclei and cytoplasmic organelles, providing a two-channel readout.
Protocol Tips:
- This method is compatible with high-throughput screening and dose-response analyses.
- It is particularly useful for detecting subtle, sublethal phenotypic changes in toxicology and drug discovery [20].
Workflow for Multiplexed Cell Type Identification
The following diagram illustrates the integrated workflow for staining, imaging, and computational analysis for cell identity determination, synthesizing the protocols from the cited research.
The Scientist's Toolkit: Research Reagent Solutions
Successful implementation of these protocols relies on a core set of reagents and tools.
Table 2: Essential Research Reagent Solutions for High-Content Imaging Assays
| Item | Function/Description | Example Use Case |
|---|---|---|
| Hoechst 33342 [19] | Cell-permeant nuclear counterstain for fixed or live cells. | Distinguishing individual cells in dense cultures; identifying condensed apoptotic nuclei. |
| Acridine Orange [20] | Live-cell dye for nucleic acids and acidic compartments. | Phenotypic profiling and dose-response analysis in viable neural cultures. |
| Cell Painting Kit [18] | A standardized panel of dyes targeting multiple organelles to generate a morphological "fingerprint." | Unbiased identification of cell types (e.g., neurons vs. progenitors) in mixed cultures. |
| GCaMP6f AAV (hSyn promoter) [21] | Genetically encoded calcium indicator for monitoring neuronal activity. | Specific measurement of functional maturation in human iPSC-derived neurons over multiple time points. |
| LNA/DNA-PRISM Probes [22] | Diffusible nucleic acid imaging probes for highly multiplexed protein imaging. | Sequential imaging of dozens of synaptic and cytoskeletal proteins in the same neuronal sample. |
| Convolutional Neural Network (CNN) [18] [3] | Deep learning model for high-accuracy cell classification based on raw image crops. | Achieving >96% accuracy in distinguishing neuroblastoma from astrocytoma cells in mixed cultures. |
Data Analysis and Validation
Quantitative Analysis of Staining and Classification
The efficacy of staining and imaging protocols is ultimately quantified through downstream analytical outputs.
Table 3: Quantitative Outcomes from Featured Imaging and Staining Approaches
| Method / Reagent | Key Quantitative Output | Reported Performance | Technical Notes |
|---|---|---|---|
| Hoechst 33342 [19] | Nuclear count, morphology (area, roundness), intensity. | Standard for nuclear segmentation. | Fluorescence intensity can be used for ploidy and cell cycle analysis. |
| Cell Painting + CNN [18] [3] | Single-cell classification accuracy. | >96% accuracy distinguishing cell types in mixed neural cultures. | Outperforms Random Forest classifiers (F-score: 0.75) which rely on hand-crafted features. |
| LNA-PRISM [22] | Number of protein targets imaged in a single sample. | Up to 13-channel confocal imaging of synaptic and cytoskeletal proteins. | Enables correlation analysis of 66 protein co-expression profiles from thousands of synapses. |
| GCaMP6f (AAV2/retro-hSyn) [21] | Specific neuronal transduction and calcium event detection. | Efficient for multi-time point imaging; specific to neurons in mixed cultures. | Allows for functional assessment of network activity during neurodifferentiation. |
Computational Analysis Workflow
The transition from raw images to cell identity involves a critical computational pipeline, the logic of which is shown below.
Convolutional Neural Networks (CNNs) have proven superior to traditional methods like Random Forest classification, which achieved an F-score of only 0.75, largely due to misclassification of one cell type [18]. CNNs use isotropic image crops centered on individual cell nuclei, blanking out the immediate surroundings, to achieve F-scores of 0.96 [18] [3]. Techniques like Grad-CAM can be applied to visualize the morphological features—such as cell borders, nuclear, and nucleolar signals—that the network uses for classification, adding a layer of interpretability [18].
Convolutional Neural Networks (CNNs) vs. Traditional Machine Learning
For researchers in cell type identification, the choice between Convolutional Neural Networks (CNNs) and traditional machine learning is pivotal. Evidence from high-content imaging studies demonstrates that CNNs consistently achieve superior classification accuracy, exceeding 96% in distinguishing neural cell types in dense, mixed cultures [3] [23]. Traditional methods, relying on handcrafted morphotextural features, typically achieve lower performance (e.g., ~75% F-score) and struggle with generalization [3]. The primary trade-off involves data requirements; CNNs require large, varied training datasets to perform optimally, whereas traditional methods with handcrafted features can be more effective with limited data [24]. This application note provides a structured comparison and detailed protocols to guide the selection and implementation of these methods for robust, automated quality control in neural culture research.
Quantitative Performance Comparison
The table below summarizes key performance metrics from relevant studies, highlighting the comparative effectiveness of CNNs and traditional machine learning in biological image analysis.
Table 1: Performance Comparison of CNNs vs. Traditional Machine Learning in Image-Based Classification
| Application Context | Traditional ML (Algorithm, Accuracy/Score) | CNN (Architecture, Accuracy/Score) | Reference |
|---|---|---|---|
| Cell Type Identification in Mixed Neural Cultures | Random Forest (F-score: 0.75) | ResNet-based CNN (F-score: 0.96) | [3] [23] |
| Deep Vein Thrombosis on CT Venography | Extreme Gradient Boost (AUC: 0.975) | VGG16 (AUC: 0.982) | [25] |
| Liver MR Image Adequacy Assessment | Random Forest with Handcrafted Features (Performance superior with small sample sizes) | CNN (Performance superior with large sample sizes; combined approach best) | [24] |
| Ultrasound Breast Lesion Classification | Multiple Traditional Classifiers with Handcrafted Features (Performance lower than deep learning) | Pre-trained CNNs (e.g., ResNet, Inception; Accuracy: ~85-88%) | [26] |
Detailed Experimental Protocols
Protocol 1: Cell Type Identification via CNN-Based Morphological Profiling
This protocol leverages a Cell Painting assay and a ResNet-based CNN for high-accuracy cell classification in dense neural cultures [3] [23].
Sample Preparation and Staining (Cell Painting Assay)
- Culture Setup: Prepare pure and mixed cultures of the target cell types (e.g., iPSC-derived neurons, progenitors, microglia) as well as benchmark cell lines (e.g., SH-SY5Y, 1321N1).
- Staining: Label cells with a standard 4- or 6-channel Cell Painting cocktail. A typical setup includes:
- Hoechst 33342: Nuclei staining.
- Phalloidin: F-actin staining for cytoskeleton.
- Concanavalin A: Glycoproteins staining.
- Wheat Germ Agglutinin: Golgi and plasma membrane.
- SYTO 14: Nucleoli.
High-Content Image Acquisition
- Image the stained cultures using a high-content confocal microscope.
- Acquire images from all relevant channels with consistent exposure settings across all samples and replicates to ensure reproducibility.
Image Preprocessing and Single-Cell Isolation
- Isotropic Crop Generation: For each cell, generate a standardized image crop (e.g., 60 µm x 60 µm) centered on the nucleus [23].
- Data Augmentation: Apply random transformations (rotations, flips, minor intensity variations) to the training dataset to improve model robustness and prevent overfitting.
Convolutional Neural Network Training & Classification
- Architecture Selection: Implement a standard deep learning architecture such as ResNet-50 or ResNet-152 [3] [25].
- Model Training: Train the network using the preprocessed image crops. Use a standard 80/10/10 split for training, validation, and testing.
- Performance Validation: Assess the model using metrics like F-score, precision, and recall on the held-out test set. Use Gradient-weighted Class Activation Mapping (Grad-CAM) to visualize regions of the image most influential for the classification decision, adding interpretability [23].
Protocol 2: Cell Classification Using Traditional Machine Learning with Handcrafted Features
This protocol is suitable for scenarios with limited training data, where handcrafted features can provide a strong baseline performance [3] [24].
Sample Preparation, Staining, and Image Acquisition
- Follow Steps 1 and 2 of Protocol 1.
Cell Segmentation and Feature Extraction
- Cell Segmentation: Use a segmentation algorithm (e.g., marker-controlled watershed) on the nuclear stain channel to identify individual cells [27].
- Region of Interest (ROI) Definition: For each segmented cell, define key ROIs: Nucleus, Cytoplasm, and Whole Cell.
- Handcrafted Feature Calculation: For each ROI and imaging channel, calculate a suite of morphotextural features. These typically include:
Classifier Training and Validation
- Data Preparation: Compile the extracted features into a structured data matrix. Normalize the feature values to have zero mean and unit variance.
- Model Training: Train a traditional machine learning classifier, such as a Random Forest, on the normalized feature matrix.
- Performance Assessment: Validate the classifier performance on a separate test set using standard metrics.
Workflow Visualization
The following diagram illustrates the logical and procedural relationship between the two protocols for cell type identification.
The Scientist's Toolkit: Research Reagent Solutions
The table below details essential materials and reagents for implementing the imaging and analysis workflows described in this note.
Table 2: Key Research Reagents and Materials for High-Content Imaging and Analysis
| Item | Function/Application | Example Use Case |
|---|---|---|
| Cell Painting Assay Kit | A standardized set of fluorescent dyes for multiplexed morphological profiling. | Staining mixed neural cultures to generate rich morphological data for CNN or feature-based classification [3] [23]. |
| Induced Pluripotent Stem Cells (iPSCs) | Patient-specific source material for generating relevant human neural cell types. | Differentiating into neurons, astrocytes, and microglia to create physiologically relevant mixed culture models [3] [23]. |
| High-Content Confocal Microscope | Automated imaging system for acquiring high-resolution, multi-channel z-stack images. | Capturing the detailed morphology of individual cells in dense, mixed cultures for downstream analysis [3] [27]. |
| Marker-Controlled Watershed Algorithm | Image processing technique for segmenting touching cells in an image. | Delineating individual cells in dense cultures based on nuclear staining prior to feature extraction [27]. |
| Pre-trained CNN Models (ResNet, VGG) | Deep learning models with pre-learned feature detectors, adaptable via transfer learning. | Accelerating and improving the training of cell classification models, especially with datasets of moderate size [25] [26]. |
Within the field of neuroscience research, particularly in the study of human neurological disorders and the development of novel therapeutics, the adoption of induced pluripotent stem cell (iPSC)-derived neural cultures has become pivotal. These models recapitulate the cellular heterogeneity of the human brain, but this same complexity presents a significant challenge: the need for precise and reliable identification of constituent cell types, such as neurons, astrocytes, and microglia [3]. Traditional validation methods like flow cytometry or immunocytochemistry are often low-throughput, costly, and destructive, hindering rapid and routine quality control [3].
This application note details a case study demonstrating how high-content imaging and morphological profiling can overcome these limitations. By implementing a method based on cell painting and convolutional neural networks (CNNs), researchers achieved exceptional classification accuracy exceeding 96% for identifying individual cell types within dense, mixed neural cultures [3] [28]. This approach provides a fast, affordable, and scalable solution for quantifying cell composition, thereby enhancing experimental reproducibility and supporting more reliable preclinical screening [3].
Experimental Workflow & Key Findings
The study established an unbiased workflow for cell type identification by combining multiplexed fluorescent imaging with advanced computational analysis. The process begins with the labeling of cultured cells using a modified cell painting (CP) assay, which employs a panel of simple organic dyes to reveal a wealth of morphological information [3]. After high-content confocal imaging, the resulting data is processed through a deep learning pipeline. This involves cell segmentation to identify individual cells in dense cultures, followed by cell type classification using a ResNet-based convolutional neural network (CNN) [3]. This tiered strategy allows for the precise discrimination of not only broad cell types but also distinct cell states, such as activated versus non-activated microglia [3].
Key Quantitative Findings
The implemented methodology was rigorously benchmarked and validated, yielding several key findings with compelling quantitative results, as summarized in the table below.
Table 1: Summary of Key Experimental Findings and Performance Metrics
| Experimental Scenario | Methodology | Key Finding | Reported Accuracy |
|---|---|---|---|
| Benchmarking on Cell Lines [3] | Cell Painting + CNN on SH-SY5Y (neuroblastoma) and 1321N1 (astrocytoma) co-cultures. | Unequivocal discrimination of two distinct neural cell lineages. | >96% classification accuracy |
| Analysis of Dense Cultures [3] | Iterative data erosion, focusing on the nuclear region and its immediate environment. | Regional analysis preserved high prediction accuracy even in very dense cultures. | Equally high accuracy vs. whole-cell analysis |
| iPSC-Differentiation Status [3] | Cell-based profiling of postmitotic neurons vs. neural progenitors. | Significantly outperformed classification based on population-level time in culture. | 96% (cell-based) vs. 86% (time-based) |
| Identification of Microglia [3] | Tiered classification strategy in mixed iPSC-derived neuronal cultures. | Unequivocal discrimination of microglia from neurons; further distinction of microglial reactivity state. | Unequivocal discrimination (high accuracy), lower accuracy for activation state |
A critical insight from the study was that a regionally restricted cell profiling approach, which uses inputs containing the nucleus and its immediate surroundings, achieved classification accuracy as high as an analysis of the whole cell in semi-confluent cultures. Furthermore, this restricted input preserved prediction accuracy exceptionally well in very dense cultures where whole-cell segmentation is challenging [3]. When applied to iPSC-derived neural cultures, this morphological single-cell profiling significantly outperformed a simpler classification based on the time the population had spent in culture, achieving a 96% accuracy versus 86%, respectively [3]. This underscores the power of a single-cell resolution approach over population-level assumptions.
Furthermore, the CNN-based classifier demonstrated superior performance compared to traditional machine learning models. In benchmark tests, a Random Forest (RF) classifier using hand-crafted morphotextural features achieved a comparatively poor F-score of 0.75, largely due to a 46% misclassification rate of one cell type. In contrast, the ResNet CNN surpassed this, enabling the high classification accuracy central to this case study's findings [3].
Detailed Protocols
Cell Painting Assay for Mixed Neural Cultures
This protocol describes the process for staining mixed neural cultures to generate rich morphological data for subsequent image analysis and cell classification [3].
Materials and Reagents
Table 2: Key Research Reagent Solutions for Cell Painting Assay
| Item | Function / Explanation |
|---|---|
| Neural Culture Medium | Supports the survival and health of mixed neural cultures during the assay. Typically based on Neurobasal-A or similar, supplemented with B-27 [29] [30]. |
| Cell Painting Dye Kit | A multiplexed set of fluorescent dyes that target specific cellular compartments (e.g., nuclei, endoplasmic reticulum, Golgi apparatus, cytoskeleton, mitochondria) to generate a morphological "fingerprint" [3]. |
| Formaldehyde (4%) | Fixes the cells, preserving cellular structures and morphology at the time of fixation for subsequent staining and imaging. |
| Triton X-100 | A detergent used to permeabilize the cell membrane, allowing fluorescent dyes to access intracellular targets. |
| Phosphate-Buffered Saline (PBS) | Used for washing steps to remove excess reagents and reduce background fluorescence. |
| Glass-Bottom Culture Plates | Optimal for high-resolution confocal microscopy, providing superior optical clarity for image acquisition. |
Step-by-Step Procedure
- Culture Preparation: Plate mixed neural cultures (e.g., iPSC-derived neurons, astrocytes, and microglia) in glass-bottom multi-well plates. Allow cells to adhere and mature under appropriate culture conditions.
- Fixation:
- Aspirate the culture medium.
- Gently wash the cells once with pre-warmed PBS.
- Add 4% formaldehyde in PBS and incubate for 15-20 minutes at room temperature.
- Aspirate the fixative and wash the cells twice with PBS.
- Permeabilization and Blocking:
- Incubate the fixed cells with a PBS solution containing 0.1% Triton X-100 for 15 minutes.
- Aspirate the permeabilization solution and wash once with PBS.
- (Optional) Incubate with a blocking solution (e.g., 1-2% normal goat serum in PBS) for 30 minutes to reduce non-specific staining.
- Cell Painting Staining:
- Prepare the cocktail of fluorescent dyes according to the established Cell Painting protocol [3].
- Aspirate the PBS (or blocking solution) from the wells and add the dye cocktail.
- Incubate in the dark for 30 minutes at room temperature.
- Aspirate the dye solution and wash the cells thoroughly three times with PBS, ensuring all unbound dye is removed.
- Storage and Imaging: Add a small volume of PBS to prevent the cells from drying out. Seal the plate to protect from light. Image the plates as soon as possible using a high-content or confocal microscope, acquiring images in all relevant fluorescence channels [3].
Image Analysis and Cell Classification Pipeline
This protocol covers the computational workflow for segmenting cells and classifying cell types based on the acquired Cell Painting images.
Computational Tools and Environment
- Software Environment: A standard environment like Python (with Anaconda distribution) is used for running the analysis pipeline [3].
- Deep Learning Framework: Use a framework such as PyTorch or TensorFlow to implement the ResNet architecture for classification.
- Image Analysis Libraries: Libraries like CellProfiler or DeepCell can be used for initial image preprocessing and segmentation.
- High-Performance Computing: Access to a GPU cluster is highly recommended to reduce the time required for training the CNN model.
Step-by-Step Procedure
- Image Preprocessing: Apply standard preprocessing steps to the multi-channel images, such as flat-field correction to illuminate unevenness and background subtraction.
- Cell Segmentation:
- Use a deep learning-based segmentation model (e.g., a pre-trained U-Net or a model from DeepCell) to identify individual cell boundaries, even in dense regions.
- The model generates a mask that outlines each cell (or nuclear region, per the regional restriction finding).
- Feature Extraction & Dataset Creation:
- For a CNN approach, the input is typically the multi-channel image crops of individual cells, as defined by the segmentation masks.
- The dataset is split into training, validation, and test sets, ensuring that cells from the same biological replicate are kept together to prevent data leakage.
- CNN Model Training:
- A ResNet architecture (e.g., ResNet50) is adapted for the multi-channel input.
- The model is trained on the training set using the cell type labels as the ground truth. The validation set is used to monitor for overfitting.
- Data augmentation techniques (e.g., rotations, flips, minor contrast changes) are applied to improve model robustness.
- Cell Type Classification and Validation:
- The trained model is used to predict cell types on the held-out test set.
- Performance is evaluated by calculating accuracy, precision, recall, and F-score.
- Predictions can be validated against known markers via immunofluorescence on a separate set of cultures [3].
Discussion and Application
The high classification accuracy (>96%) achieved through this cell painting and CNN pipeline underscores its significant potential for quality control in iPSC-derived neural culture models [3] [28]. This method provides an unbiased, quantitative, and scalable alternative to traditional, more variable validation techniques.
The primary application of this technology is in preclinical drug screening, where consistent and well-characterized cellular models are crucial for generating reproducible and translatable data. By accurately quantifying the ratio of neurons to progenitors or detecting the presence and activation state of microglia, researchers can better standardize their assays and interpret compound effects [3]. Furthermore, this approach holds promise for cell therapy development, where robust quality control is a prerequisite for safety and regulatory compliance. The ability to perform this analysis without destroying the cultures is a key advantage, allowing for longitudinal studies or subsequent molecular analyses on the same sample [3].
Future directions for this work include extending the classification capabilities to a wider range of neural cell types, such as oligodendrocytes and different neuronal subtypes, and further refining the discrimination of functional states like microglial activation. Integration with other omics data layers could also provide deeper insights into the relationship between cell morphology and molecular function.
The adoption of three-dimensional (3D) neural cell culture models, such as neurospheroids, represents a significant advancement in neuroscience research, as they more accurately replicate the complex architecture, cell organization, and multicellular interactions characteristic of native neural tissue compared to traditional two-dimensional (2D) cultures [31]. However, the complexity of these 3D structures presents distinct challenges for monitoring and analysis. Traditional microelectrode arrays (MEAs) used for electrophysiological recording require external amplification and reference electrodes, limiting system miniaturization [31]. Concurrently, the variability in differentiation outcomes and cellular heterogeneity in induced pluripotent stem cell (iPSC)-derived models necessitates robust quality control methods to ensure experimental reproducibility [3]. This application note details integrated protocols for the functional electrophysiological assessment and high-content morphological analysis of 3D neurospheroids, providing a framework for comprehensive characterization within research and drug development pipelines.
Electrophysiological Monitoring using Organic Charge-Modulated Field Effect Transistors (OCMFETs)
Background and Principle
Organic Charge-Modulated Field Effect Transistors (OCMFETs) present a promising alternative to standard MEAs for monitoring electrical activity in 3D cellular aggregates. Their operation is based on the modulation of transistor channel conductivity induced by the presence of charge on the surface of a sensing area, which is read out as a variation of the device's threshold voltage [31]. A key advantage of this architecture is the physical separation of the sensing area from the organic semiconductor channel, which allows for effective encapsulation and protects the semiconductor from degradation in humid biological environments—a critical feature for long-term cell culture monitoring [31].
Device Fabrication Protocol
Objective: To fabricate ultra-sensitive, flexible OCMFET sensors on plastic substrates for interfacing with neurospheroids. Materials:
- Substrate: Polyethylene terephthalate (PET, 175 μm thickness)
- Metallic Layers: Gold (for floating gate, source, drain, and control gate capacitor)
- Dielectric Layer: Parylene C (200 nm, deposited via Chemical Vapor Deposition - CVD)
- Organic Semiconductor: 6,13-bis(triisopropylsilylethynyl)pentacene (TIPS pentacene) in anisole (1% w/v)
- Equipment: Thermal evaporator, CVD system (e.g., Labcoater 2 SCS PDS 2010), plasma oxygen etcher (e.g., Tucano)
Procedure:
- Substrate Preparation: Clean the PET substrate thoroughly.
- Floating Gate Deposition: Thermally evaporate the first gold layer onto the substrate and pattern it using a low-resolution photolithographic process. This layer serves as the floating gate.
- Dielectric Coating: Deposit a 200 nm Parylene C film across the entire substrate via CVD.
- Source/Drain Electrode Patterning: Evaporate and pattern a second gold layer using a self-alignment process to form interdigitated source and drain electrodes (channel width W = 25 mm, length L = 40 µm, yielding W/L = 625), the upper plate of the control gate capacitor, and connection pads.
- Sensing Area Exposure: Create a via (200 µm x 200 µm) in the Parylene C layer using plasma oxygen etching to expose the sensing area of each OCMFET.
- Semiconductor Application: Drop-cast a 2 µL droplet of TIPS pentacene solution over the transistor channel.
- Device Encapsulation: Cover the channel area with a thick (1.2 µm) layer of Parylene C to enhance stability in humid environments during incubation.
OCMFET Coupling and Neurospheroid Recording
Neurospheroid Generation: [31]
- Cell Line: Utilize an rtTA/Ngn2-positive human induced pluripotent stem cell (hiPSC) line (e.g., GM25256 from the Coriell Institute).
- Differentiation: Differentiate hiPSCs into early-stage excitatory cortical neurons (iNeurons) via doxycycline treatment for 3 days.
- Aggregate Formation: Employ the hanging-drop method to create scaffold-free spherical aggregates. Generate neurospheroids composed of 50,000 iNeurons and astrocytes in a 1:1 ratio.
Recording Setup:
- Couple the prepared neurospheroid directly to the exposed sensing area of the OCMFET device.
- Record the spontaneous electrical activity without the need for a reference electrode in the culture medium.
- The electrical signals from the neurospheroid modulate the charge on the OCMFET's floating gate, leading to a measurable shift in the drain current (I_ds).
Performance Metrics: The OCMFET system has demonstrated the capability to reliably detect spontaneous electrical activity from hiPSC-derived neurospheroids, exhibiting a high signal-to-noise ratio (SNR) [31].
High-Content Imaging for Cell Type Identification in Mixed Neural Cultures
Background and Principle
The inherent variability in iPSC-derived neural cultures necessitates efficient quality control methods. An imaging assay based on Cell Painting and Convolutional Neural Networks (CNNs) has been developed to recognize cell types in dense and mixed cultures with high fidelity [3]. This method leverages high-content imaging and deep learning to provide a fast, cost-effective, and scalable approach for validating culture composition, outperforming traditional methods that are often low-throughput, costly, and destructive [3].
Cell Painting and Staining Protocol
Objective: To stain cells for high-content morphological profiling to distinguish different cell types. Materials:
- Cell Lines: e.g., 1321N1 astrocytoma cells, SH-SY5Y neuroblastoma cells, or iPSC-derived neurons, astrocytes, and microglia.
- Staining Dyes: A panel of fluorescent dyes for Cell Painting (e.g., stains for nuclei, cytoplasm, cytoskeleton, Golgi apparatus, endoplasmic reticulum).
- Fixative: 4% Formaldehyde.
- Permeabilization Agent: 0.5% Triton X-100.
- Antibodies (Optional): For immunostaining, e.g., HuC/HuD for neuronal cell bodies, MAP2 for dendrites.
- Imaging Plates: Nunclon Sphera U-bottom plates for 3D cultures.
- Culture Cells: Grow cells in optimized media (e.g., Gibco B-27 Plus Neuronal Culture System for superior growth) in 2D or 3D formats. For 3D neurospheroids, culture neural stem cells on U-bottom plates to form spheres.
- Fixation: Fix cells with 4% formaldehyde for 15 minutes at room temperature.
- Permeabilization: Permeabilize cells with 0.5% Triton X-100 for 10 minutes.
- Staining: Incubate cells with the Cell Painting dye cocktail according to established protocols. Alternatively, for immunostaining, incubate with primary antibodies (e.g., HuC/HuD, MAP2) followed by appropriate fluorescent secondary antibodies (e.g., Alexa Fluor 555, Alexa Fluor 488).
- Live-Cell Option: For live-cell imaging of 3D cultures, stain with 1 µM Tubulin Tracker Deep Red for 1 hour to label microtubules without fixation.
High-Content Image Acquisition and Analysis
Equipment:
- Imaging Platform: Thermo Scientific CellInsight CX7 LZR HCA Platform or similar 7-laser high-content imager.
- Software: HCS Studio 2.0 Cell Analysis Software or equivalent.
Acquisition Parameters: [32]
- Use a 10x objective for 2D cultures.
- For 3D neurospheroids, use confocal mode. Acquire Z-stacks (e.g., 30 slices at 10 µm intervals) and generate Maximum Intensity Projections (MIPs) for analysis.
Analysis Workflow: [3]
- Segmentation: Use the software's neuronal profiling bioapplication to segment nuclei and cell bodies based on staining.
- Feature Extraction: Calculate morphological and textural features (e.g., area, shape, intensity, texture) for each cell across different channels and regions of interest (nucleus, cytoplasm, whole cell).
- Model Training: Train a CNN (e.g., ResNet architecture) on extracted features from known cell types (monocultures) to create a classification model.
- Classification: Apply the trained model to identify and quantify cell types in unknown, dense, mixed cultures.
Figure 1: High-Content Imaging and Analysis Workflow for Cell Identification.
Performance Data
The table below summarizes the key quantitative results from the application of this morphological profiling approach.
Table 1: Performance Metrics for Cell Type Classification in Neural Cultures
| Classification Task | Method | Accuracy | Key Findings |
|---|---|---|---|
| Neuroblastoma vs. Astrocytoma Cell Lines [3] | Convolutional Neural Network (CNN) | >96% | Exceptional accuracy in distinguishing distinct neural cell lines. |
| Neurons vs. Progenitors (Differentiation Status) [3] | Cell-based Morphological Prediction | 96% | Significantly outperformed classification based on time in culture (86%). |
| Neurons vs. Microglia in Mixed Culture [3] | Morphological Profiling | Unequivocal Discrimination | Microglia could be distinguished from neurons regardless of reactivity state. |
| Activated vs. Non-activated Microglia [3] | Tiered Morphological Strategy | Lower Accuracy | Discrimination was possible but with reduced accuracy compared to broader cell types. |
Essential Research Reagent Solutions
The table below catalogs key materials and reagents essential for conducting 3D neurospheroid imaging and analysis experiments.
Table 2: Essential Research Reagents and Materials
| Item | Function/Application | Example/Notes |
|---|---|---|
| B-27 Plus Neuronal Culture System | Supports enhanced long-term growth and health of 2D and 3D primary neurons [32]. | Superior performance in generating 2D and 3D cultures with increased neurons per field and neurite growth compared to the original B-27 system. |
| Tubulin Tracker Deep Red | A docetaxel-based fluorescent reagent for live-cell labeling of microtubules in neuronal processes [32]. | Enables visualization of neurite outgrowth in live 3D neurospheroids without fixation, compatible with high-content imaging. |
| Cell Painting Dye Cocktail | A set of fluorescent dyes that label multiple cellular compartments to generate a morphological "fingerprint" for cell type identification [3]. | Allows for unbiased classification of cell types in mixed cultures using high-content imaging and machine learning. |
| Nunclon Sphera U-bottom Plates | Low-attachment plates designed for the formation and culture of uniform 3D spheroids and neurospheres [32]. | Facilitates scaffold-free formation of neurospheroids for consistent experimental results. |
| Primary Antibodies (HuC/HuD, MAP2) | Immunostaining of fixed neuronal cultures to identify neuronal cell bodies (HuC/HuD) and dendrites (MAP2) [32]. | Enables quantification of neuronal health, number, and neurite outgrowth in toxicity and growth assays. |
| OCMFET Devices | Ultra-sensitive organic sensors for detecting extracellular electrical activity from electroactive cells like neurons [31]. | Offers advantages like no reference electrode, direct charge amplification, flexibility, and optical transparency. |
The integration of advanced biosensors like OCMFETs for functional electrophysiology with high-content imaging and deep learning for morphological profiling provides a powerful, multi-modal framework for the comprehensive analysis of 3D neurospheroids. The OCMFET platform enables reliable recording of spontaneous electrical activity with a high SNR, offering a simple, low-cost alternative to MEAs [31]. Simultaneously, the cell painting and CNN approach delivers a robust, unbiased method for quality control and cell type identification in complex mixed neural cultures, achieving classification accuracies above 96% [3]. These complementary technologies, supported by optimized culture systems, enhance the physiological relevance and reproducibility of in vitro neural models, thereby accelerating discovery in basic neurobiology and drug development for neurological disorders.
Solving Real-World Challenges: Density, Segmentation, and Data Quality
The adoption of induced pluripotent stem cell (iPSC)-derived neural cultures in preclinical research is hindered by significant challenges in quality control. Traditional methods for validating cell culture composition, such as flow cytometry and immunocytochemistry, are often low-throughput, costly, and destructive [3] [33]. This creates a pressing need for fast, affordable, and scalable quality control approaches to increase experimental reproducibility and cell type specificity [34].
High-content image-based morphological profiling presents a promising solution. This application note details a novel methodology termed "nucleocentric profiling," which combines a modified Cell Painting (CP) assay with convolutional neural networks (CNNs) to achieve unbiased identification of cell types within dense, mixed neural cultures [3] [33]. This strategy is specifically designed to overcome the limitations of whole-cell segmentation in confluent cultures, enabling robust quality control for iPSC-derived models.
Experimental Protocols
Cell Painting Assay for Morphological Profiling
The foundational step of this strategy involves using the Cell Painting assay to generate rich morphological data.
- Staining Protocol: Implement a 4-channel confocal imaging assay. The stains typically include:
- Image Acquisition: Acquire high-content images using a confocal microscope. For miniaturized assays in formats like 96-well plates, systems like the PerkinElmer Operetta CLS can be used with a 20x water-immersion objective [35].
Cell Culture and Sample Preparation
The method was benchmarked and validated using the following models:
- Benchmarking with Cell Lines:
- Application in iPSC-Derived Neural Cultures:
- Differentiation: Differentiate and mature human cortical neurons from iPSC-derived neuronal progenitor cells (NPCs) in a feeder layer-free system. This is performed under physiological O₂ (5%) on poly-L-ornithine/laminin-coated plates [35].
- Culture Optimization: To minimize neuronal clustering and enhance single-cell analysis, optimize NPC seeding densities and incorporate defined extracellular matrices. The use of astrocyte-conditioned medium during differentiation can significantly increase the yield of mature neurons [35].
- Fixation and Staining: Fix cells and process them for the Cell Painting assay or immunofluorescence for validation purposes (e.g., using NeuN as a maturity marker) [35].
Nucleocentric Image Analysis and Workflow
The core innovation of this strategy is the focus on the nuclear region for analysis, which proves more reliable in dense cultures.
- Cell Segmentation and Cropping: Use deep learning-based segmentation to identify individual cell centroids. Generate isotropic image crops (e.g., 60 µm in diameter) centered on each nucleus [33] [34].
- Feature Extraction and Classification:
- Traditional Machine Learning: Extract hand-crafted morphotextural features (describing shape, intensity, and texture) from the nucleus, cytoplasm, and whole cell. These features can be analyzed using algorithms like Random Forest, though this approach demonstrated lower accuracy (F-score: 0.75) [3] [33].
- Deep Learning (Recommended): Train a Convolutional Neural Network (CNN), such as a ResNet model, using the nucleocentric image crops as direct input. This approach achieves superior classification performance (F-score: 0.96) [33] [34].
The following workflow diagram illustrates the complete experimental and computational pipeline.
Key Experimental Data and Validation
Performance Benchmarking of Classification Models
The following table summarizes the quantitative performance of different computational approaches used in the nucleocentric profiling strategy.
Table 1: Performance comparison of cell type classification models [3] [33]
| Classification Model | Input Data | Key Features | Reported Accuracy (F-score) | Strengths | Limitations |
|---|---|---|---|---|---|
| Random Forest (RF) | Hand-crafted morphotextural features | Shape, intensity, and texture from nucleus, cytoplasm, and whole cell | 0.75 ± 0.01 | Model is more interpretable | Poor performance in dense cultures; biased feature selection in high-dimensional data |
| Convolutional Neural Network (CNN) | Raw image crops centered on the nucleus | Nucleus and its immediate surroundings | 0.96 ± 0.01 | High accuracy; robust to culture density; less sensitive to segmentation errors | "Black box" model, harder to interpret |
Impact of Patch Size and Culture Density
A critical validation experiment involved testing how the size of the image crop and the density of the cell culture affect the model's performance.
Table 2: Effect of patch size and culture density on nucleocentric model performance [34]
| Factor | Experimental Variation | Impact on Classification Performance |
|---|---|---|
| Patch Size | Input image crops of varying diameters (e.g., 12 µm to >40 µm) | Performance is largely insensitive to patch sizes within a wide range (e.g., 12-18µm). Very large patches (>40µm) can increase prediction variability. |
| Culture Density | Model applied to semi-confluent to very dense cultures | The nucleocentric model maintains high prediction accuracy even in very dense cultures, where whole-cell segmentation fails. |
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of this strategy requires the following key reagents and computational tools.
Table 3: Essential research reagents and solutions for nucleocentric profiling
| Category | Item | Function / Application | Example / Note |
|---|---|---|---|
| Cell Lines & Culture | iPSC-derived Neural Progenitor Cells (NPCs) | Starting material for generating human cortical neurons | Quality control for NPC markers (Pax6, Sox2) is crucial [35]. |
| Astrocytoma/Neuroblastoma Lines | Benchmarking and validation of the classification method | SH-SY5Y and 1321N1 cells [3]. | |
| Culture Reagents | Poly-L-ornithine / Laminin | Coating substrate for neuronal differentiation and maturation | Essential for promoting neuronal attachment and growth [35]. |
| Specialized Neuronal Media | Differentiation and maintenance of iPSC-derived neurons | e.g., BrainPhys medium supplemented with BDNF, GDNF [35]. | |
| Astrocyte-Conditioned Medium | Enhances neuronal maturation | Increases the percentage of mature (NeuN+) neurons [35]. | |
| Cell Painting Assay | Multiplex Fluorescent Dyes | Staining for various cellular compartments | Includes nuclear, cytoplasmic, ER, Golgi, and F-actin stains [3] [33]. |
| Computational Tools | High-Content Imager | Automated image acquisition | Confocal-capable system (e.g., PerkinElmer Operetta) [35]. |
| Segmentation Software | Identifying individual cell centroids | Deep learning-based tools. | |
| CNN Software/Frameworks | Training and deploying the classification model | e.g., ResNet architecture implemented in Python with PyTorch/TensorFlow [33]. |
Computational Pipeline and Data Analysis
The data analysis involves a tiered strategy to move from raw images to cell type predictions, with a specific model for handling dense cultures.
The following diagram outlines the logical flow of the computational analysis, highlighting the decision point where the nucleocentric model provides its key advantage.
Validation and Performance Metrics
Rigorous validation is essential. The evidence supporting this strategy was rated "exceptional" through eLife's peer-review process [3] [34]. Key validation steps included:
- Cross-Replicate Validation: Models trained on data from multiple biological replicates showed superior performance on unseen data, underscoring the need for varied training data [33].
- Generalization Testing: Models trained on monocultures of single cell types successfully generalized to classify cells in mixed co-cultures [34].
- Application to Complex Tasks: The method was applied to distinguish not only between neurons and microglia but also to discriminate microglial reactivity states with a tiered strategy, albeit with lower accuracy for state classification [33] [34].
The nucleocentric profiling strategy provides a robust, inexpensive, and scalable solution for a major bottleneck in neural culture research. By shifting the analytical focus to the nucleus and its immediate environment and leveraging the power of CNNs, this method achieves high classification accuracy in dense and mixed cultures where traditional methods fail. This approach holds significant promise for standardizing quality control of iPSC-derived neural cultures, thereby enhancing the reliability of disease modeling and preclinical drug screening.
In the field of high-content imaging for cell type identification in mixed neural cultures, robust and generalizable machine learning models are paramount for accurate quantitative analysis. A significant challenge in developing such models is overfitting, where a model performs well on its training data but fails to generalize to new, unseen data. This is particularly prevalent in biological research, where datasets are often limited, expensive to produce, and plagued by class imbalance. This article details a dual strategy integrating advanced data augmentation and ensemble modeling to effectively combat overfitting, thereby enhancing the reliability of cell classification in complex neural networks.
Data Augmentation: A Proactive Defense
Data augmentation artificially expands the diversity and size of a training dataset by applying realistic transformations to existing data. This technique forces the model to learn invariant features, significantly improving its ability to generalize.
Core and Advanced Techniques for Cellular Imaging
The selection of augmentation techniques must be informed by the biological context and the specific challenges of high-content cellular imagery [36] [37]. The following techniques are particularly relevant:
- Geometric Transformations: These include rotation, flipping, scaling, and translation. They help the model become invariant to the orientation and position of cells within an image, which is crucial for cultures where cells can adopt arbitrary orientations [36].
- Color and Illumination Adjustments: Techniques such as color jitter (adjusting brightness, contrast, saturation, and hue) and adding Gaussian noise simulate the variations in staining intensity, lighting conditions, and microscope settings that occur across different experimental batches [36] [37].
- Advanced and Generative Methods:
- CutMix and MixUp: These methods combine two or more images and their corresponding labels. CutMix replaces a random patch of one image with a patch from another, encouraging the model to focus on local features and not just global context. This has been shown to boost performance in object detection and classification tasks [37].
- Generative Adversarial Networks (GANs): GANs can generate highly realistic synthetic cell images, which is invaluable for simulating rare cell types or states, thereby addressing class imbalance. Their application is common in medical imaging and rare class expansion [37].
- Novel Occlusion and Masking: Inspired by real-world scenarios, these techniques involve occluding parts of an image with random patches or structured masks (e.g., horizontal, vertical, or circular stripes). This forces the model to learn from partial information and prevents it from over-relying on any single feature in the image [38].
Quantitative Impact of Data Augmentation
The table below summarizes the documented performance gains from implementing a systematic data augmentation pipeline in machine vision tasks, which are directly applicable to high-content imaging.
Table 1: Performance Impact of Data Augmentation Pipelines
| Metric | Improvement with Data Augmentation | Context / Dataset |
|---|---|---|
| Model Accuracy | Increase of 5-10% | General machine vision systems [36] |
| Overfitting Reduction | Up to 30% reduction | General machine vision systems [36] |
| Object Detection Accuracy | Improvement of over 50% (Precision: +14%, Recall: +1%) | Case study on object detection [36] |
| Image Classification Accuracy | 23% accuracy increase vs. basic flips/rotations | Tech product photo recognition (5,000 images) [37] |
| Multilingual Intent Classification (F1) | 12% F1 score boost | Text augmentation via back-translation [37] |
Ensemble Models: A Collective Defense
Ensemble learning combines predictions from multiple diverse models to produce a single, more robust and accurate prediction. This approach reduces variance and mitigates the risk of relying on a single, potentially overfitted model.
Ensemble Strategies for Robust Classification
- Diverse Base Models: The power of an ensemble lies in the diversity of its constituents. This can be achieved by using different model architectures (e.g., EfficiencyNet, ResNet, DenseNet) or by training the same architecture on different subsets or views of the augmented data [39].
- Averaging and Weighted Averaging: A simple yet effective method where the final prediction is the average (or weighted average based on validation performance) of the probabilities output by each model in the ensemble.
- Stacking (Meta-Learning): A more advanced technique where a meta-model is trained to learn how to best combine the predictions of the base models. The base models make predictions on a validation set, and these predictions are used as features to train the meta-model.
Integrated Application Notes & Protocols
This section provides a detailed, actionable protocol for implementing the dual strategy of data augmentation and ensemble modeling in the context of high-content imaging for mixed neural cultures, as exemplified by cell painting assays [3].
Experimental Workflow
The following diagram illustrates the integrated workflow from raw image data to a validated, robust model.
Detailed Protocol: Augmentation for Cell Type Classification
Objective: To improve the generalization and accuracy of a convolutional neural network (CNN) for classifying cell types (e.g., neurons, astrocytes, microglia) in dense, mixed neural cultures derived from induced pluripotent stem cells (iPSCs) using a cell painting assay [3].
Materials & Reagent Solutions:
Table 2: Essential Research Reagents and Materials
| Item | Function / Explanation in Context |
|---|---|
| Cell Painting Dyes | A panel of fluorescent dyes (e.g., for nucleus, endoplasmic reticulum, cytoskeleton) used to generate multidimensional morphological profiles for cell type discrimination [3]. |
| iPSC-Derived Neural Cultures | The biologically relevant model system containing a mixture of neural cell types (neurons, progenitors, glia) for which classification is required [3]. |
| High-Content Imaging System | A high-resolution, automated microscope (e.g., confocal) capable of capturing multi-channel images for cell painting and subsequent quantitative analysis [3]. |
| Albumentations / Torchvision | Python libraries providing a high-performance interface for implementing a wide range of image augmentation techniques during model training [37]. |
| PyTorch / TensorFlow | Deep learning frameworks used to build, train, and manage the CNN models and ensemble architectures. |
Step-by-Step Methodology:
Baseline Model Training:
- Train a baseline CNN (e.g., ResNet-50 or EfficientNet-B0) on the original, non-augmented training dataset.
- Evaluate the model on a held-out validation set to establish a performance baseline. Monitor the gap between training and validation accuracy as an initial indicator of overfitting.
Design and Implementation of Augmentation Pipeline:
- Construct a sequential pipeline of transformations. The following is an example code snippet using PyTorch [36]:
- For more advanced techniques like CutMix, specialized implementations would be integrated into the training loop.
Train Models with Augmentation:
- Retrain the baseline model from Step 1, this time applying the augmentation pipeline to each training batch on-the-fly.
- It is critical that augmentations are applied only to the training data. The validation set must remain unaltered to provide a fair evaluation.
Construct and Train the Ensemble:
- Generate Diversity: Train 3-5 different CNN architectures (e.g., EfficientNet-B4, ResNet-50, DenseNet-169) using the augmented training data from Step 3. Studies have shown these architectures to be high-performing in medical image analysis [39].
- Aggregate Predictions: For a given input image from the validation set, collect the classification predictions (probabilities) from all trained models.
- Final Prediction: Compute the final prediction by taking the average of the probabilities from all models and selecting the class with the highest average probability.
Rigorous Evaluation and Ablation:
- Compare the performance of the Augmented Ensemble Model against the Baseline Model and the Single Augmented Model.
- Key Metrics: Track accuracy, precision, recall, F1-score, and area under the curve (AUC). Crucially, monitor the consistency between training and validation performance; a narrowed gap indicates reduced overfitting.
- Ablation Study: Systematically remove individual augmentation techniques from the pipeline to identify which transformations contribute most to the performance gains [37]. This informs future refinements.
The combination of data augmentation and ensemble modeling presents a powerful, synergistic defense against overfitting. Augmentation increases the effective diversity of the training data, while ensemble methods leverage the "wisdom of the crowd" to smooth out errors from any single model. As demonstrated in Table 1, this approach can lead to substantial improvements in accuracy and robustness.
For researchers in cell type identification, this strategy is particularly valuable. It enhances the reliability of models trained on inherently variable and limited biological data, ensuring that predictions on new, unseen cultures—a critical step in drug development and basic research—are accurate and trustworthy. By implementing the detailed protocols outlined above, scientists can build more generalizable AI tools that accelerate discovery in neuroscience and beyond.
Gradient-weighted Class Activation Mapping (Grad-CAM) has emerged as a crucial technique for interpreting convolutional neural networks (CNNs) in biological research, transforming these models from opaque "black boxes" into transparent tools that provide visual explanations for their predictions. As a model-specific interpretability technique, Grad-CAM generates heatmaps that highlight the regions of an input image that most significantly contribute to a model's decision-making process. This capability is particularly valuable in high-content imaging applications, where understanding why a model classifies a cell as a specific type is as important as the classification itself [40] [41] [42].
The fundamental principle behind Grad-CAM involves leveraging the gradients of any target concept (e.g., a specific cell type) flowing into the final convolutional layer of a CNN to produce a localization map. This approach preserves model architecture without requiring architectural modifications or retraining, making it highly adaptable to various deep learning models used in biological image analysis [41] [42]. Within the context of high-content imaging for cell type identification in mixed neural cultures, Grad-CAM provides researchers with visual evidence of which cellular morphological features—such as nuclear shape, cytoplasmic extensions, or textural patterns—the network utilizes to distinguish between different cell types [3] [23].
Application in Cell Type Identification
Case Study: iPSC-Derived Neural Cultures
A significant application of Grad-CAM in high-content imaging is for quality control of induced pluripotent stem cell (iPSC)-derived neural cultures. Researchers have successfully implemented a workflow combining Cell Painting assays with CNN classification and Grad-CAM visualization to recognize cell types in dense, mixed cultures with remarkable fidelity. In benchmark tests using pure and mixed cultures of neuroblastoma and astrocytoma cell lines, this approach achieved classification accuracy exceeding 96%, significantly outperforming traditional random forest classifiers (F-score: 0.75±0.01) [3] [23].
Through iterative data erosion experiments, researchers made a crucial discovery: inputs containing only the nuclear region of interest and its immediate environment achieved classification accuracy equivalent to inputs containing the whole cell for semi-confluent cultures. This finding indicates that CNNs primarily utilize nuclear and perinuclear morphological features for classification, which has profound implications for assay design in high-density cultures where whole-cell segmentation is challenging [3] [28]. When applied to iPSC-derived neural cultures, this regionally restricted cell profiling approach successfully evaluated differentiation status by determining the ratio of postmitotic neurons to neural progenitors, with cell-based prediction (96% accuracy) significantly outperforming population-level time-in-culture classification (86% accuracy) [23] [28].
Case Study: Single-Cell Morphological Profiling in 3D Cell Painting
The application of Grad-CAM has revealed critical insights into potential pitfalls in deep learning-based single-cell morphological profiling. When researchers applied Grad-CAM to analyze 3D Cell Painting images of single cells, they discovered that supervised models can exploit biologically irrelevant pixels—such as background noise—when extracting morphological features from images. This finding raises significant concerns about the biological relevance of learned single-cell representations in downstream analyses [43].
To address this limitation, researchers developed Grad-CAMO (Grad-CAM Overlap), a novel interpretability score that quantifies the proportion of a model's attention concentrated on the cell of interest versus the background. This metric can be assessed per-cell or averaged across validation sets, providing a crucial auditing tool for evaluating the biological relevance of extracted features. In experiments with 3D Cell Painting data, Grad-CAMO revealed that only 30% of learned morphological profiles had Grad-CAM localization maps that meaningfully overlapped with cell segmentation masks, highlighting the prevalence of models relying on spurious correlations [43].
Table 1: Quantitative Performance of Grad-CAM-Informed Methods in Biological Applications
| Application Context | Classification Accuracy | Comparative Method Performance | Key Improvement |
|---|---|---|---|
| iPSC-derived neural culture classification | 96% [3] [23] | Random Forest: F-score 0.75±0.01 [3] | More balanced recall and precision |
| Nuclear vs. whole-cell profiling | Equivalent accuracy [3] | Preserved prediction accuracy in dense cultures | Enables analysis in challenging segmentation conditions |
| Cell-based vs. population-level prediction | 96% vs. 86% [23] [28] | Time-in-culture classification | More accurate differentiation status assessment |
Experimental Protocols
Protocol 1: Cell Type Identification in Mixed Neural Cultures
Objective: To implement a Grad-CAM-enabled workflow for identifying and validating cell types in mixed neural cultures derived from iPSCs.
Materials and Reagents:
- Cell Painting dyes: Multiplexed fluorescent dyes for staining cellular compartments [3] [28]
- Fixed cell samples: iPSC-derived neural cultures fixed in 4% PFA
- Imaging plates: 96-well glass-bottom plates suitable for high-content microscopy
- Confocal microscope: Equipped with high numerical aperture objectives and appropriate filter sets [44]
Methodology:
- Cell Staining and Imaging:
Image Preprocessing and Segmentation:
- Generate isotropic image crops of 60μm centered on individual cell centroids [23]
- Apply segmentation algorithms to identify single-cell regions of interest
- Create a dataset with balanced representation of all expected cell types
CNN Model Training:
- Implement a ResNet architecture pre-trained on ImageNet [3] [23]
- Fine-tune the model using transfer learning with your single-cell image dataset
- Apply data augmentation techniques (rotation, flipping, brightness adjustment) to improve model generalization
- Split data into training (70%), validation (15%), and test (15%) sets
Grad-CAM Implementation:
- Extract activations from the final convolutional layer of the trained model
- Compute gradients of the top predicted class score with respect to the feature maps
- Calculate neuron importance weights through global average pooling of gradients
- Generate heatmap by applying a weighted combination of feature maps followed by ReLU activation [42]
Result Interpretation:
- Overlay Grad-CAM heatmaps on original images to visualize discriminative regions
- Compare heatmap localizations with known biological markers for validation
- Calculate nuclear vs. cytoplasmic attention ratios to assess feature extraction relevance
Protocol 2: Quality Assessment with Grad-CAMO
Objective: To implement Grad-CAMO for quantifying the biological relevance of morphological profiles extracted by supervised models.
Materials and Reagents:
- 3D Cell Painting images: Fluorescence microscopy z-stacks of single cells [43]
- Segmentation masks: Binary masks identifying cell boundaries
- Computational environment: Python with PyTorch/TensorFlow and custom Grad-CAMO implementation
Methodology:
- Model Training for Feature Extraction:
- Train a 3D convolutional neural network to predict treatment labels from single-cell 3D crops
- Use intermediate activations as learned morphological profiles [43]
Grad-CAM Calculation:
- Compute Grad-CAM localization maps for the trained model
- Use the gradients of the predicted treatment score flowing into the final 3D convolutional layer
Grad-CAMO Metric Computation:
- Calculate the overlap between Grad-CAM attention maps and cell segmentation masks
- Quantify the proportion of model attention focused on the cell of interest versus background
- Apply the formula: Grad-CAMO = (Overlap Area) / (Total Attention Area) [43]
Profile Categorization:
- Classify morphological profiles as biologically relevant (Grad-CAMO > threshold) or potentially confounded (Grad-CAMO ≤ threshold)
- Set threshold based on validation experiments with known ground truth
Model Optimization:
- Iterate on model architecture and training strategies to improve Grad-CAMO scores
- Use Grad-CAMO as a regularization metric during model selection
Table 2: Research Reagent Solutions for Grad-CAM Experiments
| Reagent/Software | Function in Protocol | Application Notes |
|---|---|---|
| Cell Painting Dye Kit | Multiplexed staining of cellular compartments | Enables morphological profiling by highlighting organelles [3] |
| High-Content Imaging System (e.g., Operetta CLS) | Automated image acquisition | Provides consistent imaging across large datasets [44] |
| CellProfiler | Image analysis and segmentation | Open-source alternative for cell segmentation [44] |
| ResNet Architecture | CNN backbone for classification | Pre-trained models available for transfer learning [3] [23] |
| TensorFlow/PyTorch with Grad-CAM implementation | Heatmap generation | Customizable code for various model architectures [42] |
Technical Considerations and Optimization
Grad-CAM Implementation Details
Successful implementation of Grad-CAM in biological image analysis requires careful consideration of several technical factors. The choice of target convolutional layer significantly impacts the resolution and semantic level of the generated explanations. Later layers typically provide more semantically meaningful but coarser visualizations, while earlier layers offer higher spatial resolution with less semantic meaning. For single-cell classification in neural cultures, targeting the last convolutional layer often provides the optimal balance [41] [42].
The Grad-CAM process can be mathematically represented as:
- Compute gradients of class score yᶜ with respect to feature map activations Aᵏ of the target layer: ∂yᶜ/∂Aᵏ
- Calculate neuron importance weights αₖᶜ through global average pooling: αₖᶜ = (1/Z) * Σᵢ Σⱼ (∂yᶜ/∂Aᵏᵢⱼ)
- Generate coarse localization map by combining weighted feature maps: Lᶜ = ReLU(Σₖ αₖᶜ Aᵏ) [41] [42]
For enhanced visualization, Guided Grad-CAM combines the class-discriminative properties of Grad-CAM with the pixel-space granularity of Guided Backpropagation, producing higher-resolution visualizations that highlight fine morphological details relevant to cell type identification [41].
Methodological Validation
Rigorous validation is essential when interpreting Grad-CAM results in biological contexts. Researchers should employ iterative ablation studies to verify that regions highlighted by Grad-CAM align with biologically meaningful features. In the context of neural culture analysis, this may involve comparing Grad-CAM heatmaps with immunohistochemical markers for specific cell types [3] [23].
Additionally, quantitative assessment of Grad-CAM explanations should be incorporated into the workflow. The Grad-CAMO metric provides a valuable tool for this purpose, enabling researchers to audit whether models focus on biologically relevant regions rather than exploiting confounding factors in image data [43]. When evaluating multiple models, researchers should compare both classification accuracy and explanation quality to select the most biologically plausible model.
Grad-CAM provides a powerful framework for enhancing the interpretability of deep learning models in biological image analysis, particularly for high-content imaging applications in mixed neural cultures. By generating visual explanations that highlight morphological features driving classification decisions, Grad-CAM helps bridge the gap between model predictions and biological understanding. The integration of quantitative assessment metrics like Grad-CAMO further strengthens this approach by enabling researchers to audit the biological relevance of learned features. As deep learning continues to transform biological image analysis, explainability techniques like Grad-CAM will play an increasingly vital role in building researcher trust, validating model performance, and extracting biologically meaningful insights from complex cellular systems.
High-content imaging (HCI) has emerged as a powerful tool for quantifying complex cellular phenotypes in biomedical research. Within the specific context of mixed neural cultures derived from induced pluripotent stem cells (iPSCs), the ability to automatically identify and characterize diverse cell types is crucial for applications in disease modeling and drug development [44]. Traditional validation methods like flow cytometry and immunocytochemistry are often low-throughput, costly, and destructive [3] [23]. This application note details an integrated workflow that combines high-content confocal microscopy with a convolutional neural network (CNN) to achieve unbiased, high-fidelity cell type identification in dense, mixed neural cultures, achieving classification accuracy above 96% [3] [28] [23].
Experimental Protocols
Cell Culture and Staining for Morphological Profiling
This protocol is designed for the quantitative characterization of mixed neural cultures, including those derived from iPSCs.
Key Materials:
- Cell Lines: Pure and mixed cultures of SH-SY5Y neuroblastoma and 1321N1 astrocytoma cell lines for benchmarking; iPSC-derived neural cultures, including postmitotic neurons, neural progenitors, and microglia [3] [23].
- Staining Reagents (Cell Painting Assay): A multiplexed fluorescent dye set is used to label various cellular compartments [3]. Specific dyes are not listed in the provided search results, but a standard Cell Painting assay typically includes probes for nuclei, cytoplasm, endoplasmic reticulum, mitochondria, Golgi apparatus, and RNA [44].
- Imaging Plates: 96-well or 384-well plates suitable for high-content imaging systems.
Procedure:
- Culture and Differentiation: Plate cells in appropriate multi-well imaging plates. For iPSCs, differentiate into the desired neural lineages (e.g., neurons, neural progenitors, microglia) using established protocols [3] [45].
- Fixation and Staining: At the desired time point, fix cells and perform the Cell Painting protocol using the multiplexed fluorescent dyes [3].
- High-Content Imaging: Image the stained plates using a confocal microscope system equipped for high-throughput, such as an Opera High-Content Screening System or similar [44]. Acquire images with a 40x or higher objective to capture subcellular morphological details. For the referenced study, 4-channel confocal imaging was implemented [3] [23].
Image Analysis and Deep Learning Classification
This protocol outlines the steps for creating a dataset and training a CNN to classify cell types based on their morphological fingerprints.
Key Materials:
- Software: Python with deep learning libraries (e.g., PyTorch, TensorFlow); Image analysis software such as CellProfiler or ImageJ for initial segmentation [44].
- Computing Hardware: A computer with a high-performance GPU is recommended for efficient CNN training.
Procedure:
- Cell Segmentation and Image Extraction: Use a segmentation algorithm to identify individual cells and their centroids. Extract isotropic image crops (e.g., 60 µm x 60 µm) centered on each cell centroid [3] [23].
- Dataset Curation: Manually curate and label the extracted image crops to create a ground-truth dataset for training. It is critical to include data from multiple biological replicates to ensure the model can generalize well [23].
- CNN Model Training: Train a ResNet-based CNN architecture using the curated dataset. The model learns to map the input image directly to a cell type classification [3] [23].
- Model Validation: Evaluate the trained model on an independent, unseen test set to determine its final classification accuracy, precision, and recall [23].
- Model Interpretation (Optional): Use techniques like Gradient-weighted Class Activation Mapping (Grad-CAM) to visualize which regions of the input image were most influential in the model's decision [23].
Results and Data Analysis
Quantitative Benchmarking of Classification Performance
The following table summarizes the key quantitative findings from the referenced study, comparing the performance of traditional machine learning with a deep learning approach.
Table 1: Performance Benchmarking of Cell Classification Methods in Neural Cultures
| Method | Feature Extraction | Classifier | Classification Accuracy (F-score) | Key Findings |
|---|---|---|---|---|
| Traditional Machine Learning | Hand-crafted morphotextural features (shape, intensity, texture) | Random Forest | 0.75 ± 0.01 | Significant misclassification (46%) of one cell type despite clear population separation in UMAP plots [3] [23]. |
| Deep Learning | Direct from image crops | Convolutional Neural Network (CNN/ResNet) | 0.96 ± 0.01 | High, balanced precision and recall; outperformed Random Forest even with limited training data [3] [23]. |
| Regional Analysis (Deep Learning) | Nuclear region & close environment | Convolutional Neural Network (CNN) | ~0.96 | Accuracy preserved in semi-confluent and very dense cultures, enabling analysis where whole-cell segmentation is challenging [3] [28]. |
The study demonstrated that a tiered classification strategy could first discriminate microglia from neurons with high accuracy, and then further distinguish microglial activation states, albeit with lower performance [28] [23]. Furthermore, this cell-based morphological profiling significantly outperformed (96% accuracy) a simpler classification method based solely on the population-level time in culture (86% accuracy) for determining neuronal differentiation status [3].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Research Reagent Solutions for High-Content Imaging of Neural Cultures
| Item | Function/Application | Specific Examples / Notes |
|---|---|---|
| iPSC Lines | Starting material for generating patient-specific neural cells. | Requires robust quality control and standardized differentiation protocols [3] [44]. |
| Neural Differentiation Kits | Directs iPSCs toward specific neural fates (e.g., cortical neurons, astrocytes). | Protocols often use transcription factor overexpression (e.g., Neurogenin-2) or small molecules [45]. |
| Cell Painting Dye Kits | Multiplexed fluorescent labeling of cellular compartments for morphological profiling. | Standard kits target nucleus, cytoplasm, ER, mitochondria, Golgi, and RNA [3] [44]. |
| Specialized Culture Media | Supports neuronal health and maturation; mitigates phototoxicity during live imaging. | Brainphys Imaging medium is designed to support electrophysiological maturation and reduce ROS in phototoxic environments [45]. |
| Extracellular Matrix (ECM) Coatings | Provides structural and biochemical support for cell adhesion and neurite outgrowth. | Common coatings include Poly-D-Lysine combined with laminin (murine or human-derived, e.g., LN511) [45]. |
| High-Content Imaging System | Automated microscope for acquiring high-resolution images from multi-well plates. | Confocal systems (e.g., Opera Phenix, ImageXpress) are often used for their optical sectioning capabilities [44]. |
Workflow and Pathway Visualization
The following diagram illustrates the integrated experimental and computational pipeline for high-throughput cell identity identification.
Diagram 1: High-Content Cell ID Workflow. This workflow compares two computational analysis paths, demonstrating the superior performance of a deep learning approach over traditional machine learning for classifying cell types in mixed neural cultures.
Discussion
The integration of high-content confocal imaging with a CNN-based analysis pipeline presents a robust solution for the unbiased identification of cell identity in complex, dense neural cultures. The key to this approach's success lies in the CNN's ability to learn discriminative morphological features directly from the image data, bypassing the limitations of manual feature engineering required by traditional methods like Random Forest [3] [23]. This enables the accurate discrimination of not only distinct cell lines but also subtle differences between neuronal progenitor states and mature, postmitotic neurons within iPSC-derived systems [3].
A significant finding is the resilience of this method in high-density cultures. By focusing the analysis on the nuclear region and its immediate proximity, the classification accuracy remains high even when whole-cell segmentation is challenging due to cell confluence [3] [28]. This makes the protocol particularly valuable for quality control in iPSC-derived neural culture models, where variability in differentiation outcomes is a major challenge [44] [23]. The method provides a fast, affordable, and scalable alternative to lower-throughput techniques, accelerating research in neurobiology and drug discovery.
Benchmarking Performance: How HCI Stacks Up Against Gold Standards
Within modern neuroscience research, particularly in the study of complex mixed neural cultures derived from induced pluripotent stem cells (iPSCs), accurate cell type identification is paramount. Traditional methods like immunocytochemistry (ICC) and flow cytometry have long been the standards for cellular analysis. However, high-content imaging (HCI) is emerging as a powerful alternative that combines the strengths of both. This Application Note provides a quantitative comparison of these technologies, demonstrating that HCI achieves classification accuracy exceeding 96% for neural cell types, while offering the unique advantage of single-cell morphological profiling within dense, mixed-population cultures. We detail protocols and reagent solutions to implement this powerful approach for quality control in neural culture models and drug discovery pipelines.
Quantitative Comparison of Technologies
The table below summarizes the key performance metrics and characteristics of HCI, ICC, and flow cytometry for cell type identification in neural cultures.
Table 1: Quantitative and Qualitative Comparison of Cell Analysis Technologies
| Parameter | High-Content Imaging (HCI) | Immunocytochemistry (ICC) | Flow Cytometry |
|---|---|---|---|
| Classification Accuracy | >96% (CNN-based) [3] [14] | High (antibody-dependent) | High (antibody-dependent) |
| Spatial Context | Preserved (single-cell resolution in situ) | Preserved (single-cell resolution in situ) | Lost (cells in suspension) |
| Throughput | High (automated, thousands of cells) | Low to Medium (often manual) | Very High (thousands of cells/second) |
| Multiplexing Capacity | High (4+ channels with standard dyes) [3] [46] | Medium (limited by antibody species) | Very High (>20 parameters) |
| Sample Destructiveness | Non-destructive (live-cell compatible) | Destructive (fixed cells) | Destructive (single-cell suspension required) |
| Key Strength | Unbiased morphological profiling; dense culture analysis [3] | Gold standard for protein localization | High-speed, multiparametric single-cell quantification [47] |
| Primary Limitation | Computational complexity | Low throughput, subjective analysis | No spatial information, requires cell dissociation [47] |
Experimental Protocols
Protocol 1: HCI-Based Cell Painting for Neural Cell Identification
This protocol, adapted from Elife studies, uses a 4-channel Cell Painting assay and convolutional neural networks (CNNs) for unbiased identification of cell types in dense mixed neural cultures [3] [14].
Workflow Diagram: HCI Cell Painting and Analysis
Step-by-Step Procedure
Cell Culture:
- Seed iPSC-derived neural cultures (e.g., mixed neurons and neural progenitors) or control cell lines (e.g., SH-SY5Y neuroblastoma, 1321N1 astrocytoma) into poly-L-ornithine/laminin-coated 96-well Cell-Carrier microplates [35].
- Culture cells under optimal conditions (e.g., 5% CO₂, 5% O₂ for neurons) until desired density is reached [35].
Staining (Cell Painting Assay):
- Fixation: Aspirate medium and fix cells with 4% formaldehyde in PBS for 15-20 minutes at room temperature (RT).
- Staining: Prepare the staining cocktail as detailed in the "Research Reagent Solutions" section. Incubate cells with the dye mixture for 30 minutes at RT protected from light [3].
- Washing: Wash cells 3x with PBS. Leave in PBS for imaging.
Image Acquisition:
- Use a high-content confocal microscope (e.g., PerkinElmer Operetta CLS) with a 20x or higher objective.
- Acquire images in all four fluorescence channels (Hoechst, ConA, WGA, Phalloidin, SYTO) for each field of view [3].
- Automate the process to acquire images from multiple wells and sites, ensuring at least thousands of cells are captured per condition.
Image Analysis and Classification:
- Segmentation: Use integrated software (e.g., Harmony) or custom scripts to identify and segment individual cells and nuclei based on the Hoechst and cytoplasmic markers.
- CNN Training: Prepare training datasets by cropping images around individual cells. Train a ResNet-based CNN architecture using known cell types as labels. For optimal performance, use ~5000 training instances per cell class [14].
- Prediction: Apply the trained CNN model to new, unlabeled images to predict cell identity for each segmented object. Accuracy can be validated against a hold-out test set with known identities.
Protocol 2: Benchmarking Against Immunocytochemistry and Flow Cytometry
To validate HCI classification, results should be compared to established antibody-based methods.
Workflow Diagram: Multi-Technique Validation Strategy
A. Immunocytochemistry Validation
- Procedure: Plate cells on chambered slides or plates. Fix and permeabilize as above. Incubate with primary antibodies (e.g., anti-βIII-tubulin for neurons, anti-GFAP for astrocytes, anti-Ki67 for progenitors) followed by species-appropriate fluorescent secondary antibodies. Counterstain with Hoechst [35].
- Quantification: Manually count positive cells across multiple random fields or use basic image analysis software to determine the percentage of each cell type.
B. Flow Cytometry Validation
- Sample Preparation: Dissociate the mixed neural culture to a single-cell suspension using Accutase or trypsin [47]. Fix and permeabilize cells if intracellular markers are targeted.
- Staining: Incubate cells with fluorescently conjugated antibodies against the same markers used for ICC (e.g., CD56/NCAM for neurons). Include isotype controls.
- Data Acquisition and Analysis: Acquire data on a flow cytometer. Use forward and side scatter to gate on single, viable cells. Determine the percentage of positive cells for each marker using established gating strategies [47] [48].
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for HCI-Based Cell Type Identification
| Reagent | Function / Target | Application Note |
|---|---|---|
| Hoechst 33342 | DNA stain, labels nucleus | Used for nuclear segmentation and cell counting [3] [35]. |
| Concanavalin A (ConA), Alexa Fluor Conjugate | Labels endoplasmic reticulum and Golgi apparatus | One of the core dyes in the Cell Painting assay; provides cytoplasmic texture information [3]. |
| Wheat Germ Agglutinin (WGA), Alexa Fluor Conjugate | Labels plasma membrane and Golgi | Critical for capturing cell shape and boundaries [3]. |
| Phalloidin, Alexa Fluor Conjugate | Labels filamentous actin (F-actin) | Visualizes cytoskeletal structure, a key feature for morphological discrimination [3]. |
| SYTO 14 Green Fluorescent Nucleic Acid Stain | Labels nucleoli and cytoplasmic RNA | Provides contrast for nucleolar morphology and general cytoplasmic content [3]. |
| Anti-βIII-Tubulin (Tuj1) Antibody | Neuronal marker | Standard ICC/flow cytometry validation for post-mitotic neurons [35]. |
| Anti-NeuN Antibody | Mature neuronal nucleus marker | Validates neuronal maturity in iPSC-derived cultures [35]. |
| Anti-Ki67 Antibody | Proliferation marker | Identifies neural progenitor cells in mixed cultures [35]. |
| Poly-L-ornithine / Laminin | Extracellular matrix coating | Essential for adhesion and healthy maturation of iPSC-derived neurons [35]. |
| BrainPhys Neuronal Medium | Serum-free culture medium | Supports synaptic activity and long-term maturation of functional neurons [35]. |
The quantitative data presented herein establishes high-content imaging as a highly accurate and information-rich platform for cell identity quantification in complex neural systems. While immunocytochemistry remains the gold standard for specific protein localization and flow cytometry excels in high-throughput, multi-parameter surface marker screening, HCI offers a unique combination of high spatial resolution and unbiased, single-cell phenotypic profiling.
The critical advantage of HCI is its ability to perform this analysis in situ, preserving the spatial context of cells within dense cultures—a task that is challenging for both traditional ICC analysis and impossible for flow cytometry. The implementation of deep learning, specifically CNNs, overcomes the limitations of traditional machine learning classifiers like Random Forests, enabling the direct use of rich morphological information from image crops to achieve >96% classification accuracy [3] [14]. This approach is particularly valuable for the quality control of iPSC-derived neural cultures, where variability in differentiation outcomes is a major hurdle for research and drug development [3] [35]. By providing a fast, affordable, and scalable method for quantifying culture composition, HCI stands ready to improve the reproducibility and translational value of neuroscientific research.
Single-Cell Profiling Outperforms Population-Level Metrics
This application note demonstrates the superior accuracy of single-cell morphological profiling over population-level metrics for classifying cell identity in dense, mixed neural cultures. Quantitative results from a validated imaging assay show that convolutional neural network (CNN)-based analysis of single-cell morphological features achieves 96% classification accuracy, significantly outperforming population-level classification based on time-in-culture criteria (86% accuracy). We provide detailed protocols for implementing this high-content imaging workflow, which enables robust quality control for induced pluripotent stem cell (iPSC)-derived neural culture models without disrupting subsequent molecular assays.
Traditional methods for characterizing cell culture composition often rely on population-level metrics such as time-in-culture or bulk sequencing, which obscure cellular heterogeneity. In induced pluripotent stem cell (iPSC)-derived neural cultures, this approach is particularly limiting due to the inherent variability between individual iPSC lines and differentiation batches [3]. The inability to comprehensively characterize iPSC-derived cell types at single-cell resolution hinders adoption in routine preclinical screening settings [3] [28].
High-content imaging based on Cell Painting (CP) offers an alternative approach that preserves single-cell resolution while capturing rich morphological information [3]. This method uses multiplexed fluorescent dyes to label various cellular compartments, generating morphotextural fingerprints that can distinguish cell types with high fidelity even in dense, mixed cultures [14]. When combined with deep learning algorithms, this approach enables unbiased identification of individual cell types without the destructive processing required by sequencing-based methods [3].
Comparative Performance Analysis
Quantitative Comparison of Classification Accuracy
Table 1: Performance comparison of single-cell profiling versus population-level metrics
| Method | Classification Basis | Accuracy | Precision | Recall | Application Context |
|---|---|---|---|---|---|
| Single-cell CNN classification | Morphological profiling using convolutional neural networks | 96.0% ± 1.8% | Balanced across cell types | Balanced across cell types | Mixed neural cultures at various densities |
| Population-level time classification | Time-in-culture as proxy for differentiation stage | 86.0% | Variable by cell type | Variable by cell type | iPSC-derived neural cultures |
| Random Forest classification | Hand-crafted morphotextural features | 71.0% ± 1.0% | Imbalanced (46% misclassification of 1321N1 cells) | Imbalanced | Monocultures of neural cell lines |
Density-Dependent Performance
Table 2: Effect of culture density on single-cell classification accuracy
| Culture Density (%) | Classification Accuracy | Key Observations | Recommended Input Region |
|---|---|---|---|
| 0-80% | >96% | Consistent high performance | Whole cell or nucleocentric |
| 80-95% | >96% | Maintained accuracy | Nucleocentric preferred |
| 95-100% | 92.0% ± 1.7% | Significant decrease due to segmentation challenges | Nucleocentric required |
Experimental Protocols
Cell Painting Assay for Neural Cultures
Principle: Cell Painting uses multiplexed fluorescent dyes to label multiple cellular compartments, generating rich morphological profiles that serve as distinctive fingerprints for different cell types [3] [14].
Reagents and Equipment:
- 1321N1 astrocytoma cells and SH-SY5Y neuroblastoma cells for benchmarking
- Cell Painting dyes: Hoechst 33342 (nuclei), Concanavalin A (endoplasmic reticulum), Wheat Germ Agglutinin (plasma membrane), SYTO 14 (nucleoli), Phalloidin (cytoskeleton), and MitoTracker (mitochondria)
- Confocal microscope with 4-channel capability
- Image analysis software (CellProfiler or equivalent)
Procedure:
- Culture neural cells on appropriate substrates to achieve desired density (0-100% confluency)
- Apply Cell Painting dye cocktail according to established protocols [3]
- Acquire 4-channel confocal images with consistent exposure settings across samples
- Export images in standardized format (TIFF preferred) for downstream analysis
Convolutional Neural Network Classification
Workflow Overview:
Detailed Protocol:
Image Preprocessing
- Segment individual cells using nuclear markers as reference points
- Extract bounding boxes around cell centroids (minimum 50×50 pixel regions)
- Apply data augmentation (rotation, flipping, brightness adjustment)
- Normalize pixel intensities across batches
Model Training
- Implement ResNet architecture with pre-trained weights
- Use balanced training set (minimum 5000 instances per cell type)
- Include multiple biological replicates in training data
- Train for 50-100 epochs with early stopping
- Validate on held-out test set from independent cultures
Classification and Interpretation
- Generate predictions for individual cells in mixed cultures
- Apply Gradient-weighted Class Activation Mapping (Grad-CAM) to identify discriminative features
- Calculate accuracy metrics per cell type and culture condition
Research Reagent Solutions
Table 3: Essential reagents for single-cell morphological profiling
| Reagent/Category | Specific Examples | Function | Application Notes |
|---|---|---|---|
| Cell Painting Dyes | Hoechst 33342, Concanavalin A, Wheat Germ Agglutinin, SYTO 14, Phalloidin, MitoTracker | Multiplexed labeling of cellular compartments | Optimize concentration for neural cells; avoid cytotoxicity |
| Cell Lines | 1321N1 astrocytoma, SH-SY5Y neuroblastoma | Benchmarking and validation | Use early passages; maintain consistent culture conditions |
| iPSC-Derived Neural Cells | Cortical neurons, astrocytes, microglia, neural progenitors | Primary application models | Characterize with canonical markers; account for maturation state |
| Imaging Reagents | PBS, fixation buffer (4% PFA), mounting media | Sample preparation and preservation | Validate compatibility with Cell Painting dyes |
| Software Tools | CellProfiler, Python (PyTorch/TensorFlow), ImageJ | Image analysis and deep learning | Implement standardized pipelines for reproducibility |
Technical Considerations
Optimizing Input Regions for Classification
Decision Framework for Input Selection:
Method Selection Guidelines
For culture densities below 80% confluency, both whole-cell and nucleocentric inputs deliver >96% classification accuracy. As density increases to 95-100% confluency, the nuclear region of interest (ROI) with its immediate environment maintains higher prediction accuracy (92.0% ± 1.7%) than whole-cell inputs, as nuclei remain largely separated and segmentable even in densely packed cultures [3]. This nucleocentric approach preserves classification performance when cell boundaries become obscured by cell-cell contacts.
Single-cell morphological profiling significantly outperforms population-level metrics for cell type identification in mixed neural cultures, achieving 96% classification accuracy versus 86% for time-in-culture based classification. This approach enables robust, non-destructive quality control for iPSC-derived neural models, addressing a critical need in drug screening and cell therapy development. The provided protocols offer researchers a comprehensive framework for implementing this powerful method in their own experimental workflows.
The adoption of human induced pluripotent stem cell (iPSC)-derived neural cultures is revolutionizing neuroscience research and preclinical drug screening by providing physiologically relevant human models of the brain [3]. However, a significant obstacle hindering their routine use is the inherent variability between individual iPSC lines and differentiation protocols, leading to inconsistent and potentially misleading results [3]. High-content image-based morphological profiling has emerged as a powerful, affordable, and scalable solution for quantifying cell culture composition [3]. This Application Note details protocols for cross-platform validation to ensure consistent and accurate cell type identification across different imaging modalities, a critical requirement for robust quality control in iPSC-derived mixed neural culture research.
Experimental Design and Workflow
The core objective is to implement a validation pipeline that ensures morphological profiling results are consistent and reproducible, regardless of the specific imaging platform used. This involves a multi-stage process from sample preparation through to cross-platform data alignment.
Core Experimental Workflow
The following diagram outlines the primary workflow for cross-platform validation of cell type identification.
Key Materials and Reagents
Table 1: Essential Research Reagent Solutions for Cross-Platform Validation
| Item | Function / Description | Key Considerations |
|---|---|---|
| iPSC-Derived Neural Cells | Primary model system containing mixed cell types (neurons, progenitors, glia). | Account for line-to-line and batch variability; maintain consistent differentiation protocols [3]. |
| Cell Painting Dyes | Multiplex fluorescent kit for morphological profiling (e.g., stains for nucleus, cytoplasm, ER, mitochondria, F-actin) [3]. | Ensure dye compatibility and minimal spectral overlap across all imaging platforms used. |
| Validated Antibody Panels | For immunocytochemistry (ICC) validation of cell types (e.g., MAP2 for neurons, GFAP for astrocytes, IBA1 for microglia) [49]. | Use as a ground truth reference; panels should be optimized for each imaging platform. |
| Annotation Software (ImageJ) | Publicly available software for manual cell annotation to generate training data [49]. | Critical for creating gold-standard datasets; requires consensus among multiple annotators. |
| Consensus Annotations | Manually curated datasets of cellular boundaries (whole cell and nucleus) from multiple experts [49]. | Serves as the ground truth for training and benchmarking segmentation and classification algorithms. |
Protocols for Multi-Platform Imaging and Analysis
Protocol: Sample Preparation and Staining for Cross-Platform Validation
Principle: Standardize sample processing to minimize technical variability when preparing slides for different imaging systems.
Materials:
- Cultured iPSC-derived mixed neural cells (e.g., 2D cultures or 3D organoids)
- Cell Painting dye kit [3]
- Fixation solution (e.g., 4% Paraformaldehyde in PBS)
- Permeabilization buffer (e.g., 0.1% Triton X-100 in PBS)
- Blocking buffer (e.g., 1-5% BSA in PBS)
- Validated antibody panels for ICC [49]
Procedure:
- Culture and Fixation: Grow iPSC-derived neural cultures to the desired density. Fix cells with 4% PFA for 15-20 minutes at room temperature.
- Permeabilization and Blocking: Permeabilize cells with 0.1% Triton X-100 for 10 minutes. Incubate with blocking buffer for 1 hour to reduce non-specific binding.
- Multiplex Staining: Perform the Cell Painting protocol according to established methods [3]. Simultaneously, stain a parallel set of samples with validated antibody panels for key neural cell markers to serve as a biological ground truth.
- Mounting: Mount coverslips using a compatible, anti-fade mounting medium.
Protocol: Image Acquisition Across Modalities
Principle: Acquire comparable image data from multiple fluorescent imaging platforms to assess consistency.
Materials:
- Stained samples from Protocol 3.1
- Compatible imaging platforms (e.g., Akoya Vectra 3.0, Ultivue InSituPlex with Zeiss Axioscan, Akoya CODEX) [49]
Procedure:
- Platform Calibration: Calibrate each imaging system using standard fluorescence slides to ensure intensity measurements are within a linear range.
- Multi-Platform Acquisition: Image the same biological samples, or technical replicates from the same batch, across the different platforms.
- For Sequential IF with Unmixing (e.g., Akoya Vectra): Acquire images using a multispectral imaging system. Apply spectral unmixing to generate pure marker-specific channels [49].
- For Sequential IF with Narrowband Capture (e.g., Ultivue): Use a system where each target is labeled with a spectrally distinct fluorophore and imaged without the need for unmixing [49].
- For Cyclical IF (e.g., Akoya CODEX): Perform cyclical staining and imaging, where fluorescent probes are added, imaged, and then stripped before the next cycle [49].
- Metadata Logging: Record all acquisition parameters (exposure times, laser powers, filters) for each platform to ensure reproducibility.
Protocol: Cross-Platform Image Analysis and Data Integration
Principle: Segment cells, extract morphological features, and align data structures to enable direct comparison of classification results across platforms.
Materials:
- Image datasets from Protocol 3.2
- Image analysis software (e.g., ImageJ, CellProfiler, Python with OpenCV/scikit-image)
- Computational environment for machine learning (e.g., Python with TensorFlow/PyTorch)
Procedure:
- Cell Segmentation:
- Use a convolutional neural network (CNN) trained on manual consensus annotations [49] to segment individual cells in images from all platforms.
- Manually review and validate segmentation accuracy against a subset of consensus annotations for each platform.
Morphological Feature Extraction:
- For each segmented cell, extract morphological and textural features (e.g., area, eccentricity, intensity, texture) from each fluorescence channel in defined regions of interest (nucleus, cytoplasm, whole cell) [3].
- Note: Studies have shown that inputs containing the nuclear region and its immediate environment can achieve classification accuracy as high as 96%, even in dense cultures [3].
Cross-Platform Data Alignment and Validation:
- The following diagram illustrates the tiered strategy for validating model consistency across imaging platforms.
Data Analysis and Interpretation
Quantitative Analysis of Classification Performance
The success of cross-platform validation is quantitatively assessed by measuring cell type classification accuracy across different imaging systems. The following table summarizes expected outcomes from a well-executed validation study.
Table 2: Expected Quantitative Outcomes for Cell Type Classification
| Cell Type / System | Benchmark Accuracy (Single Platform) | Minimum Target Accuracy (Cross-Platform) | Key Morphological Features |
|---|---|---|---|
| Neuron vs. Progenitor | >96% [3] | >90% | Cellular area, nuclear texture, cytoplasmic complexity |
| Neuron vs. Microglia | "Unequivocally discriminated" [3] | >95% | Somatic shape, process complexity, intensity profiles |
| Microglia (Activated vs. Non-Activated) | Lower accuracy than broad type ID [3] | Tiered analysis required | Cell body roundness, process thickness, branching |
| CNN vs. Random Forest | CNN significantly outperforms RF (F-score: 0.75) [3] | Use CNN for all platforms | Leverages deep morphological patterns vs. hand-crafted features |
Interpretation Guidelines
- Successful Validation: A classification model trained on data from one imaging platform maintains high accuracy (>90% for broad types) when applied to data from another platform. This indicates that the morphological descriptors are robust to technical variations.
- Platform-Specific Bias: If a model performs well on its training platform but poorly on others, this suggests platform-specific biases in staining, acquisition, or feature representation. Investigate and normalize the discrepant steps.
- Tiered Discrimination: Consistent with published findings, expect a tiered discrimination power where broad cell types (e.g., neuron vs. microglia) are identified with very high accuracy, while finer distinctions (e.g., activation states) are more challenging and may achieve lower, yet biologically useful, accuracy [3].
Approaching Human-Level Concordance and Identifying Ambiguous Cells
Within the field of high-content imaging for cell type identification in mixed neural cultures, achieving classification accuracy that rivals human expert annotation is a critical goal. This application note details a robust methodology that combines a multiplexed fluorescent staining assay (Cell Painting) with a deep learning-based classification model to quantitatively identify and characterize neural cell types in dense, mixed cultures with high fidelity. This protocol is designed to address the central challenge of variability in induced pluripotent stem cell (iPSC)-derived neural cultures, which hinders their adoption in routine preclinical screening and cell therapy pipelines [3] [14]. By providing a fast, affordable, and scalable quality control approach, this method enables researchers to move beyond population-level assumptions and gain single-cell resolution of culture composition, ultimately improving experimental reproducibility and translational value [3].
Experimental Protocols & Workflows
Core Experimental Workflow: From Cell Culture to Cell Classification
The following diagram outlines the primary experimental and computational workflow for unbiased cell identity identification.
Tiered Strategy for Resolving Ambiguous Cell Identities
For complex classification tasks, a tiered strategy improves discrimination of subtly different cell states, such as activated vs. non-activated microglia [3].
Detailed Step-by-Step Protocols
Cell Painting and Staining Protocol for Mixed Neural Cultures
This protocol adapts the Cell Painting assay for use in dense, mixed neural cultures, enabling the acquisition of rich morphological data [3] [14].
- Materials:
- Fixed mixed neural cultures (e.g., iPSC-derived neurons, progenitors, microglia)
- Cell Painting staining kit or individual dyes: Hoechst 33342 (nuclei), Concanavalin A (actin cytoskeleton), Wheat Germ Agglutinin (Golgi/ER), MitoTracker (mitochondria), and Phalloidin (F-actin) [14]
- Staining buffer (e.g., PBS with 1% BSA)
- Blocking solution (e.g., 1-3% BSA in PBS)
- Permeabilization solution (0.1% Triton X-100 in PBS, if required)
- Procedure:
- Fixation: Fix cells with 4% paraformaldehyde for 15-20 minutes at room temperature.
- Permeabilization (if needed): For intracellular targets, permeabilize with 0.1% Triton X-100 for 10 minutes.
- Blocking: Incubate with blocking solution for 30-60 minutes to reduce non-specific binding.
- Staining: Prepare the multiplexed dye cocktail in staining buffer according to manufacturer recommendations. Incubate cells with the cocktail for 30-60 minutes, protected from light.
- Washing: Perform 3x5 minute washes with staining buffer to remove unbound dye.
- Mounting (if applicable): Mount coverslips with an anti-fade mounting medium. Seal edges with nail polish.
- Storage: Store stained samples at 4°C in the dark until imaging, preferably within 24-48 hours.
High-Content Confocal Imaging Protocol
Optimal image acquisition is critical for high-fidelity classification. The following protocol is based on the use of a Laser Scanning Confocal Microscope (LSCM) [50] [51].
- Materials:
- Stained samples
- Laser Scanning Confocal Microscope (e.g., Nikon A1 HD25/A1R HD25, C2+ systems)
- High Numerical Aperture (NA) objective lenses (e.g., 40x/NA 1.2, 60x/NA 1.4)
- Procedure:
- Objective Lens Selection: Select a high-NA objective (≥1.2) to maximize resolution and light collection. A 40x or 60x lens is typically ideal for single-cell resolution [50].
- Laser and Detector Setup: Configure lasers to match the excitation spectra of the dyes used. Use an Acousto-Optic Tunable Filter (AOTF) for precise laser control. Set photomultiplier tube (PMT) detector gains and offsets to avoid signal saturation.
- Pinhole Adjustment: Set the confocal pinhole diameter to 1 Airy Unit (AU) to achieve optimal optical sectioning and lateral resolution (~0.2 µm) without unnecessarily sacrificing signal [51].
- Multi-Channel Sequential Scanning: Acquire images for each fluorescent channel sequentially to prevent bleed-through (cross-talk) between channels.
- Z-stack Acquisition (Optional): For 3D information, acquire a z-stack of images through the sample depth. Set the z-step size to approximately 0.5 µm, which is less than the axial resolution (~0.6 µm) [51].
- Field of View and Replication: Image multiple, randomly selected fields of view per well/replicate to ensure a representative sample of the culture. Include at least three biological replicates.
Computational Pipeline for Model Training and Classification
This protocol covers the analysis of acquired images to train a convolutional neural network (CNN) for classification [3] [14].
- Materials:
- High-content confocal images (4 channels)
- Computational environment (e.g., Python with PyTorch/TensorFlow, Anaconda distribution)
- Deep learning framework with a ResNet architecture
- Procedure:
- Image Pre-processing and Segmentation: Use a segmentation algorithm (e.g., CellPose) to identify individual cells and their nuclei. In dense cultures, prioritize nuclear segmentation for robustness.
- Input Generation ("Nucleocentric" Crops): For each segmented cell, extract an image crop centered on the nuclear centroid. Blank out the surrounding cell areas to focus the model on the most informative regions, which enhances performance in dense cultures [14].
- Dataset Curation: Split the dataset of image crops into training, validation, and test sets (e.g., 70/15/15). Ensure all sets contain data from all biological replicates to prevent batch bias.
- Model Training: Train a ResNet CNN using the training set. Use data augmentation (random flips, rotations, minor contrast changes) to improve model generalization.
- Model Validation and Testing: Evaluate the model's accuracy on the validation set during training to fine-tune hyperparameters. Final performance metrics (e.g., accuracy, F-score) should be reported on the held-out test set.
- Ambiguity Identification: Analyze misclassified cells from the test set. Use techniques like Gradient-weighted Class Activation Mapping (Grad-CAM) to visualize image regions leading to misclassification, helping to identify dead cells, debris, or truly ambiguous morphological states [14].
Key Experimental Data and Performance Benchmarks
Quantitative Performance of Classification Models
This table summarizes the key quantitative findings from benchmarking the described approach against traditional methods.
Table 1: Performance comparison of cell classification methods in neural cultures [3] [14]
| Classification Method | Input Data | Tested Culture System | Reported Accuracy | Key Strengths |
|---|---|---|---|---|
| Convolutional Neural Network (CNN) | 4-channel image crops (nucleocentric) | Mixed neuroblastoma/astrocytoma cell lines | 96.0% ± 1.8% | High accuracy, robust to density, minimal feature engineering |
| Random Forest (RF) | Hand-crafted morphotextural features | Mixed neuroblastoma/astrocytoma cell lines | 71.0% - 75.0% | Model interpretability, lower computational cost |
| Cell-based Prediction (CNN) | Nucleocentric image crops | iPSC-derived neurons vs. progenitors | 96.0% | Superior to population-level metrics |
| Population-level (Time in culture) | Culture day metadata | iPSC-derived neurons vs. progenitors | 86.0% | Simple to implement, but less accurate |
Impact of Culture Density on Classification Accuracy
This table shows how the nucleocentric profiling approach maintains high performance even in challenging, dense cultures.
Table 2: Model robustness across increasing culture confluency [14]
| Culture Confluency | Classification Accuracy (CNN) | Notes |
|---|---|---|
| 0% - 80% | ~96% (No significant decrease) | Robust performance across low to high density. |
| 80% - 95% | ~96% | Maintained accuracy. |
| 95% - 100% | 92.0% ± 1.7% | Slight decrease due to extreme cell crowding and shape deformation. Nuclear ROI remains reliable. |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential reagents and materials for the cell identity identification workflow
| Item | Function / Role in Protocol | Example / Specification |
|---|---|---|
| Cell Painting Dye Cocktail | Multiplexed fluorescent labeling of cellular compartments. | Hoechst 33342 (Nuclei), Concanavalin A (ER), Phalloidin (F-actin), etc. [14] |
| Laser Scanning Confocal Microscope (LSCM) | High-resolution, optical sectioning fluorescence imaging. | Systems with AOTF laser control, PMT detectors, and high-NA objectives (40x/NA 1.2, 60x/NA 1.4) [50] [51]. |
| High-NA Objective Lens | Maximizes light collection and resolution for detailed morphology. | 60x oil immersion, NA 1.4 [50]. |
| ResNet CNN Architecture | Deep learning model for image-based classification. | Standard ResNet (e.g., ResNet50) implemented in PyTorch/TensorFlow [3] [14]. |
| Cell Segmentation Software | Identifies individual cells and nuclei in dense images. | CellPose or other deep learning-based segmentation tools. |
| iPSC-Derived Neural Cells | Physiologically relevant human model system. | Co-cultures of neurons, neural progenitors, and microglia [3]. |
Application to Drug Development and Basic Research
This methodology provides a powerful tool for quality control in iPSC-based disease modeling and preclinical drug screening. By accurately quantifying the ratio of postmitotic neurons to neural progenitors, researchers can standardize cultures across experiments and batches, increasing the reproducibility of functional assays and toxicity screens [3]. Furthermore, the ability to discriminate microglia and their activation state in a mixed culture opens new avenues for studying neuroinflammation in vitro, a key mechanism in many neurological disorders. The tiered classification strategy allows for the systematic identification of ambiguous cells—such as those undergoing cell death or possessing intermediate states—which can be isolated for further molecular analysis, turning a classification challenge into a discovery opportunity [14].
Conclusion
High-content imaging, particularly when powered by deep learning, has emerged as a robust, unbiased, and scalable solution for cell type identification in complex mixed neural cultures. It successfully addresses the critical need for quality control in variable iPSC-derived models, a key bottleneck in translational neuroscience. The technology's ability to provide single-cell resolution data, even in dense cultures, surpasses the limitations of traditional, population-averaging methods. As these automated systems now approach or even match the accuracy of human experts, they pave the way for more reproducible and physiologically relevant drug screening and disease modeling. The future of this field lies in the deeper integration of HCI with other omics technologies, the development of more interpretable AI models, and the establishment of standardized, shareable image data repositories to accelerate the discovery of new therapeutics for neurological disorders.
References
- 1. Advances and challenges in modeling inherited peripheral ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11263568/]
- 2. Addressing variability in iPSC-derived models of human ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6994963/]
- 3. Unbiased identification of cell identity in dense mixed ... [https://elifesciences.org/articles/95273]
- 4. Quantitative assessment of neural outgrowth using spatial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5482290/]
- 5. Network science characteristics of brain-derived neuronal ... [https://www.nature.com/articles/s41598-020-72013-7]
- 6. High Content, Phenotypic Assays and Screens for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6532062/]
- 7. Automated high-content imaging in iPSC-derived neuronal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10602900/]
- 8. Assay Development Guidelines for Image-Based High ... - NCBI [https://www.ncbi.nlm.nih.gov/books/NBK100913/]
- 9. High-Content Analysis (Quantitative Imaging) Information [https://www.thermofisher.com/us/en/home/life-science/cell-analysis/cell-analysis-learning-center/cell-imaging-information/high-content-analysis-information.html]
- 10. Combining phenomics with transcriptomics reveals cell- ... [https://www.nature.com/articles/s41467-025-61547-x]
- 11. High-content screening in drug discovery: A brief guide [https://alitheagenomics.com/blog/high-content-screening-in-drug-discovery-a-brief-guide]
- 12. AI for High-Content Screening in Drug Discovery [https://lifesciences.danaher.com/us/en/library/ai-high-content-screening-drug-discovery]
- 13. Cell Painting, a high-content image-based assay for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5223290/]
- 14. Unbiased identification of cell identity in dense mixed ... [https://elifesciences.org/reviewed-preprints/95273v1]
- 15. Cell Painting High-Content Image Assay [https://www.thermofisher.com/us/en/home/life-science/cell-analysis/cell-structure/cell-painting-assay.html]
- 16. HCA Platforms for Automated, Quantitative Analyses [https://www.thermofisher.com/us/en/home/references/newsletters-and-journals/bioprobes-journal-of-cell-biology-applications/bioprobes-76/hca-platforms-automated-quantitative-analyses.html]
- 17. Protocol for the Differentiation of Human Induced ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5608344/]
- 18. Unbiased identification of cell identity in dense mixed ... [https://elifesciences.org/reviewed-preprints/95273v2]
- 19. Hoechst 33342 Protocol for Imaging [https://www.thermofisher.com/us/en/home/references/protocols/cell-and-tissue-analysis/protocols/hoechst-33342-imaging-protocol.html]
- 20. Image-Based Profiling in Live Cells Using Live Cell Painting [https://pubmed.ncbi.nlm.nih.gov/41080447/]
- 21. A robust and reliable methodology to perform GECI-based ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10560759/]
- 22. Multiplexed and high-throughput neuronal fluorescence ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6763432/]
- 23. Unbiased identification of cell identity in dense mixed ... [https://elifesciences.org/reviewed-preprints/95273]
- 24. Comparison of handcrafted features and convolutional ... [https://www.nature.com/articles/s41598-020-77264-y]
- 25. Comparison between Deep Learning and Conventional ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8871174/]
- 26. Evaluation of the performance of traditional machine learning ... [https://qims.amegroups.org/article/view/59009/html]
- 27. Utilizing convolutional neural networks for discriminating ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11344342/]
- 28. Unbiased identification of cell identity in dense mixed ... [https://pubmed.ncbi.nlm.nih.gov/39819559/]
- 29. Optimized Protocol for Culture of Mouse Fetal Hindbrain ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12153581/]
- 30. An optimized method for directed differentiation of ... [https://www.nature.com/articles/s41598-025-02847-6]
- 31. Frontiers | Organic charge-modulated transistor for electrophysiological... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2025.1571011/full]
- 32. - High and Analysis of 2D and 3D Content ... Imaging Neuronal Cultures [https://www.thermofisher.com/pk/en/home/life-science/cell-culture/cell-culture-learning-center/cell-culture-resource-library/cell-culture-transfection-application-notes/high-content-imaging-analysis-2d-3d-neuronal-cultures-grown-optimized-media.html]
- 33. Unbiased identification of cell identity in dense mixed ... [https://elifesciences.org/reviewed-preprints/95273v3]
- 34. Unbiased identification of cell identity in dense mixed neural cultures [https://sciety.org/articles/activity/10.7554/elife.95273.3]
- 35. High content screening miniaturization and single cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10119332/]
- 36. Steps to Build a Data Augmentation Pipeline in 2025 [https://www.unitxlabs.com/resources/data-augmentation-pipeline-2025/]
- 37. Data Augmentation: Techniques That Work in Real-World ... [https://labelyourdata.com/articles/data-augmentation]
- 38. Enhancing Image Classification with Augmentation: Data ... [https://arxiv.org/html/2502.18691v1]
- 39. Evaluating pre-processing and deep learning methods in ... [https://www.sciencedirect.com/science/article/pii/S1110016825001176]
- 40. Grad-CAM (Gradient-weighted Class Activation Mapping) [https://www.sciencedirect.com/science/article/abs/pii/S0010482525015537]
- 41. Grad-CAM: A Gradient-based Approach to Explainability in ... [https://medium.com/@kdk199604/grad-cam-a-gradient-based-approach-to-explainability-in-deep-learning-871b3ab8a6ce]
- 42. A Guide to Grad-CAM in Deep Learning [https://www.analyticsvidhya.com/blog/2023/12/grad-cam-in-deep-learning/]
- 43. Grad-CAMO: Learning Interpretable Single-Cell ... [https://arxiv.org/html/2403.17615v1]
- 44. Recent Advances in High-Content Imaging and Analysis in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10572296/]
- 45. Optimizing the in vitro neuronal microenvironment to mitigate ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-025-04591-0]
- 46. High-Content Screening of Primary Neurons: Ready for Prime ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2787795/]
- 47. An Update on Flow Cytometry Analysis of Hematological ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12190783/]
- 48. Design and validation of novel flow cytometry panels to ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2024.1432816/full]
- 49. Cross-platform dataset of multiplex fluorescent cellular ... [https://www.nature.com/articles/s41597-023-02108-z]
- 50. Specimen Preparation and Imaging - Confocal [https://www.microscopyu.com/techniques/confocal/specimen-preparation-and-imaging]
- 51. Confocal Microscopy: Principles and Modern Practices - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6961134/]