The Addicted Brain: A Neurocircuitry Analysis of the Basal Ganglia, Extended Amygdala, and Prefrontal Cortex
This article provides a comprehensive neurobiological framework for substance use disorders, focusing on the dynamic interplay between the basal ganglia, extended amygdala, and prefrontal cortex.
The Addicted Brain: A Neurocircuitry Analysis of the Basal Ganglia, Extended Amygdala, and Prefrontal Cortex
Abstract
This article provides a comprehensive neurobiological framework for substance use disorders, focusing on the dynamic interplay between the basal ganglia, extended amygdala, and prefrontal cortex. Grounded in the well-established three-stage addiction cycle (binge/intoxication, withdrawal/negative affect, preoccupation/anticipation), it synthesizes current evidence from animal and human imaging studies to explain the transition from voluntary use to compulsive addiction. Aimed at researchers and drug development professionals, the content explores foundational circuitry, methodological approaches for investigation, challenges in modeling and treating these neuroadaptations, and a comparative analysis with non-drug addictions to validate the model's broader applicability. The review concludes by outlining future directions for leveraging this neurocircuitry knowledge to develop novel biomedical interventions.
Core Neurocircuitry of Addiction: Deconstructing the Basal Ganglia, Extended Amygdala, and Prefrontal Cortex
Addiction, increasingly understood through the framework of substance use disorder (SUD), is a chronically relapsing condition characterized by compulsion to seek and take a drug, loss of control over intake, and emergence of a negative emotional state during withdrawal [1] [2]. The neurobiological understanding of addiction has transformed the field, moving past historical perceptions of moral failing to a model of chronic brain disease [3] [4]. This whitepaper elaborates on a predominant heuristic in addiction neuroscience: the three-stage cycle of binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation. This framework not only describes behavioral phenotypes but is also mapped to specific neurocircuitry, neuroplasticity, and neurochemical dysregulations, providing a powerful tool for guiding research and therapeutic development [5] [1] [2]. The cycle is deeply rooted in the dysregulation of three key brain regions: the basal ganglia, the extended amygdala, and the prefrontal cortex [3] [6] [7]. The following sections provide a neurocircuitry-based analysis of each stage, supported by key experimental data and methodologies.
Stage 1: Binge/Intoxication
The binge/intoxication stage is defined by the rewarding or pleasurable effects of initial substance use, which positively reinforces drug-taking behavior [6] [8]. This stage is primarily mediated by the basal ganglia, a key node of the brain's reward circuit [3] [7].
Core Neurocircuitry and Neurotransmitters
The ventral tegmental area (VTA) and the nucleus accumbens (NAc) within the ventral striatum are focal points for this stage [5] [1]. All addictive substances produce powerful surges of neurotransmitters in this circuit:
- Dopamine (DA): Nearly all addictive substances, particularly stimulants like cocaine and amphetamine, directly or indirectly increase dopamine signaling from the VTA to the NAc [6] [7]. This surge is often larger and more prolonged than those produced by natural rewards, powerfully reinforcing the drug-taking behavior [7] [8].
- Opioid Peptides: The brain's endogenous opioid system (involving endorphins and mu, delta, and kappa receptors) plays a critical role in the rewarding effects of opioids and alcohol, often stimulating the NAc directly or via modulation of GABA and dopamine systems [6].
- GABA: Sedative-hypnotic drugs like benzodiazepines target GABA receptors, while THC from cannabis targets GABA neurons that in turn stimulate the dopamine system [6].
This drug-induced neuroactivation leads to incentive salience, whereby drugs and associated cues acquire powerful motivational properties [2].
Key Experimental Data and Protocols
A wealth of data on this stage comes from animal models, particularly the intravenous self-administration (IVSA) paradigm, which is considered the gold standard for studying drug-seeking behavior [8]. The table below summarizes quantitative findings from key experiments.
Table 1: Key Experimental Findings from the Binge/Intoxication Stage
| Experimental Measure | Key Finding | Implication | Primary Source |
|---|---|---|---|
| Dopamine Release | Drugs of abuse cause larger, faster dopamine surges in NAc compared to natural rewards. | Underpins powerful reinforcement and learning of drug-associations. [7] [8] | |
| Breakpoint in Progressive Ratio | Animals with escalated intake will press levers hundreds of times for a single drug infusion. | Measures increased motivational drive ("wanting") for the drug. [8] | |
| Metabolic Tolerance | Chronic ethanol exposure induces elevated liver enzyme activity (e.g., CYP2E1). | Leads to faster drug degradation, reducing bioavailability. [8] | |
| Receptor Downregulation | Chronic drug exposure reduces D2 dopamine receptor expression in the striatum. | Contributes to tolerance and reduced sensitivity to reward. [8] |
Detailed Protocol: Intravenous Self-Administration (IVSA) Purpose: To investigate the reinforcing properties of a drug and the motivation to obtain it. Workflow:
- Surgery: A catheter is surgically implanted into the jugular vein of a rodent (e.g., rat or mouse) and connected to an external syringe pump containing a sterile solution of the drug under investigation.
- Acquisition Training: The animal is placed in an operant chamber equipped with levers or nosepokes. A response on the "active" lever results in:
- An intravenous infusion of the drug.
- Presentation of a conditioned stimulus (CS), such as a light or tone.
- Responses on an "inactive" lever have no consequence.
- Motivation Assessment (Progressive Ratio): To measure how much work an animal will expend for the drug, the required number of lever presses for a single infusion is progressively increased within a session (e.g., 1, 2, 4, 6, 9, 12...). The highest ratio completed before the animal ceases to respond is the "breakpoint," a metric of the drug's reinforcing efficacy.
- Data Analysis: Primary dependent variables include the number of infusions earned, the breakpoint, and the discrimination between active and inactive levers.
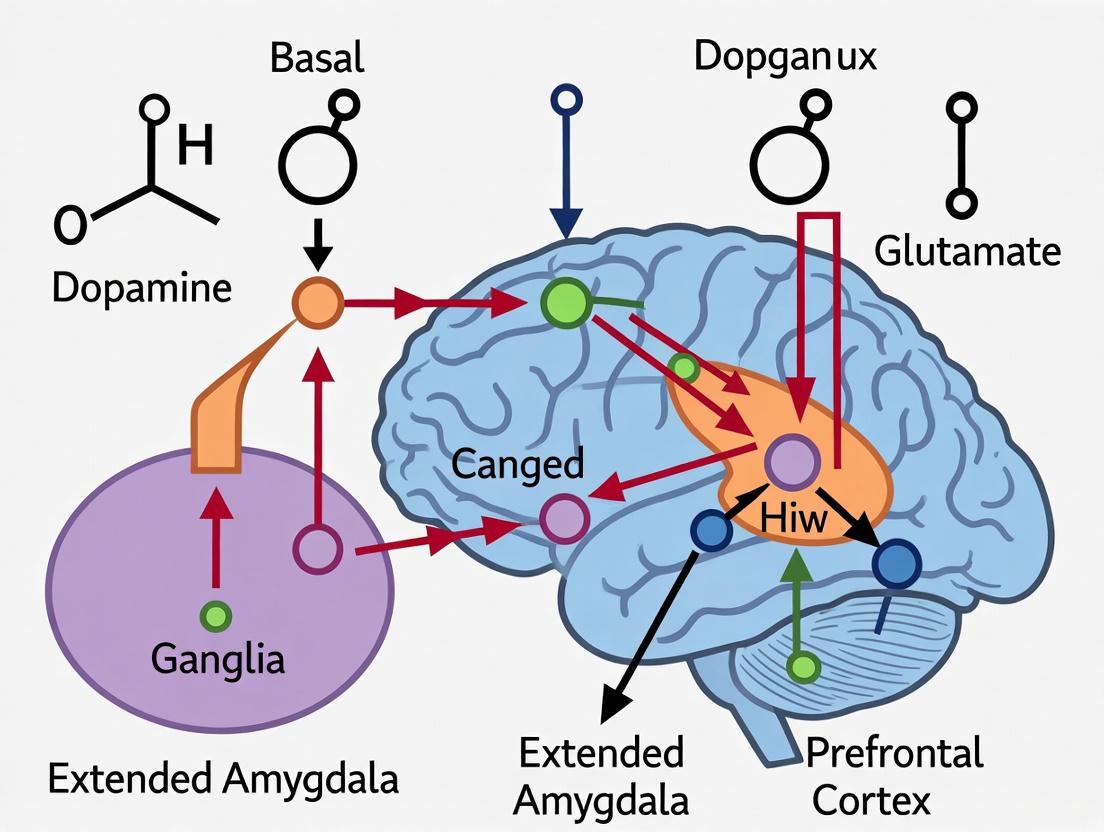
The following diagram illustrates the core neurocircuitry and neuropharmacological interactions of the binge/intoxication stage:
Stage 2: Withdrawal/Negative Affect
The withdrawal/negative affect stage emerges when drug consumption ceases, characterized by a negative emotional state—including dysphoria, anxiety, irritability, and emotional pain—that defines the "dark side" of addiction [1] [2]. This stage is a key driver of negative reinforcement, where individuals resume drug use to alleviate this aversive state [6].
Core Neurocircuitry and Neurotransmitters
This stage is primarily mediated by the extended amygdala, a macrostructure that includes the central amygdala, bed nucleus of the stria terminalis (BNST), and a part of the nucleus accumbens shell [1] [8]. Key neuroadaptations include:
- Reward Deficit: Chronic drug use leads to a reduction in the sensitivity of the brain's reward systems. This is evidenced by decreased dopamine release and downregulation of dopamine D2 receptors in the reward circuit, elevating the reward threshold and diminishing the capacity to experience pleasure from natural reinforcers [6] [7] [8].
- Stress Surfeit: The extended amygdala shows increased activation of brain stress systems. Key mediators include:
- Corticotropin-Releasing Factor (CRF): A primary driver of the stress response, CRF is upregulated in the extended amygdala during withdrawal [6] [2].
- Dynorphin: This kappa-opioid receptor agonist, which has dysphoric effects, is upregulated in the nucleus accumbens [2].
- Norepinephrine: This neurotransmitter's systems are also hyperactive during withdrawal [6].
Key Experimental Data and Protocols
Research on this stage utilizes models of conditioned withdrawal and measures of affective states in animals, often paired with intracranial pharmacological manipulations.
Table 2: Key Experimental Findings from the Withdrawal/Negative Affect Stage
| Experimental Measure | Key Finding | Implication | Primary Source |
|---|---|---|---|
| Intracranial Self-Stimulation (ICSS) Threshold | Withdrawal from all major drugs of abuse elevates brain reward thresholds. | Quantifies the anhedonic (loss of pleasure) state associated with withdrawal. [2] | |
| Anxiety-like Behavior (e.g., Elevated Plus Maze) | Rodents undergoing withdrawal spend less time in the open arms of the maze. | Models the anxiety and increased stress reactivity seen in human withdrawal. [8] | |
| CRF Antagonists | Blocking CRF receptors in the extended amygdala reduces excessive drug self-administration. | Validates the CRF system as a critical mediator of negative reinforcement. [6] [2] | |
| Dopamine Receptor Availability | Human imaging shows long-lasting decreases in striatal D2 receptors, even after detoxification. | Provides a biological basis for prolonged anhedonia and vulnerability to relapse. [6] |
Detailed Protocol: Conditioned Place Aversion (CPA) to Measure Affective Withdrawal Purpose: To assess the aversive motivational properties of drug withdrawal. Workflow:
- Pre-Test: A rodent is allowed to freely explore a two-chamber apparatus with distinct visual and tactile cues in each chamber. The time spent in each chamber is recorded to establish a baseline preference.
- Conditioning: Over several sessions, the animal is subjected to a state of drug withdrawal (e.g., by administering a drug antagonist like naloxone for opioids) and confined to one chamber. On alternate sessions, the animal is given a saline injection and confined to the other chamber.
- Post-Test: The animal is again allowed free access to both chambers in a drug-free state. A significant avoidance of the chamber previously paired with the withdrawal state indicates that the withdrawal is aversive (Conditioned Place Aversion).
- Pharmacological Validation: The protocol can be repeated while administering potential therapeutic compounds (e.g., CRF antagonists) to test if they can block the expression of CPA.
The following diagram illustrates the neurobiological mechanisms driving the withdrawal/negative affect stage:
Stage 3: Preoccupation/Anticipation
The preoccupation/anticipation (or craving) stage involves the persistent desire for the drug and its reinstatement after a period of abstinence, representing a core challenge in treating this chronic relapsing disorder [5] [1]. This stage is characterized by deficits in executive function and inhibitory control.
Core Neurocircuitry and Neurotransmitters
The prefrontal cortex (PFC) and its projections are central to this stage [3] [6]. Key regions and systems include:
- Prefrontal Cortex (PFC): This area, responsible for executive function (decision-making, impulse control, self-regulation), becomes dysregulated in addiction. Neuroimaging studies show abnormal activity and impaired performance on cognitive tasks dependent on the PFC [6] [8].
- Orbitofrontal Cortex (OFC) and Dorsolateral PFC (dlPFC): The OFC is involved in assigning value to rewards, and its dysfunction contributes to the overvaluation of the drug. The dlPFC is critical for cognitive control, and its impairment underlies loss of control over drug intake [5] [1].
- Anterior Cingulate Cortex (ACC) and Inferior Frontal Cortex: Involved in error detection, conflict monitoring, and inhibitory control [1].
- Glutamate: The primary excitatory neurotransmitter from the PFC to the basal ganglia and extended amygdala is critically involved in relapse. Drug-seeking reinstatement is linked to increased glutamate release in the nucleus accumbens [2].
- Hippocampus and Insula: The hippocampus is involved in memory and context-associated cravings, while the insula is implicated in interoception and conscious drug urges [5] [1] [8].
Key Experimental Data and Protocols
The reinstatement model in animals is the primary tool for studying relapse, mirroring the preoccupation/anticipation stage in humans.
Table 3: Key Experimental Findings from the Preoccupation/Anticipation Stage
| Experimental Measure | Key Finding | Implication | Primary Source |
|---|---|---|---|
| Reinstatement of Drug Seeking | After extinction, drug-seeking returns upon exposure to drug cues, stress, or a prime dose. | Models relapse in humans and is used to screen anti-craving medications. [8] | |
| Cognitive Task Impairment | Humans with cocaine/opioid use disorders show deficits in Stroop and Go/No-Go tasks. | Demonstrates impaired executive function and behavioral inhibition. [6] | |
| Glutamate Release in NAc | Cue-induced reinstatement of drug-seeking is correlated with increased glutamate in the NAc core. | Highlights glutamate as a key transmitter mediating relapse. [2] | |
| Insula Activation | Human neuroimaging shows insula activation during cue-elicited craving; insula damage disrupts addiction. | Suggests the insula integrates bodily signals into conscious urges to take drugs. [5] [1] |
Detailed Protocol: Reinstatement of Drug-Seeking Behavior Purpose: To model relapse and identify neural mechanisms and potential treatments for craving. Workflow:
- Self-Administration Training: Animals are trained to self-administer a drug as described in the IVSA protocol.
- Extinction: The drug and the conditioned stimulus (CS) are withheld. Lever presses no longer result in drug infusion or cue presentation. This phase continues until the drug-seeking behavior (e.g., active lever presses) declines to a predetermined criterion.
- Reinstatement Test: Drug-seeking behavior is provoked by one of three main triggers:
- Cue-Induced: Presentation of the drug-associated CS (light/tone) non-contingently.
- Drug-Induced (Priming): A non-contingent, small injection of the drug.
- Stress-Induced: Exposure to an acute stressor, such as a brief footshock.
- Data Analysis: The primary dependent variable is the number of responses on the previously active lever during the reinstatement test session, compared to responses during the last extinction session. An increase in lever pressing indicates reinstatement of drug-seeking.
The following diagram illustrates the complex neurocircuitry governing the preoccupation/anticipation stage:
The Scientist's Toolkit: Essential Research Reagents & Materials
This section details key reagents, tools, and technologies essential for conducting research within the three-stage framework of addiction.
Table 4: Essential Research Reagents and Materials for Addiction Neurobiology
| Reagent / Material | Function / Application | Example Use Case |
|---|---|---|
| Selective Receptor Agonists/Antagonists | Pharmacological probes to activate or block specific neurotransmitter receptors in vivo or in vitro. | CRF receptor antagonists to test blockade of stress-induced reinstatement [6] [2]. Dopamine D1/D2 antagonists to probe role in self-administration. |
| Viral Vector Systems (e.g., AAV) | For targeted gene delivery to manipulate gene expression in specific brain regions (knockdown, overexpression, CRISPR). | Herpes simplex virus (HSV) vectors for transient gene transfer in neuropsychiatric research [2]. Channelrhodopsin (Chemogenetics DREADDs) for circuit mapping. |
| Radioligands for PET/SPECT Imaging | Molecules that bind to specific neuroreceptors (e.g., dopamine D2, mu-opioid) for quantification in living brain. | [[11C]raclopride PET to measure decreases in striatal D2 receptor availability in cocaine addiction [6]. |
| Microdialysis Probes | In vivo sampling of extracellular fluid to measure dynamic changes in neurotransmitters (e.g., DA, Glutamate). | Measuring drug-induced surges in accumbal dopamine or glutamate during reinstatement [2]. |
| cAMP & PKA Activity Assays | Biochemical kits to measure intracellular second messenger signaling, which is upregulated by chronic drug exposure. | Quantifying cAMP upregulation in the NAc following chronic stimulant exposure [2]. |
| Transgenic Rodent Models | Animals with specific genes knocked out or mutated to study their role in addiction vulnerability. | Studying addiction-related behaviors in mice with knockout of genes like CHRNA2 (linked to cannabis use disorder) [4]. |
| fMRI/MRI for Human Studies | Non-invasive neuroimaging to assess brain structure (MRI) and function (fMRI) during cognitive tasks or cue exposure. | Identifying hypoactivity in the PFC and hyperactivity in the extended amygdala in individuals with SUD [5] [3]. |
Integrated View and Therapeutic Implications
The three-stage cycle is a dynamic and escalating framework. The binge/intoxication stage progressively loads the basal ganglia, strengthening incentive salience and habits. The withdrawal/negative affect stage engages the extended amygdala, establishing negative reinforcement. The preoccupation/anticipation stage, governed by a dysregulated prefrontal cortex, allows cues, stress, or a single dose to trigger the cycle anew [1] [6] [2]. This neurocircuitry-based analysis directly informs medication development, suggesting multi-target approaches:
- Binge/Intoxication: Dopamine or opioid receptor partial agonists/antagonists.
- Withdrawal/Negative Affect: CRF antagonists, kappa-opioid antagonists, and neurokinin-1 receptor antagonists.
- Preoccupation/Anticipation: Glutamate modulators (e.g., N-acetylcysteine, modafinil) and cognitive enhancers to improve PFC function [2].
Furthermore, this framework is relevant to behavioral addictions (e.g., gambling, internet addiction), which share underlying mechanisms of incentive salience, reward deficits, and executive dysfunction [2]. Recognizing adolescence as a period of heightened vulnerability due to ongoing PFC maturation is also critical for prevention strategies [3] [9]. Continued research within this heuristic framework promises to yield more precise and effective therapeutics for substance use disorders.
The binge/intoxication stage of addiction is centrally mediated by the brain's reward circuitry, with the basal ganglia playing a critical role in the transition from voluntary drug use to compulsive habits. This whitepaper provides a comprehensive technical analysis of the neurobiological mechanisms underlying this stage, detailing how drugs of abuse hijack dopaminergic, opioidergic, and glutamatergic systems within the basal ganglia to produce lasting neuroadaptations. We synthesize evidence from molecular, cellular, and systems neuroscience to elucidate the circuit-level dysregulations that drive the progressive narrowing of behavioral repertoire toward drug-seeking, focusing on the transition from ventral to dorsal striatal control. The review integrates quantitative neurochemical data, experimental methodologies for studying addiction phenotypes, and visualizations of key signaling pathways to provide researchers and drug development professionals with a foundational resource for understanding and targeting this critical phase of the addiction cycle.
Drug addiction represents a dramatic dysregulation of motivational circuits characterized by compulsion to seek and take drugs, loss of control over intake, and emergence of negative emotional states when access to the drug is prevented [10]. The disorder is conceptualized as a three-stage cycle—binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation—that worsens over time and involves distinct but interacting neurocircuitry adaptations [10] [11]. Within this framework, the binge/intoxication stage is primarily mediated by the basal ganglia and serves as the initial driver of the addiction process.
The binge/intoxication stage is defined by the rewarding effects of drugs of abuse and the development of incentive salience and drug-seeking habits [10]. This stage encompasses both the acute pharmacological reinforcement that initiates drug use and the neuroplastic changes that occur with repeated administration, ultimately leading to the transition from controlled use to compulsive drug-taking. Understanding the precise mechanisms through which addictive substances alter basal ganglia function is crucial for developing interventions that can prevent or reverse the progression to addiction.
This technical review focuses on the neuroadaptations within the basal ganglia and associated reward circuits during the binge/intoxication stage, with particular emphasis on: (1) the acute effects of drugs on reward systems; (2) the transition from goal-directed to habitual drug seeking; (3) the molecular and cellular mechanisms underlying this transition; and (4) experimental approaches for studying these processes. The analysis is framed within the broader context of addiction neurocircuitry involving the basal ganglia, extended amygdala, and prefrontal cortex [3].
Neurobiological Mechanisms of the Binge/Intoxication Stage
Acute Drug Reward and Dopamine Signaling
The rewarding effects of virtually all drugs of abuse involve direct or indirect activation of the ascending mesocorticostriatal dopamine system, particularly dopamine projections from the ventral tegmental area (VTA) to the ventral striatum (nucleus accumbens) [10]. Drugs of abuse cause rapid, steep increases in dopamine concentration in the nucleus accumbens that activate low-affinity dopamine D1 receptors, which are necessary for the rewarding effects of drugs and for triggering conditioned responses [10]. In humans, positron emission tomography (PET) studies have demonstrated that intoxicating doses of alcohol and drugs release dopamine and opioid peptides into the ventral striatum, with fast and steep dopamine release associated with the subjective sensation of being "high" [10].
Table 1: Key Neurotransmitter Systems in the Binge/Intoxication Stage
| Neurotransmitter/Neuromodulator | Direction of Change | Primary Brain Regions | Functional Consequences |
|---|---|---|---|
| Dopamine | Increase | VTA, Nucleus Accumbens | Reinforcement, incentive salience |
| Opioid Peptides | Increase | Nucleus Accumbens, VTA | Reward, hedonic processing |
| Serotonin | Increase | Dorsal Raphe, Striatum | Mood modulation, impulse control |
| γ-aminobutyric acid (GABA) | Increase | VTA, Striatum | Inhibition of dopamine neurons |
| Acetylcholine | Increase | Striatum | Arousal, attention |
| Glutamate | Increase | Prefrontal Cortex, Striatum | Learning, synaptic plasticity |
| Endocannabinoids | Variable | Striatum, VTA | Modulation of neurotransmitter release |
The specific circuitry associated with drug reward has been broadened to include multiple neural inputs and outputs that interact with the basal forebrain, including not only dopamine and opioid peptides but also GABA, glutamate, serotonin, acetylcholine, and endocannabinoid systems that act at the level of either the VTA or nucleus accumbens [10]. These neurotransmitter systems form complex mini-circuits that contribute to the overall rewarding effects of drugs and initiate the neuroplastic changes that lead to addiction.
From Incentive Salience to Habits: The Ventral to Dorsal Striatal Shift
A critical neuroadaptation occurring during the binge/intoxication stage is the progressive engagement of more dorsal aspects of the striatum as drug use transitions from occasional to chronic [11]. Initially, drug use is mediated by the ventral striatum (nucleus accumbens), which processes information about reward prediction and incentive salience. With repeated drug exposure, control over drug-seeking behavior shifts dorsally to the dorsolateral striatum, which mediates stimulus-response habits that are executed with minimal conscious awareness or goal direction [11].
This ventral-to-dorsal progression represents a fundamental shift from flexible, goal-directed actions to rigid, automatic habits characterized by compulsivity [10]. The transition is supported by serial connectivity between the ventral and dorsal striatum, with dopamine-dependent interactions critical for the development of compulsive-like drug-seeking behavior [11]. This shift explains why addicted individuals continue to seek drugs even when the drug is no longer pleasurable and despite negative consequences.
Table 2: Striatal Subregions in the Transition to Addiction
| Striatal Subregion | Primary Function in Addiction | Key Inputs | Key Outputs | Behavioral Manifestation |
|---|---|---|---|---|
| Ventral Striatum (Nucleus Accumbens) | Processing drug reward, incentive salience | Ventral Tegmental Area, Amygdala, Hippocampus, Prefrontal Cortex | Ventral Pallidum, VTA | Goal-directed drug seeking |
| Dorsomedial Striatum | Goal-directed actions, action-outcome learning | Prefrontal Cortex, Orbitofrontal Cortex | Globus Pallidus internus, Substantia Nigra pars reticulata | Flexible drug-seeking based on expected value |
| Dorsolateral Striatum | Stimulus-response habits | Sensorimotor Cortex | Globus Pallidus internus, Substantia Nigra pars reticulata | Compulsive, automatic drug-seeking |
Molecular and Cellular Neuroadaptations
Repeated drug administration during the binge/intoxication stage induces profound neuroplasticity at molecular and cellular levels within the basal ganglia. These changes include alterations in gene expression, synaptic plasticity, and neuronal morphology that collectively contribute to the transition to addiction.
Key molecular adaptations include changes in transcription factors such as ΔFosB, which accumulates in the striatum with repeated drug exposure and promotes sensitized responses to drugs [11]. Additionally, drugs of abuse alter glutamate receptor trafficking and function in the striatum, particularly increasing the surface expression of calcium-permeable AMPA receptors that lack GluR2 subunits, which enhances synaptic strength and contributes to the persistence of drug-seeking behaviors [11].
At the cellular level, chronic drug exposure induces changes in the spine density and morphology of medium spiny neurons in the striatum, with particularly prominent effects on neurons expressing dopamine D1 receptors, which form the direct pathway that promotes reward and reinforcement [11]. These structural changes facilitate the efficient processing of drug-related information and enhance the ability of drug-associated cues to trigger drug-seeking behavior.
Experimental Approaches for Studying the Binge/Intoxication Stage
Animal Models of Addiction
Animal models have been essential for elucidating the neurobiological mechanisms underlying the binge/intoxication stage of addiction [10]. While no animal model fully emulates the human condition, they permit investigations of specific signs or symptoms associated with addiction. Key models for studying this stage include:
Drug Self-Administration: This procedure allows animals to voluntarily administer drugs of abuse (typically by pressing a lever) and is considered the gold standard for modeling drug-taking behavior. Variations include fixed-ratio schedules (where each response produces drug) and progressive-ratio schedules (where increasing numbers of responses are required for each subsequent drug dose, providing a measure of motivation).
Intracranial Self-Stimulation (ICSS): ICSS measures changes in brain reward threshold following drug administration and can detect both reward-enhancing and reward-attenuating effects of drugs.
Conditioned Place Preference: This model assesses the rewarding properties of drugs by measuring the development of preference for environments paired with drug administration.
More recently developed animal models incorporate individual and strain diversity in responses to drugs, complex environments with access to and choices of alternative reinforcers, and effects of stressful stimuli, allowing investigation of neurobiological processes underlying risk for addiction and environmental factors that provide resilience [10].
Table 3: Key Research Reagent Solutions for Studying Basal Ganglia Reward Circuitry
| Research Tool Category | Specific Examples | Primary Applications | Technical Considerations |
|---|---|---|---|
| Neuroanatomical Tracers | Fluoro-Gold, Cholera Toxin B, Herpes Simplex Virus-1 | Mapping neural connections between basal ganglia subregions and cortical areas | Direction of transport (anterograde vs. retrograde), transsynaptic capability |
| Dopamine Receptor Ligands | SCH-23390 (D1 antagonist), Raclopride (D2 antagonist) | Pharmacological manipulation of dopamine signaling | Receptor specificity, blood-brain barrier penetration |
| Genetically Encoded Indicators | GRAB-DA sensors, dLight | Real-time measurement of dopamine dynamics in behaving animals | Temporal resolution, specificity, expression levels |
| Chemogenetic Tools | DREADDs (Designer Receptors Exclusively Activated by Designer Drugs) | Selective manipulation of neural activity in specific basal ganglia pathways | Receptor expression specificity, ligand pharmacokinetics |
| Optogenetic Tools | Channelrhodopsin-2, Halorhodopsin | Precise temporal control of specific neural populations in basal ganglia circuits | Light delivery limitations, tissue penetration |
| In Vivo Imaging | Fiber photometry, Microendoscopy | Monitoring neural activity during drug self-administration | Motion artifacts, spatial resolution limitations |
Human Laboratory Models and Neuroimaging
Human laboratory models parallel animal models of the specific stages of the addiction cycle and can be studied with neuroimaging techniques such as functional magnetic resonance imaging (fMRI) and positron emission tomography (PET) [10]. These approaches have been particularly valuable for validating findings from animal studies and identifying human-specific aspects of addiction neurobiology.
Key human laboratory approaches for studying the binge/intoxication stage include:
Drug Self-Administration Paradigms: Controlled laboratory settings where human participants have the opportunity to self-administer drugs of abuse, allowing researchers to measure both the subjective effects and the behavioral choices associated with drug use.
Cue-Reactivity Studies: Measurement of physiological, neural, and subjective responses to drug-related cues, which can predict relapse risk and treatment outcomes.
Neuroimaging Techniques: fMRI studies have revealed that drug-related cues activate the ventral and dorsal striatum in individuals with substance use disorders, with the magnitude of activation correlating with craving and addiction severity. PET imaging has demonstrated drug-induced dopamine release in the striatum and alterations in dopamine receptor availability associated with chronic drug use.
Signaling Pathways and Neurocircuitry Diagrams
Basal Ganglia Circuits in the Binge/Intoxication Stage
The following diagram illustrates the key neural circuits involved in the binge/intoxication stage, highlighting the transition from ventral to dorsal striatal control as addiction progresses:
Neurotransmitter Interactions in Acute Drug Reward
The following diagram illustrates the complex neurotransmitter interactions within the nucleus accumbens and VTA that mediate the acute rewarding effects of drugs during the binge/intoxication stage:
Implications for Therapeutic Development
Understanding the precise neuroadaptations occurring in the basal ganglia during the binge/intoxication stage provides critical insights for developing targeted interventions for substance use disorders. Several promising approaches emerge from this neurobiological framework:
First, medications that target specific components of the dopamine system, particularly those that normalize rather than completely block dopamine signaling, may reduce drug reward without producing profound anhedonia or other undesirable side effects [10]. Second, interventions that prevent or reverse the transition from goal-directed to habitual drug seeking by targeting the dorsal striatum may help prevent the development of compulsive drug use [11]. Third, approaches that strengthen prefrontal cortical control over striatal function may enhance the ability to resist drug-seeking impulses.
Recent research has also explored novel therapeutic targets beyond traditional neurotransmitter systems. For example, glucagon-like peptide-1 receptor agonists (GLP-1RAs), currently used for diabetes and obesity, show promise in reducing alcohol and drug consumption in preclinical models and early clinical trials [12]. These medications may modulate the neurobiological pathways underlying addictive behaviors, potentially providing a new approach to treating substance use disorders.
Additionally, research on non-opioid pain relief medications that target specific pain receptors without producing euphoria or dependence may help address the opioid crisis by providing alternatives to opioid analgesics [13]. Approaches targeting specific pain-related enzymes or heteromeric receptor complexes may provide effective pain relief without the addiction liability associated with traditional opioids.
The basal ganglia play a central role in the binge/intoxication stage of addiction, mediating both the initial rewarding effects of drugs and the progressive development of compulsive drug-seeking habits. The transition from ventral to dorsal striatal control represents a critical shift from voluntary drug use to addiction, characterized by loss of behavioral flexibility and emergence of compulsive drug-seeking. This transition is supported by complex neuroadaptations in dopaminergic, glutamatergic, and other neurotransmitter systems that alter the function of basal ganglia circuits.
Detailed understanding of these processes provides a heuristic framework for identifying novel molecular targets for medication development and behavioral interventions. Future research should focus on elucidating the precise mechanisms underlying individual differences in vulnerability to the transition from controlled use to addiction, as well as developing interventions that can specifically reverse the neuroadaptations in dorsal striatal circuits that drive compulsive drug seeking. By targeting the specific neurobiological processes underlying the binge/intoxication stage, it may be possible to develop more effective strategies for preventing and treating substance use disorders.
Addiction is a chronically relapsing disorder characterized by a compulsive cycle of intoxication, withdrawal, and craving [11]. The withdrawal/negative affect stage represents a critical phase in this cycle, often termed the "dark side" of addiction, where a shift from positive reinforcement (taking drugs to feel good) to negative reinforcement (taking drugs to avoid feeling bad) occurs [10] [14]. This stage is defined by the emergence of a negative emotional state—including dysphoria, anxiety, and irritability—when access to the drug is prevented [10] [11]. The neurobiological substrate for this phenomenon is the extended amygdala, a macrostructure that integrates brain reward and stress systems and becomes dysregulated during the development of dependence [14]. This technical guide provides an in-depth analysis of the extended amygdala's role in stress system dysregulation during the withdrawal/negative affect stage, framed within the broader context of the neural circuitry of addiction involving the basal ganglia and prefrontal cortex.
Neuroanatomical Framework: The Extended Amygdala
Definition and Core Structures
The extended amygdala is a neuroanatomical construct that forms a continuous basal forebrain macrostructure, linking the basal ganglia with the limbic system [14]. It comprises three primary components:
- Bed nucleus of the stria terminalis (BNST)
- Central nucleus of the amygdala (CeA)
- Sublenticular substantia innominata (which includes a transition zone in the medial shell of the nucleus accumbens) [14]
This structure serves as a common anatomical substrate for both the acute rewarding effects of drugs and the negative motivational state associated with compulsive drug administration [14]. It receives afferent connections from limbic structures such as the basolateral amygdala and hippocampus and projects to hypothalamic and brainstem areas that mediate the behavioral and physiological manifestations of emotional states [14].
Functional Role in Motivation
The extended amygdala is positioned as a key interface in the integration of emotional and motivational information. It is a fundamental component of the brain's reward circuitry, with acute drug administration producing robust dopamine release in the shell of the nucleus accumbens, a key ventral striatal region within the extended amygdala network [14]. However, with repeated drug administration and the transition to dependence, the extended amygdala undergoes significant neuroadaptations that shift its role from processing reward to mediating the stressful and aversive aspects of withdrawal [14].
Neurobiological Mechanisms of the Withdrawal/Negative Affect Stage
Key Neurotransmitter Systems
The dysregulation of the extended amygdala during withdrawal involves a complex interplay of multiple neurotransmitter systems. The table below summarizes the primary neurochemical changes observed during the withdrawal/negative affect stage.
Table 1: Key Neurotransmitter Changes in the Withdrawal/Negative Affect Stage
| Neurotransmitter/Neuromodulator | Direction of Change | Primary Brain Region(s) | Functional Consequence |
|---|---|---|---|
| Corticotropin-Releasing Factor (CRF) | Increase [10] | Central Amygdala, BNST [14] | Increased anxiety-like behavior and stress responsiveness |
| Dynorphin | Increase [10] | Extended Amygdala [10] | Dysphoria and aversive states via kappa-opioid receptor activation |
| Norepinephrine | Increase [10] | Extended Amygdala [10] | Enhanced stress and anxiety responses |
| Dopamine | Decrease [10] | Ventral Tegmental Area [10] | Reduced reward sensitivity, anhedonia |
| Serotonin | Decrease [10] | Not Specified | Mood disturbances, dysphoria |
| Endocannabinoids | Decrease [10] | Not Specified | Reduced modulation of stress and reward |
| Neuropeptide Y | Decrease [10] | Not Specified | Diminished anti-stress, anxiolytic effects |
The Allostatic Model of Addiction
The neuroadaptations in the extended amygdala can be understood through an allostatic model of addiction [14]. Allostasis refers to the process of maintaining stability through change; in addiction, this involves a chronic deviation of the brain's reward set point to counteract the repeated perturbing effects of drugs [14]. This model posits that:
- Counter-adaptive mechanisms are engaged to overcome the chronic presence of the drug and restore normal function.
- These mechanisms do not return to the original homeostatic set point but instead create a new allostatic state.
- This state is characterized by chronic decreases in reward system function and recruitment of brain stress systems.
- The resulting allostatic load manifests as the persistent negative emotional state that defines the "dark side" of addiction, driving compulsive drug taking through negative reinforcement [14].
The following diagram illustrates the key neurocircuits and neurotransmitter systems within the extended amygdala that are dysregulated during the withdrawal/negative affect stage, creating the allostatic state that drives negative reinforcement.
Experimental Approaches and Methodologies
Animal Models of Motivational Withdrawal
Research on the neurobiology of the withdrawal/negative affect stage has been advanced through the development and use of sophisticated animal models that capture the motivational aspects of dependence, beyond mere physical withdrawal symptoms [14]. Key validated models include:
- Intracranial Self-Stimulation (ICSS) Thresholds: This model measures brain reward thresholds. During withdrawal from all major drugs of abuse, elevations in ICSS thresholds are observed, reflecting a decrease in the rewarding value of electrical brain stimulation—an operational measure of dysphoria [14].
- Conditioned Place Aversion: Animals learn to avoid an environment previously paired with the aversive state of withdrawal, providing a direct measure of the negative motivational component of withdrawal [14].
- Drug Self-Administration: Dependent animals self-administer greater quantities of the drug, particularly during periods of anticipated withdrawal, demonstrating the negative reinforcement process [14]. These models have construct and predictive validity for the human condition, as they parallel the dysphoria, anxiety, and irritability reported by individuals with substance use disorders during abstinence [10].
Neuropharmacological and Neurochemical Techniques
The specific neuroadaptive mechanisms within the extended amygdala have been elucidated using a range of experimental protocols:
- Microinjection Studies: Cannula-directed injections of receptor agonists and antagonists into discrete components of the extended amygdala (e.g., CeA, BNST) during withdrawal. For example, microinjection of a CRF receptor antagonist into the CeA reverses the anxiogenic-like effects of ethanol withdrawal [14].
- In Vivo Microdialysis: This technique allows for the measurement of extracellular neurotransmitter levels in specific brain regions of awake, freely moving animals. Studies using microdialysis have documented increased extracellular CRF in the CeA and BNST during withdrawal from alcohol, opiates, cocaine, and tetrahydrocannabinol (THC) [14].
- Electrophysiological Recordings: These assess changes in neuronal excitability and synaptic transmission within the extended amygdala circuit during withdrawal states.
- Molecular and Genetic Approaches: These include measures of gene expression (e.g., in situ hybridization), protein levels (immunohistochemistry, Western blot), and the use of knockout mice or viral vector-mediated gene transfer to manipulate specific genes within the extended amygdala.
Table 2: Key Experimental Protocols for Studying Extended Amygdala in Withdrawal
| Experimental Protocol | Key Measurement | Example Application | Technical Considerations |
|---|---|---|---|
| Intracranial Self-Stimulation (ICSS) | Brain reward threshold | Quantifying dysphoria during drug withdrawal | Requires stereotaxic surgery; threshold elevations indicate negative emotional state |
| In Vivo Microdialysis | Extracellular neurotransmitter levels | Measuring CRF release in CeA during withdrawal | High temporal resolution; technically challenging in small brain regions |
| Local Microinjection | Behavioral response to receptor ligands | CRF antagonist reversal of withdrawal-anxiety | Verifies anatomical specificity of neurochemical mechanisms |
| Conditioned Place Aversion | Time spent in withdrawal-paired context | Assessing motivational aversion to withdrawal | Requires careful conditioning protocol; reflects learned aversion |
| Fos Expression Mapping | Neuronal activation patterns | Identifying brain regions activated during withdrawal | Provides snapshot of activation; does not imply necessity for behavior |
The Scientist's Toolkit: Research Reagent Solutions
The following table catalogs essential research reagents and tools used in the featured experiments for investigating the role of the extended amygdala in addiction.
Table 3: Essential Research Reagents for Extended Amygdala Research
| Reagent / Tool | Category | Primary Function/Application | Example Use Case |
|---|---|---|---|
| CRF Receptor Antagonists | Pharmacological Tool | Block CRF receptors to test their role in stress responses | Reverses anxiety-like effects of ethanol withdrawal when microinjected into CeA [14] |
| Dopamine Receptor Ligands | Pharmacological Tool | Target specific dopamine receptor subtypes (D1, D2, D3) | SCH 23390 (D1 antagonist) decreases cocaine reinforcement [14] |
| GABA Receptor Agonists/Antagonists | Pharmacological Tool | Modulate inhibitory neurotransmission in extended amygdala | GABA-A antagonism in extended amygdala decreases ethanol self-administration [14] |
| Cre-loxP System | Genetic Tool | Enables cell-type-specific gene knockout or expression | Targeting gene deletion in CRF neurons of the extended amygdala |
| AAV Vectors | Viral Vector | For targeted gene expression or silencing in specific brain regions | Overexpressing or knocking down neuropeptide Y in BNST to study anxiety |
| Radioactive Tracers | Imaging Tool | Label receptors or transporters for autoradiography or PET | Quantifying dopamine transporter levels in striatum during recovery [15] |
| c-Fos Antibodies | Immunological Tool | Marker of neuronal activation following behavioral challenges | Mapping neuronal activation in extended amygdala during withdrawal |
Integration with Broader Addiction Circuitry
The dysfunction of the extended amygdala during the withdrawal/negative affect stage does not occur in isolation. It is part of a broader network of interconnected neural circuits that become dysregulated in addiction, including the basal ganglia and prefrontal cortex [10] [3] [16].
- Basal Ganglia: The ventral striatum (nucleus accumbens) is a key node in the binge/intoxication stage, mediating the initial rewarding and reinforcing effects of drugs. With the development of addiction, there is a progressive shift in control from the ventral to the dorsal striatum, which mediates the habitual and compulsive drug-seeking behaviors that characterize addiction [17].
- Prefrontal Cortex: This region, particularly the orbitofrontal cortex (OFC), anterior cingulate cortex (ACC), and dorsolateral prefrontal cortex (DLPFC), is critical for executive function, including inhibitory control, decision-making, and salience attribution [16]. In addiction, the prefrontal cortex becomes compromised, leading to the syndrome of impaired response inhibition and salience attribution (iRISA), which contributes to the craving and relapse observed in the preoccupation/anticipation stage [16].
The extended amygdala interacts with these circuits, and its dysregulation contributes to the negative emotional state that drives negative reinforcement, thereby fueling the compulsive cycle of addiction. The following diagram illustrates this broader integrated circuitry.
The extended amygdala serves as a critical nexus for the stress dysregulation that defines the "dark side" of addiction in the withdrawal/negative affect stage. The recruitment of stress neurotransmitters like CRF and dynorphin, combined with the weakening of anti-stress systems, within this circuitry creates a powerful allostatic state that drives compulsive drug seeking through negative reinforcement [10] [14]. A comprehensive understanding of this framework not only elucidates the neurobiological mechanisms underlying addiction but also highlights promising targets for therapeutic intervention. Future research should focus on further delineating the specific microcircuits and cell populations within the extended amygdala that contribute to this dysregulation, as well as exploring the dynamic interactions between the extended amygdala, basal ganglia, and prefrontal cortex across the different stages of the addiction cycle. Such work will be vital for developing novel, effective strategies for the diagnosis, treatment, and prevention of substance use disorders.
Addiction is currently understood as a chronic and relapsing brain disorder, marked by specific neuroadaptations that drive compulsive substance use despite negative consequences [18]. The neurobiological framework of addiction is conceptualized as a three-stage cycle—binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation—that worsens over time and involves distinct but interconnected brain circuits [10] [11]. This review focuses on the preoccupation/anticipation stage, a critical phase characterized by intense craving and deficits in executive function that significantly contribute to relapse. During this stage, the prefrontal cortex (PFC), the brain's central hub for executive function, becomes profoundly dysregulated. This dysruption represents a hijacking of the brain's highest-order cognitive control systems, leading to a dramatic inability to regulate the urge to seek and use substances, even after prolonged periods of abstinence [18] [10]. The PFC does not operate in isolation; its dysfunction in addiction is deeply intertwined with the altered reward processing of the basal ganglia and the stress responses of the extended amygdala, forming a dysfunctional neural triad that perpetuates the addiction cycle [3] [11].
Neurobiological Mechanisms of PFC Dysfunction
The "Go" and "Stop" Systems: A Framework for Executive Dysfunction
The executive functions governed by the PFC can be heuristically understood through the lens of a "Go" system and a "Stop" system [18] [19]. The "Go" system, involving circuits such as the dorsolateral prefrontal cortex and anterior cingulate, is responsible for motivated, goal-directed behavior and planning. In substance use disorders, this system is strengthened and co-opted by addiction, leading to heightened impulsivity and a powerful drive to seek drugs. Concurrently, the "Stop" system, critical for inhibiting impulsive actions and distracting emotions, is significantly weakened. This dual pathology creates a neurological scenario where the "gas pedal" is stuck while the "brakes" are failing, making relapse almost inevitable in the face of triggers [19]. This imbalance is a core manifestation of the compromised executive control that defines the preoccupation/anticipation stage.
Key Neurotransmitter Systems and Molecular Pathways
The dysfunction of the PFC's "Go" and "Stop" systems is mediated by specific neurotransmitter alterations. Glutamate, the primary excitatory neurotransmitter in the PFC, plays a critical role. During the preoccupation/anticipation stage, there is a documented increase in glutamate signaling from the PFC to other addiction-related regions, such as the basal ganglia and extended amygdala [10]. This heightened glutamatergic drive is thought to fuel the craving and compulsive drug-seeking behaviors. Other neurotransmitter systems are also recruited, including increases in corticotropin-releasing factor (CRF), dopamine, and hypocretin (orexin), which collectively contribute to the stress, motivation, and preoccupation associated with craving [10]. At the molecular level, these neurotransmitter changes induce neuroadaptations, including alterations in intracellular signaling and gene expression, which underlie the long-lasting nature of addiction and the vulnerability to relapse [20].
Table 1: Key Neurotransmitter Changes in the Preoccupation/Anticipation Stage
| Neurotransmitter/Neuromodulator | Change in Preoccupation/Anticipation Stage | Primary Function in this Stage |
|---|---|---|
| Glutamate [10] | Increase | Fuels craving and compulsive drug-seeking; primary transmitter from PFC to subcortical structures. |
| Dopamine [10] | Increase | Modulates motivation and the salience of drug-associated cues. |
| Corticotropin-Releasing Factor (CRF) [10] | Increase | Mediates stress responses that drive drug-seeking to relieve negative affect. |
| Hypocretin (Orexin) [10] | Increase | Regulates arousal, wakefulness, and the motivational drive for drug seeking. |
| Serotonin [10] | Increase | May influence mood and impulse control, though its role is complex. |
Experimental Models and Research Methodologies
Animal Models of Craving and Relapse
Research into the neurobiology of the preoccupation/anticipation stage has been advanced through rigorously validated animal models that probe specific behavioral constructs. Key among these are reinstatement models, which are the most widely used for studying relapse. In this model, an animal is trained to self-administer a drug, followed by a period of extinction where the drug is no longer available. Subsequently, "relapse" is triggered by exposure to a drug-associated cue, a stressor, or a small "priming" dose of the drug itself. The behavioral output—resumption of drug-seeking responses—is quantified, allowing researchers to investigate the neural substrates of relapse-like behavior [10] [11]. Another critical model is the incubation of craving model, where cue-induced craving is shown to grow, rather than diminish, during prolonged forced abstinence. This model is particularly relevant for understanding the persistent nature of craving in humans and has been instrumental in identifying time-dependent neuroadaptations in the PFC and its circuits [11].
Human Neuroimaging Approaches
Human studies, particularly using functional Magnetic Resonance Imaging (fMRI), have complemented animal research by allowing investigators to "see" inside the living human brain. fMRI studies consistently reveal that exposure to drug-related cues (e.g., pictures of drug paraphernalia) produces robust activation in a distributed network that includes the orbitofrontal cortex, dorsolateral prefrontal cortex, anterior cingulate, and insula in individuals with substance use disorders [3] [11]. This activation pattern correlates with subjective reports of craving. Furthermore, structural MRI studies often show a reduced density of grey matter and compromised integrity of white matter tracts within the PFC, providing a anatomical correlate for the observed executive function deficits [3]. These imaging technologies are vital for translating findings from animal models to the human condition and for identifying individual differences in neural circuitry that may predict vulnerability to relapse.
Table 2: Core Experimental Protocols for Studying the Preoccupation/Anticipation Stage
| Experimental Approach | Core Methodology | Measurable Outputs |
|---|---|---|
| Drug Reinstatement Model [10] [11] | 1. Train animal to self-administer drug (e.g., lever press).2. Extinguish behavior (drug unavailable).3. Apply trigger (cue, stress, prime) and measure drug-seeking responses. | - Number of active lever presses during reinstatement.- Latency to first response. |
| Incubation of Craving Model [11] | 1. Train animal to self-administer drug.2. Enforce forced abstinence (e.g., 1 day vs. 30 days).3. Present drug-associated cue and measure seeking behavior. | - Level of cue-induced drug seeking after different abstinence periods. |
| Human Cue-Reactivity fMRI [3] | 1. Present drug-related vs. neutral cues to abstinent individuals in scanner.2. Measure brain activation using BOLD signal.3. Collect simultaneous self-report of craving. | - BOLD activation in PFC, striatum, amygdala.- Correlation between neural activity and craving score. |
Visualizing the Neural Circuitry of Addiction
The following diagram illustrates the key brain regions and their interactions during the three stages of the addiction cycle, with a specific emphasis on the preoccupation/anticipation stage and the role of the prefrontal cortex.
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Research Reagents for Investigating the Preoccupation/Anticipation Stage
| Reagent / Material | Function in Experimental Research |
|---|---|
| Dopamine Receptor Antagonists (e.g., SCH 23390 for D1, Eticlopride for D2) [11] | Used to pharmacologically block specific dopamine receptor subtypes in order to dissect their contribution to cue-induced reinstatement and incentive salience. |
| Glutamate Receptor Modulators (e.g., NMDA and AMPA receptor antagonists) [10] [11] | Critical for probing the role of glutamatergic transmission from the PFC to the nucleus accumbens and other striatal regions in driving compulsive drug-seeking. |
| CRF Receptor Antagonists [10] [11] | Used to investigate the contribution of brain stress systems to relapse, particularly stress-induced reinstatement of drug-seeking behavior. |
| Viral Vector Systems (e.g., AAV for DREADDs or Cre-lox technology) [11] | Enable cell-type-specific and circuit-specific manipulation of neuronal activity (inhibition or excitation) in defined pathways, such as PFC→NAc projections. |
| Positron Emission Tomography (PET) Radioligands (e.g., for D2/3 receptors, dopamine release) [3] [10] | Allow for non-invasive quantification of receptor availability and neurotransmitter dynamics in the living human brain, linking neurochemistry to behavior. |
| c-Fos and Other Immediate Early Gene Markers | Serve as a histological marker of neuronal activation to map brain circuits (e.g., PFC, amygdala, striatum) that are engaged during cue-induced craving or relapse tests. |
Implications for Therapeutics and Future Research
The detailed neurobiological understanding of PFC dysfunction in the preoccupation/anticipation stage opens promising avenues for therapeutic intervention. Strategies aimed at normalizing glutamate transmission, such as modulating metabotropic glutamate receptors (mGluRs), are under active investigation to reduce hyperglutamatergic states that drive craving [10] [11]. Furthermore, interventions designed to strengthen "Stop" system function are a cornerstone of behavioral therapies. Techniques used in Cognitive Behavioral Therapy (CBT) and mindfulness-based relapse prevention directly target the PFC, helping individuals improve inhibitory control and emotional regulation [19]. Non-invasive brain stimulation techniques like transcranial magnetic stimulation (TMS) are also being explored to directly modulate PFC activity and reduce craving in patients with substance use disorders [18]. Future research will continue to delineate the molecular and genetic underpinnings of the individual vulnerability for PFC dysregulation, paving the way for more personalized and effective treatments for addiction.
This whitepaper synthesizes current neurobiological research on five key neurotransmitter and neuromodulator systems—dopamine, opioid peptides, corticotropin-releasing factor (CRF), glutamate, and dynorphin—and their integrated roles in the neural circuitry of addiction. The transition from controlled substance use to compulsive addiction involves specific neuroadaptations within the basal ganglia, extended amygdala, and prefrontal cortex. Understanding these mechanisms provides a framework for developing targeted therapeutic interventions for substance use disorders. This technical guide summarizes quantitative findings, experimental methodologies, and visualization tools to support ongoing research and drug development efforts.
Drug addiction is conceptualized as a chronic relapsing disorder characterized by a three-stage cycle—binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation—each mediated by specific neural circuits [11]. The progression to addiction involves neuroplasticity in three key brain regions: the basal ganglia (reward and habit formation), the extended amygdala (stress and negative affect), and the prefrontal cortex (executive control and regulation) [7] [3]. These interconnected regions undergo significant molecular and functional changes during the transition from occasional use to addiction, primarily driven by adaptations in the neurotransmitter systems detailed in this review.
Table: Key Brain Regions in Addiction Neurocircuitry
| Brain Region | Primary Function in Addiction | Associated Neurotransmitters |
|---|---|---|
| Basal Ganglia | Reward processing, habit formation, motivation | Dopamine, Opioid Peptides |
| Extended Amygdala | Stress response, negative affect, anxiety | CRF, Dynorphin |
| Prefrontal Cortex | Executive control, decision-making, impulse regulation | Glutamate, Dopamine |
Dopamine: The Reinforcement Signal
Mechanisms of Action
Dopamine, particularly within the mesolimbic pathway projecting from the ventral tegmental area (VTA) to the nucleus accumbens (NAc) in the basal ganglia, is central to the initial reinforcing effects of drugs [21]. Addictive substances hijack this evolutionarily conserved reward system. Different drug classes manipulate the dopamine system through distinct mechanisms:
- Cocaine & Amphetamines: These psychostimulants directly target dopamine transporters (DAT). Cocaine binds to DAT and inhibits dopamine reuptake, increasing extracellular dopamine concentrations [22]. Amphetamines are transported into the neuron, promoting reverse transport of dopamine through DAT and increasing its release into the synapse [22].
- Opioids & Nicotine: These drugs indirectly increase dopamine in the NAc by activating opioid receptors or nicotinic acetylcholine receptors on GABAergic interneurons in the VTA. This disinhibits dopamine neurons, leading to increased firing and dopamine release [21].
- Alcohol: Alcohol's effects are more complex, influencing multiple neurotransmitter systems (GABA, glutamate, serotonin), but its rewarding properties are also linked to increased NAc dopamine levels [21].
Role in the Addiction Cycle
While dopamine is crucial for the initial binge/intoxication stage, its role evolves with chronic drug use. The large dopamine surges "teach" the brain to associate drug use with cues and contexts, powerfully reinforcing the behavior [7]. However, with repeated exposure, the reward circuit adapts. The basal ganglia becomes less sensitive to dopamine, diminishing the person's ability to experience pleasure from naturally rewarding activities and the drug itself—a phenomenon known as tolerance [7]. This leads to a shift in motivation: drug use becomes less about seeking pleasure ("liking") and more driven by habit and the desire to relieve the negative state of withdrawal ("wanting") [7] [11].
Diagram Title: Dopamine Signaling Pathways in Addiction
Experimental Protocols for Dopamine Research
Fast-Scan Cyclic Voltammetry (FSCV) for Real-Time Dopamine Measurement
- Principle: An electroanalytical technique used to measure real-time, rapid changes in extracellular dopamine concentrations in brain slices or in vivo.
- Procedure:
- A carbon-fiber microelectrode is implanted into the target brain region (e.g., NAc).
- A triangular waveform voltage potential is applied to the electrode.
- As the voltage scans, dopamine molecules at the electrode surface are oxidized and reduced, producing a characteristic current.
- The resulting current is measured, and the dopamine concentration is determined based on the oxidation peak current.
- Key Applications: Measuring phasic dopamine release events elicited by drug administration, drug-predictive cues, or natural rewards.
Opioid Peptides and Dynorphin: The Opposing Forces of Reward
Endogenous Opioid Systems
The endogenous opioid system, comprising mu (MOR), delta (DOR), and kappa (KOR) opioid receptors and their ligands (e.g., endorphins, enkephalins, dynorphin), plays a critical modulatory role in reward, pain, and stress [23]. Mu-opioid receptor (MOR) activation in key reward regions like the NAc and VTA is a primary mechanism for the euphoric and analgesic effects of natural rewards and exogenous opioids like heroin and morphine [21]. This occurs through disinhibition of dopamine neurons, similar to the mechanism of other indirect drugs of abuse.
Dynorphin/KOR System in Addiction
In contrast to the reward-enhancing effects of the mu-opioid system, the dynorphin/kappa-opioid receptor (KOR) system is considered a powerful anti-reward system [24] [23]. Dynorphin is the primary endogenous ligand for KOR. Its activation produces dysphoric, aversive, and pro-depressive effects [23].
- Molecular Signaling: KORs are G-protein coupled receptors (GPCRs). Their activation leads to:
- Opening of G-protein-gated inwardly rectifying potassium (GIRK) channels.
- Inhibition of voltage-gated calcium channels.
- The net effect is hyperpolarization of the neuron and reduced neurotransmitter release [24].
- KOR activation also engages downstream signaling cascades like the MAPK pathway (ERK1/2, JNK, p38), which can lead to long-term transcriptional changes [24].
- Role in Stress and Addiction: Stress exposure and chronic drug use upregulate the dynorphin/KOR system in the extended amygdala and NAc [24] [23]. This upregulation is thought to be a key mechanism driving the negative emotional state (dysphoria, anxiety, irritability) characteristic of drug withdrawal and protracted abstinence. Consequently, the addicted individual may use drugs not only to get high but to achieve temporary relief from this KOR-mediated negative affect [7] [11].
Table: Key Differences Between Mu and Kappa Opioid Systems
| Feature | Mu-Opioid Receptor (MOR) System | Kappa-Opioid Receptor (KOR) System |
|---|---|---|
| Primary Endogenous Ligand | β-endorphin, Enkephalins | Dynorphin |
| Affective Response | Euphoria, Pleasure | Dysphoria, Aversion |
| Role in Addiction Cycle | Drives Binge/Intoxication stage | Drives Withdrawal/Negative Affect stage |
| Effect on Dopamine | Increases NAc dopamine release | Decreases NAc dopamine release |
| Therapeutic Target | Agonists for maintenance (methadone), antagonists to block reward (naltrexone) | Antagonists to treat negative affect and prevent relapse |
Corticotropin-Releasing Factor (CRF): The Stress Axis
Central Role in Stress Response
Corticotropin-releasing factor (CRF) is a 41-amino-acid neuropeptide that is the primary activator of the body's stress response, the hypothalamic-pituitary-adrenal (HPA) axis [25]. In response to a stressor, hypothalamic CRF is released, triggering a cascade that results in the secretion of cortisol (corticosterone in rodents) from the adrenal glands.
Extrahypothalamic CRF and Addiction
Beyond the HPA axis, extrahypothalamic CRF systems, particularly within the extended amygdala (central amygdala, bed nucleus of the stria terminalis), are critically involved in the behavioral responses to stress [25]. This system becomes dysregulated during the development of addiction.
- Acute Withdrawal: Upon cessation of drug use in a dependent individual, CRF release increases dramatically within the extended amygdala. This surge is a key mediator of the anxiety, irritability, and emotional distress that define the withdrawal/negative affect stage [25] [11].
- Protracted Abstinence: The CRF system in the extended amygdala remains hyperactive long after acute withdrawal has subsided. This persistent sensitization of brain stress systems contributes to the persistent negative emotional state and heightened vulnerability to stress-induced relapse [25].
Experimental Protocols for CRF Research
Intracranial Microinjection of CRF Receptor Antagonists
- Objective: To assess the causal role of extrahypothalamic CRF in addiction-related behaviors.
- Procedure:
- Stereotaxic surgery is performed to implant guide cannulae above the target brain region (e.g., central amygdala).
- After recovery, an injection cannula is inserted through the guide to deliver a small volume of a CRF receptor antagonist (e.g., CP-154,526 for CRF1) or vehicle.
- Behavioral assays are conducted post-injection, such as:
- Anxiety-like behaviors: Elevated plus maze, open field test.
- Drug-seeking: Stress-induced reinstatement of drug self-administration.
- Outcome Measures: Reduction in withdrawal-induced anxiety and blockade of stress-triggered relapse demonstrate the involvement of local CRF signaling [25].
Glutamate: The Driver of Plasticity and Relapse
Glutamate Homeostasis
Glutamate is the primary excitatory neurotransmitter in the brain. Its homeostasis—the balance between release and reuptake—is critical for normal synaptic communication and plasticity. Key components include:
- Vesicular Release: Action potential-dependent release from neurons.
- Astrocytic Reuptake: The excitatory amino acid transporter 2 (EAAT2/GLT-1) on astrocytes is responsible for the vast majority of glutamate clearance from the synapse, preventing excitotoxicity [22].
- Cystine-Glutamate Exchanger (xCT): Located on astrocytes, this antiporter releases glutamate into the extrasynaptic space in exchange for cystine, helping to maintain basal glutamate tone [22].
Disruption in Addiction
Chronic drug use, particularly psychostimulants, disrupts glutamate homeostasis in the NAc and prefrontal cortex [22].
- Downregulation of GLT-1: Chronic cocaine and alcohol use lead to reduced expression and function of the GLT-1 transporter. This impairs glutamate clearance, leading to elevated extracellular glutamate levels and disrupted synaptic signaling [22].
- Altered xCT Function: Drug exposure also alters the cystine-glutamate exchanger, contributing to a pathological shift in basal glutamate levels [22].
These adaptations contribute to a state of synaptic plasticity that underlies the formation of powerful drug-context associations and memories. During abstinence, cue-induced craving is linked to a transient, massive release of glutamate in the NAc, driven by projections from the prefrontal cortex, amygdala, and hippocampus [26]. This glutamate surge is a critical mediator of the preoccupation/anticipation (craving) stage and is a potent trigger for relapse.
Diagram Title: Glutamate Dysregulation in Addiction
Integrated View: Neurotransmitter Dynamics Across the Addiction Cycle
The progression of addiction involves a sequential recruitment of different neural circuits and neurotransmitter systems, as summarized in the table below.
Table: Neurotransmitter Dynamics Across the Three-Stage Addiction Cycle
| Addiction Stage | Key Brain Regions | Primary Neurotransmitter Dynamics |
|---|---|---|
| Binge/Intoxication | Basal Ganglia (VTA, NAc) | ↑ Dopamine, ↑ Opioid Peptides (MOR): Drive reward and reinforcement. |
| Withdrawal/Negative Affect | Extended Amygdala | ↑ CRF, ↑ Dynorphin: Drive stress, dysphoria, and negative emotional state. ↓ Dopamine: Reduces reward sensitivity. |
| Preoccupation/Anticipation (Craving) | Prefrontal Cortex, Hippocampus, Basolateral Amygdala, Dorsal Striatum | ↑ Glutamate: Mediates cue-induced craving and relapse. Dysregulated Dopamine: Impairs motivation and salience attribution. |
The Scientist's Toolkit: Key Research Reagents
Table: Essential Research Reagents for Studying Addiction Neurobiology
| Reagent / Tool | Class / Target | Primary Research Application | Example Use |
|---|---|---|---|
| JDTic | KOR Antagonist | Blocking KOR to study its role in stress-induced relapse and depressive-like behaviors. | Prevents stress-induced reinstatement of cocaine-seeking in rats [23]. |
| CP-154,526 | CRF1 Receptor Antagonist | Probing the role of the CRF system in anxiety and withdrawal. | Reduces anxiety-like behavior during ethanol withdrawal [25]. |
| Naltrexone | Opioid Receptor Antagonist (primarily MOR) | Studying role of opioid signaling in reward and as a pharmacotherapy. | Reduces alcohol consumption in humans and animal models [21]. |
| Ceftriaxone | β-lactam Antibiotic / GLT-1 Upregulator | Restoring glutamate homeostasis. | Upregulates GLT-1 expression, reducing cocaine and alcohol relapse in rats [22]. |
| DAT-Cre Mouse Line | Transgenic Model (Dopamine Neurons) | Cell-specific manipulation of gene expression in dopamine neurons. | Studying dopamine-specific effects of gene deletion or optogenetic stimulation on drug-related behaviors. |
| Fast-Scan Cyclic Voltammetry (FSCV) | Electrochemical Technique | Real-time measurement of dopamine dynamics in vivo. | Measuring phasic dopamine release to a drug-paired cue. |
Understanding the integrated roles of dopamine, opioid peptides, CRF, glutamate, and dynorphin provides a roadmap for novel therapeutic development. Promising avenues include:
- GLP-1 Receptor Agonists: Originally developed for diabetes and obesity, this class of drugs shows promise in reducing alcohol and drug intake in preclinical and early clinical trials. They are believed to work by modulating neurobiological pathways underlying addictive behaviors, potentially by reducing dopamine release in the reward circuit [12].
- KOR Antagonists: As the dynorphin/KOR system is a key driver of the negative affect in withdrawal, KOR antagonists are being actively investigated for their potential to treat depressive states and prevent relapse in substance use disorders [24] [23].
- CRF1 Receptor Antagonists: Targeting the brain stress system remains a compelling strategy for treating the negative emotional state that drives negative reinforcement in addiction [25].
- Glutamate Modulators: Efforts to restore glutamate homeostasis, for instance by upregulating the GLT-1 transporter with compounds like ceftriaxone, represent a promising approach to prevent cue-induced craving and relapse [22].
In conclusion, addiction is a disorder of complex, interacting neurochemical systems. The transition from voluntary use to compulsive addiction reflects a shift from dopamine-driven positive reinforcement to CRF/dynorphin-driven negative reinforcement, with a progressive loss of prefrontal cortical control and a growing influence of glutamate-driven learned responses. Effective treatments will likely require combination strategies that target multiple components of this dysfunctional circuitry.
Methodological Approaches: From Animal Models to Human Neuroimaging in Circuit Analysis
Addiction is a chronic, relapsing disorder characterized by compulsion to seek and take a drug, loss of control in limiting intake, and emergence of a negative emotional state when access to the drug is prevented [10]. The contemporary neuroscientific understanding conceptualizes addiction as a brain disease driven by dramatic dysregulation of motivational circuits, caused by a combination of exaggerated incentive salience, habit formation, reward deficits, stress surfeits, and compromised executive function [10] [3]. This understanding has emerged predominantly from carefully validated animal models that permit investigations at molecular, cellular, and circuit levels not possible in human subjects.
Research has identified three major neurocircuitry domains that undergo specific neuroadaptations during the transition to addiction: the basal ganglia (reward and habit formation), the extended amygdala (stress and negative affect), and the prefrontal cortex (executive control) [3]. These systems form the core neural substrates of the widely accepted three-stage addiction cycle—binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation—which worsens over time and involves specific neuroplastic changes in brain reward, stress, and executive function systems [10] [11]. The following sections detail how animal models have been developed to validate these constructs, elucidate their underlying mechanisms, and explore individual vulnerabilities that determine transition from casual use to addiction.
The Addiction Cycle: Neurocircuitry and Behavioral Validation
Stage 1: Binge/Intoxication
The binge/intoxication stage primarily involves the reinforcing, rewarding effects of drugs and the development of incentive salience and drug-seeking habits. This stage predominantly engages the basal ganglia, including the nucleus accumbens and dorsal striatum, with key roles for dopamine and opioid peptides [10] [3].
Core Neurocircuitry: The mesolimbic dopamine system, originating in the ventral tegmental area (VTA) and projecting to the nucleus accumbens, is central to drug reward. All abused drugs increase dopamine signaling in the nucleus accumbens, though through different initial molecular targets [3] [27]. Positron emission tomography (PET) studies in humans confirm that intoxicating doses of alcohol and drugs release dopamine and opioid peptides into the ventral striatum, with fast and steep dopamine release associated with the subjective "high" [10]. As addiction progresses, there is a shift from ventral to dorsal striatal control, mediating the transition from voluntary to habitual drug seeking [11].
Validating Animal Models and Protocols:
- Drug Self-Administration: This is the gold standard model where animals (typically rodents or nonhuman primates) perform an operant response (e.g., lever press, nose poke) to receive intravenous drug infusions. The model demonstrates that animals will readily self-administer the same drugs abused by humans, and patterns of intake often mirror human patterns [27]. Protocols typically involve daily sessions (1-3 hours) where each operant response produces a drug delivery (fixed-ratio 1 schedule). To model the binge/intoxication stage, Long Access (LgA) protocols extend session duration (e.g., 6+ hours), leading to escalation of drug intake, a key feature of this stage [28].
- Conditioned Place Preference (CPP): In this non-contingent model, animals experience drug effects in one distinct context and vehicle in another. After several pairings, drug-free testing measures preference for the drug-paired context, establishing the rewarding properties of the drug [28].
Table 1: Neurotransmitter Systems in the Binge/Intoxication Stage
| Neurotransmitter/Neuromodulator | Direction of Change | Primary Brain Regions | Behavioral Function |
|---|---|---|---|
| Dopamine | Increase | Ventral Tegmental Area, Nucleus Accumbens | Reward, incentive salience |
| Opioid Peptides | Increase | Nucleus Accumbens, Ventral Striatum | Pleasure, euphoria |
| GABA | Increase | Ventral Tegmental Area | Disinhibition of dopamine neurons |
| Glutamate | Increase | Prefrontal Cortex to Nucleus Accumbens | Learning, habit formation |
Stage 2: Withdrawal/Negative Affect
The withdrawal/negative affect stage is characterized by a "motivational withdrawal syndrome" manifesting as dysphoria, anxiety, irritability, and heightened stress sensitivity when drug access is prevented. This stage primarily involves the extended amygdala and its stress systems [10] [11].
Core Neurocircuitry: The extended amygdala (including the central nucleus of the amygdala, bed nucleus of the stria terminalis, and sublenticular substantia innominata) becomes hyperactive during withdrawal. Key neuroadaptations include decreases in dopamine function in reward systems and recruitment of brain stress neurotransmitters, particularly corticotropin-releasing factor (CRF) and dynorphin [10] [11]. This creates a powerful negative reinforcement mechanism where drug seeking is driven not by pleasure but by relief from this negative emotional state.
Validating Animal Models and Protocols:
- Affective Measures During Withdrawal: Following cessation of chronic drug administration, researchers measure behavioral indices of negative affect. These include elevated plus maze tests for anxiety-like behavior, measures of anhedonia (e.g., reduced sucrose preference), and increased thresholds for brain stimulation reward, indicating a dysphoric state [11].
- Escalation of Drug Intake: In the Long Access (LgA) self-administration model, animals with extended drug access (6+ hours) progressively increase their intake, particularly at the beginning of sessions. This escalation is driven by a compensatory response to the developing negative emotional state and is a key marker of the transition to addiction [28] [11].
Table 2: Neuroadaptations in the Withdrawal/Negative Affect Stage
| Neurotransmitter/System | Direction of Change | Primary Brain Regions | Behavioral Consequence |
|---|---|---|---|
| Corticotropin-Releasing Factor (CRF) | Increase | Extended Amygdala | Anxiety, stress sensitivity |
| Dynorphin | Increase | Nucleus Accumbens, Extended Amygdala | Dysphoria, aversion |
| Norepinephrine | Increase | Bed Nucleus of Stria Terminalis | Anxiety, autonomic arousal |
| Dopamine | Decrease | Ventral Tegmental Area, Nucleus Accumbens | Anhedonia, reward deficit |
| Neuropeptide Y | Decrease | Extended Amygdala | Reduced stress buffering |
| Serotonin | Decrease | Raphe Nuclei | Depression, irritability |
Stage 3: Preoccupation/Anticipation (Craving)
The preoccupation/anticipation stage involves intense craving and deficits in executive function that lead to relapse. This stage engages a widely distributed network involving the prefrontal cortex (orbitofrontal, anterior cingulate, and medial prefrontal regions), basolateral amygdala, hippocampus, and insula [10] [11].
Core Neurocircuitry: The prefrontal cortex, particularly its projections to the basal ganglia and extended amygdala, is critically compromised. Glutamatergic projections from the prefrontal cortex to the nucleus accumbens become dysregulated, and disrupted executive function (impulse control, decision-making) results from hypoactivity in the prefrontal regions [10] [3]. The insula is implicated in interoceptive awareness of craving states.
Validating Animal Models and Protocols:
- Reinstatement Models: These are the primary models for studying relapse. After animals acquire self-administration and undergo extinction (where responding no longer produces drug), researchers test the ability of various triggers to reinstate drug-seeking behavior. These triggers include: drug-priming (small non-contingent drug dose), drug-associated cues (lights or tones previously paired with drug), and stressors (e.g., footshock) [28]. Each trigger engages partially distinct neural circuits.
- Incubation of Craving: This phenomenon demonstrates that cue-induced craving and reinstatement actually increase, rather than decrease, during prolonged abstinence. This time-dependent increase in craving is linked to neuroadaptations in glutamate transmission from prefrontal cortex to nucleus accumbens [11].
Modeling Individual Vulnerability
A critical advancement in addiction research has been the recognition that only a subset of individuals (approximately 20-40% of those who experiment with drugs) transition to addiction [29]. Animal models have been essential for identifying vulnerability factors and their neurobiological underpinnings.
Behavioral Endophenotypes
Several measurable behavioral traits, or endophenotypes, predict increased vulnerability to addiction in both humans and animals:
- Impulsivity: Defined as "a predisposition toward rapid, unplanned reactions to internal and external stimuli without regard for negative consequences" [10]. Animals with high baseline impulsivity on tasks like the 5-choice serial reaction time task or delay discounting tasks acquire drug self-administration more rapidly and show greater drug intake [29].
- Novelty/Sensation Seeking: The High-Responder/Low-Responder Model identifies rats with high spontaneous novelty-seeking behavior. These high-responders show greater locomotor response to novelty and subsequently acquire amphetamine and cocaine self-administration more rapidly, demonstrating an innate vulnerability [28] [29].
- Sign-Tracking vs. Goal-Tracking: In a Pavlovian conditioning paradigm, "sign-trackers" (who approach and interact with the cue predicting reward) show greater cue-induced reinstatement of drug seeking compared to "goal-trackers" (who approach the reward delivery location), modeling heightened cue reactivity and relapse vulnerability [28].
Genetic and Environmental Factors
Individual vulnerability arises from complex interactions between genetic predisposition, environmental factors, and epigenetic mechanisms.
- Genetic Factors: Heritability accounts for 40-60% of the risk for addiction. Selective breeding has produced lines with high preference for specific drugs (e.g., alcohol-preferring "P" rats or Marchigian Sardinian "msP" rats) [29]. These lines stably segregate traits like excessive drinking, high motivation for drug, and vulnerability to relapse, allowing researchers to study associated neuroadaptations, such as differences in dopamine D2 receptor and CRF system function [29].
- Environmental Factors: Stress, adverse childhood experiences, and drug availability are powerful vulnerability factors. In animals, chronic stress exposure increases drug self-administration and reinstatement. Conversely, environmental enrichment can be protective, reducing drug intake and relapse [29] [30].
- Adolescent Exposure: Adolescence is a critical "at-risk period." Animal studies show that drug use during this developmental window produces more pronounced and persistent neurobiological changes and increases adult addiction vulnerability [3].
Table 3: Key Behavioral Endophenotypes Predicting Addiction Vulnerability
| Endophenotype | Operational Definition in Animals | Common Assessment Tasks | Associated Neural Substrate |
|---|---|---|---|
| Impulsivity | Inability to withhold a response or preference for smaller immediate over larger delayed rewards | 5-Choice Serial Reaction Time, Delay Discounting | Prefrontal Cortex (hypoactivity), Orbitofrontal Cortex |
| Novelty/Sensation Seeking | High spontaneous exploratory activity in a novel environment | Novelty-Induced Locomotion | Ventral Tegmental Area (dopamine reactivity), Nucleus Accumbens |
| Sign-Tracking | Propensity to approach and interact with a cue predictive of reward | Pavlovian Conditioned Approach | Nucleus Accumbens Core (dopamine) |
| High Anxiety | Heightened behavioral inhibition in threatening environments | Elevated Plus Maze, Open Field Test | Extended Amygdala (CRF system), Bed Nucleus of Stria Terminalis |
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Research Reagents and Materials in Preclinical Addiction Research
| Reagent/Material | Primary Function/Application | Specific Examples & Notes |
|---|---|---|
| Operant Conditioning Chambers | Controlled environment for self-administration and reinstatement studies | Equipped with levers/response manipulanda, cue lights, tone generators, and drug infusion pumps. |
| Intravenous Catheters | Chronic venous access for drug self-administration | Surgical implantation into jugular or femoral vein; maintained with heparinized saline. |
| Microdialysis Systems | In vivo measurement of neurotransmitter release in specific brain regions | Used to measure extracellular dopamine, glutamate, etc., in nucleus accumbens or prefrontal cortex during drug taking or withdrawal. |
| Dopamine Receptor Ligands | Pharmacological manipulation of dopamine signaling | SCH 23390 (D1 antagonist); Raclopride (D2 antagonist); used to probe receptor contributions to drug seeking. |
| CRF Receptor Antagonists | Blockade of the brain stress system | Antalarmin (non-peptide CRF1 antagonist); used to reverse stress-induced reinstatement and negative affect of withdrawal. |
| Viral Vector Systems (AAV, Lentivirus) | Targeted manipulation of gene expression in specific circuits | Cre-lox system for cell-type specific manipulation; optogenetics/chemogenetics (DREADDs) for circuit control. |
| Small Interfering RNA (siRNA) | Transient knockdown of specific gene targets | Used to study roles of specific proteins (e.g., glutamate receptors) in addiction-related behaviors. |
Animal models have proven indispensable for validating the neurocircuitry constructs of the addiction cycle and elucidating the mechanisms of individual vulnerability. The heuristic framework of the three-stage cycle—with its distinct yet interacting neural substrates in the basal ganglia, extended amygdala, and prefrontal cortex—has provided a powerful organizational structure for research that translates across species [10] [11] [3].
Future research is increasingly adopting dimensional frameworks like the Research Domain Criteria (RDoC), which investigate disruptions in core functional domains (Positive Valence, Negative Valence, Cognitive Systems) across the addiction cycle [31]. This approach, combined with increasingly sophisticated animal models that incorporate individual differences, environmental complexity, and circuit-level manipulations, promises to advance our understanding of addiction. This progress will pave the way for personalized prevention and treatment strategies targeting specific neuroadaptations in vulnerable individuals, ultimately addressing the enormous personal and societal burdens of substance use disorders.
Human brain imaging techniques, particularly functional magnetic resonance imaging (fMRI) and positron emission tomography (PET), have revolutionized our understanding of the neural circuitry underlying addiction. These in vivo methods provide unparalleled insight into the neuroadaptations and circuit dysregulations that characterize addictive disorders, focusing on core structures such as the basal ganglia, extended amygdala, and prefrontal cortex (PFC). This whitepaper details the experimental protocols, analytical frameworks, and key findings from this field, serving as a technical guide for researchers and drug development professionals. The synthesized evidence confirms that addiction manifests as a relapsing cycle driven by a syndrome of impaired response inhibition and salience attribution (iRISA), where PFC dysfunction leads to excessive drug salience, reduced sensitivity to natural rewards, and impaired inhibitory control [16].
The Neural Circuitry of Addiction: A Tripartite Model
Addiction is not a localized brain disorder but a pathology of interconnected neural circuits. The iRISA model provides a framework for understanding how dysregulation in specific networks translates to the clinical symptoms of addiction [16]. The core components of this circuit are summarized in the table below.
Table 1: Core Neural Circuits and Their Dysregulation in Addiction
| Brain Circuit | Key Structures | Primary Function in Addiction | Manifestation of Dysregulation |
|---|---|---|---|
| Reward/Salience | Ventral Striatum (including Nucleus Accumbens), Ventral Tegmental Area | Reward processing, incentive salience | Increased sensitivity to drug cues; decreased sensitivity to natural rewards |
| Executive Control | Dorsolateral Prefrontal Cortex (DLPFC), Anterior Cingulate Cortex (ACC), Inferior Frontal Gyrus (IFG) | Self-control, decision-making, error detection | Impaired inhibitory control, impulsivity, compulsivity |
| Emotional/Motivational | Extended Amygdala, Orbitofrontal Cortex (OFC), Ventromedial PFC | Stress reactivity, emotional regulation, motivation | Enhanced stress and negative affect; increased motivation to procure drugs |
The dysfunction of the prefrontal cortex (PFC) is particularly critical. Neuroimaging studies have elucidated the roles of specific PFC subregions, demonstrating that addiction is not a unitary deficit but a collection of disruptions in distinct yet interacting neuropsychological processes [16].
Table 2: Prefrontal Cortex (PFC) Dysfunction in Addiction
| Neuropsychological Process | Disruption in Addiction | Implicated PFC Regions |
|---|---|---|
| Self-control & Behavioral Monitoring | Impulsivity, compulsivity, risk-taking, impaired self-monitoring | DLPFC, dACC, IFG, vlPFC |
| Emotion Regulation | Enhanced stress reactivity, inability to suppress negative affect | mOFC, vmPFC, subgenual ACC |
| Motivation | Enhanced motivation for drugs, decreased motivation for other goals | OFC, ACC, vmPFC, DLPFC |
| Decision Making | Choice of immediate reward, discounting of future consequences | lOFC, mOFC, vmPFC, DLPFC |
| Salience Attribution | Drugs have sensitized value; non-drug reinforcers are devalued | mOFC, vmPFC |
Key Methodologies and Experimental Protocols
Functional Magnetic Resonance Imaging (fMRI)
Protocol 1: Blood Oxygen Level-Dependent (BOLD) fMRI in Drug Challenge Studies
- Objective: To measure brain activity changes following acute drug administration and the modulation of these responses by drug expectation [16].
- Procedure:
- Participant Cohort: Overnight-abstinent, cocaine-addicted individuals.
- Task Design: Participants receive intravenous cocaine in both expected and unexpected delivery conditions in a controlled, counterbalanced design.
- Image Acquisition: Acquire T2*-weighted BOLD images on a 3T MRI scanner. Standard parameters: TR/TE = 2000/30 ms, voxel size = 3x3x3 mm³.
- Data Analysis: Compare BOLD signal changes between expected and unexpected drug delivery conditions. ROI analysis is performed on PFC subregions (e.g., lateral OFC, frontopolar cortex, ACC) and subcortical reward areas [16].
- Key Findings: PFC subregions (left lateral OFC, frontopolar cortex, ACC) show BOLD responses modulated by drug expectation, whereas subcortical regions respond primarily to the pharmacological effects of cocaine [16].
Protocol 2: Task-Based fMRI for Inhibitory Control
- Objective: To assess deficits in inhibitory control using a Go/No-Go or Stop-Signal Task.
- Procedure:
- Task: Participants respond quickly to frequent "Go" stimuli and withhold responses to infrequent "No-Go" stimuli.
- Image Acquisition: Standard BOLD fMRI acquisition during task performance.
- Data Analysis: Contrast BOLD activity during successful vs. failed No-Go trials. Focus on the inferior frontal gyrus (IFG) and dorsal ACC, key nodes of the inhibitory control network.
- Key Findings: Individuals with addiction consistently show reduced activation in the IFG and ACC during successful inhibition, correlating with behavioral measures of impulsivity [16].
Positron Emission Tomography (PET)
Protocol 3: FDG-PET to Measure Brain Glucose Metabolism
- Objective: To quantify changes in overall brain metabolic activity in response to a drug challenge.
- Procedure:
- Radiopharmaceutical: Intravenous administration of ⁸F-fluorodeoxyglucose (FDG).
- Drug Challenge: Administration of a stimulant drug like methylphenidate (MPH) to active cocaine users under expected and unexpected conditions.
- Image Acquisition: Perform PET scanning following the uptake period to measure regional cerebral glucose metabolism, a marker of neural activity.
- Data Analysis: Compare whole-brain and ROI-specific glucose metabolism between conditions.
- Key Findings: MPH administration increases whole-brain glucose metabolism. The lateral OFC showed a different response pattern (greater metabolism to unexpected MPH) compared to fMRI BOLD signals, highlighting the importance of considering temporal sensitivity in imaging modalities [16].
Protocol 4: Receptor-Specific PET for Dopaminergic Signaling
- Objective: To quantify dopamine receptor (D1, D2) availability and drug-induced dopamine release in the striatum and PFC.
- Procedure:
- Radiopharmaceuticals: Use ligands like [¹¹C]raclopride (for D2/D3 receptors) or [¹¹C]SCH23390 (for D1 receptors).
- Baseline Scan: Perform a PET scan to determine baseline receptor availability.
- Challenge Scan: A second scan is performed following administration of an addictive drug (e.g., amphetamine) or a psychostimulant (e.g., MPH).
- Data Analysis: Calculate the percentage change in binding potential (BPₙᴅ) between scans. A decrease in BPₙᴅ in the challenge scan indicates dopamine release and competition with the radioligand.
- Key Findings: Addiction is associated with reduced baseline D2 receptor availability in the striatum and blunted dopamine release in response to a stimulant challenge.
Visualizing Experimental Workflows and Signaling Pathways
The following diagrams, generated using Graphviz, outline core experimental workflows and conceptual frameworks in addiction neuroimaging research.
Graphviz Diagram 1: Standard fMRI experimental workflow for addiction studies.
Graphviz Diagram 2: The iRISA model illustrating how PFC dysfunction leads to core addiction behaviors [16].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagent Solutions for Imaging Addiction Neuroadaptations
| Reagent / Material | Function / Application | Example Use Case |
|---|---|---|
| ¹¹C-Raclopride | PET radioligand for dopamine D2/D3 receptors | Quantifying striatal D2 receptor availability and amphetamine-induced dopamine release. |
| ¹⁸F-FDG (Fluorodeoxyglucose) | PET radioligand for measuring glucose metabolism | Assessing regional brain metabolic activity under drug challenge or at rest. |
| Methylphenidate (MPH) | Psychostimulant challenge agent | Probing dopamine system responsivity in fMRI and PET studies [16]. |
| fMRI Go/No-Go Task | Paradigm to probe inhibitory control | Activating the inhibitory control network (IFG, ACC) to quantify deficits. |
| High-Resolution T1 MRI Sequence | Anatomical reference and volumetric analysis | Coregistering functional data and conducting voxel-based morphometry. |
| Statistical Parametric Mapping (SPM) or FSL | Software for fMRI/PET data analysis | Preprocessing, statistical modeling, and visualization of neuroimaging data. |
Human brain imaging has unequivocally demonstrated that addiction is a disorder of brain circuits, characterized by specific neuroadaptations in the reward, executive, and stress systems. The iRISA model, supported by extensive fMRI and PET findings, provides a powerful explanatory framework for these changes, centering on the collapse of PFC regulatory function. Future research will benefit from integrating multimodal imaging (e.g., combining fMRI and PET to link hemodynamic and neurochemical changes), employing computational modeling to better parse PFC subregion functions, and utilizing these biomarkers for developing and evaluating targeted therapeutic interventions in drug development [16]. The enduring neuroadaptive changes in the brain's circuitry underscore addiction as a chronic medical condition and highlight the imperative for long-term treatment strategies.
The neurobiological understanding of addiction has evolved to frame it as a chronic brain disorder characterized by compulsive drug seeking and use, despite negative consequences. This pathology is supported by distinct maladaptations within key neural circuits, primarily involving the basal ganglia, extended amygdala, and prefrontal cortex [4]. The transition from voluntary drug use to addiction is conceptualized as a cycle with three stages—binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation—each mediated by specific brain networks [4] [32]. The basal ganglia (including the nucleus accumbens, NAc) and ventral tegmental area (VTA) form a reward circuit central to the binge/intoxication stage. The extended amygdala (including the central amygdala, CeA) underlies the negative emotional state of withdrawal. The prefrontal cortex (PFC) is critical for the preoccupation/anticipation stage, governing executive function and behavioral control, which becomes compromised in addiction [4].
Genetic and epigenetic methodologies are revolutionizing the identification of biomarkers and molecular targets within these circuits. Addiction susceptibility involves a complex interplay of genetic predisposition and environmental factors, with heritable components accounting for an estimated 20-50% of the variability in risk [33]. Epigenetic mechanisms, which regulate gene expression without altering the DNA sequence, provide a molecular interface through which environmental exposures, including chronic drug use and stress, can induce persistent changes in neural gene expression, thereby influencing addiction vulnerability and trajectory [33] [34] [35]. This technical guide details the core methodologies employed to uncover these molecular underpinnings, providing a resource for researchers and drug development professionals.
Core Epigenetic Mechanisms in Addiction Neurobiology
Epigenetic regulation provides a dynamic and persistent mechanism for adapting the genome's output in response to environmental stimuli, including drugs of abuse. The three primary epigenetic mechanisms are DNA methylation, histone modifications, and non-coding RNA regulation.
DNA Methylation
DNA methylation involves the addition of a methyl group to the 5-carbon position of cytosine bases, primarily within cytosine-guanine (CpG) dinucleotides, catalyzed by DNA methyltransferases (DNMTs) [33] [35]. This modification is typically associated with transcriptional repression when it occurs in gene promoter regions. The process is dynamic, with ten-eleven translocation (TET) enzymes catalyzing the oxidation of 5-methylcytosine (5-mC) to 5-hydroxymethylcytosine (5-hmC) as part of active demethylation pathways [33] [35]. Chronic exposure to drugs of abuse, including cocaine, alcohol, and opioids, alters the expression and activity of DNMTs and TETs in key brain regions like the NAc, leading to lasting changes in the methylation state of genes critical for synaptic plasticity and behavior [34] [35]. For instance, a genome-wide association study identified that under-expression of the CHRNA2 gene in the cerebellum, potentially regulated by methylation, is associated with cannabis use disorder [4].
Histone Modification
Histones are core proteins around which DNA is wrapped to form chromatin. They can undergo a wide array of post-translational modifications—including acetylation, methylation, phosphorylation, and ubiquitylation—on their N-terminal tails [33] [34]. These modifications alter chromatin structure and accessibility, thereby influencing gene expression.
- Histone Acetylation: Generally associated with an open, transcriptionally active chromatin state, this is regulated by the opposing actions of histone acetyltransferases (HATs) and histone deacetylases (HDACs). Drugs of abuse can induce targeted acetylation, such as at histone H3 lysine 9 and 14 (H3K9K14ac2), at specific gene promoters [33] [34].
- Histone Methylation: The effect on transcription depends on the specific lysine residue methylated and the degree of methylation (mono-, di-, or tri-methylation). For example, trimethylation of histone H3 lysine 4 (H3K4me3) is linked to promoter activation, whereas trimethylation of H3 lysine 27 (H3K27me3) is associated with transcriptional repression [33].
Non-Coding RNA Regulation
Non-coding RNAs (ncRNAs), which include microRNAs (miRNAs), small interfering RNAs (siRNAs), and long non-coding RNAs (lncRNAs), regulate gene expression post-transcriptionally or by guiding chromatin-modifying complexes to specific genomic loci [33]. For example, siRNA can promote DNA and histone methylation to silence gene expression, while lncRNAs can form complexes with proteins to influence chromatin architecture and gene silencing [33].
Table 1: Core Epigenetic Mechanisms and Their Roles in Addiction
| Mechanism | Key Enzymes/Effectors | General Functional Outcome | Example in Addiction |
|---|---|---|---|
| DNA Methylation | DNMTs, TETs, MeCP2 | Transcriptional repression (typically) | Altered DNMT3A expression in NAc after cocaine exposure [35] |
| Histone Acetylation | HATs, HDACs | Transcriptional activation (HATs) or silencing (HDACs) | Acetylation changes at plasticity gene promoters in reward circuits [34] |
| Histone Methylation | HMTs, HDMs | Activation or repression (context-dependent) | H3K4me3 changes associated with sustained transcriptional adaptations [33] |
| Non-Coding RNAs | miRNAs, lncRNAs | mRNA degradation, translational repression, chromatin remodeling | miRNA regulation of genes involved in synaptic plasticity [33] |
Methodologies for Mapping and Modifying Brain Circuits
Understanding the molecular targets within addiction circuits requires sophisticated tools to map, observe, and manipulate specific neural populations with high precision.
Neural Circuit Mapping and Monitoring
Viral Tracing techniques are fundamental for delineating the complex connectivity of addiction-related circuits. Adeno-associated virus (AAV)-based tracers and monosynaptic rabies virus systems enable the precise mapping of neural inputs and outputs at single-synapse resolution [36]. For example, these methods have revealed distinct input patterns to different neuron populations in the retrosplenial cortex (RSC), which projects to areas like the thalamus and secondary motor cortex [36].
Nanostructured photonic probes represent a recent advancement, allowing for ultra-high-resolution observation and control of brain activity at cellular and subcellular levels with remarkable spatial (~100 nm) and temporal (~ms) accuracy, surpassing traditional electrodes [36].
Precision Neuromodulation
These techniques allow for causal inferences about the function of specific circuits and cell types.
- Optogenetics: This method uses light-sensitive ion channels (e.g., channelrhodopsin) to exert millisecond-scale control over the activity of genetically defined neuronal populations [36]. For instance, optogenetic silencing of M2-projecting RSC neurons was shown to impair object-location memory, highlighting the functional specificity of parallel pathways [36].
- Chemogenetics: Chemogenetics employs engineered receptors (e.g., DREADDs - Designer Receptors Exclusively Activated by Designer Drugs) that are activated by inert synthetic ligands to modify neural activity over a longer timescale (hours) [36] [37]. A key application is demonstrated in the central amygdala (CeA), where chemogenetic inhibition of distinct neuronal populations (PKC-δ, CRF, or SST) differentially reduced fentanyl self-administration or withdrawal symptoms in mice [37].
- Photopharmacology: This innovative approach uses small drug molecules tethered to their target receptor (e.g., mGluR2) that can be selectively "switched on" in specific brain circuits using light. A Weill Cornell Medicine study used this method to dissect anxiety circuits, finding that activating mGluR2 in an insula-to-amygdala pathway reduced anxiety without the cognitive impairments caused by activating the same receptor in a prefrontal-to-amygdala pathway [38].
The following diagram illustrates a typical experimental workflow integrating these methodologies to identify and validate circuit-specific molecular targets.
Figure 1: Experimental Workflow for Target Identification
Methodologies for Genetic and Epigenetic Analysis
A suite of high-throughput and targeted molecular techniques is used to identify genetic and epigenetic biomarkers within the defined neural circuits.
Genomic and Epigenomic Profiling
Genome-Wide Association Studies (GWAS) scan the genome for genetic variants (single nucleotide polymorphisms, SNPs) that occur more frequently in individuals with a substance use disorder compared to controls. This approach has identified loci associated with addiction risk, such as a region on chromosome 8 linked to cannabis use disorder [4].
Epigenome-Wide Analysis leverages several key technologies:
- Bisulfite Sequencing: Treatment of DNA with bisulfite converts unmethylated cytosines to uracils, allowing for base-resolution mapping of methylated cytosines. Methods include Whole Genome Bisulfite Sequencing (WGBS) for a comprehensive view and Reduced Representation Bisulfite Sequencing (RRBS) for a more cost-effective analysis of CpG-rich regions [34].
- Chromatin Immunoprecipitation Sequencing (ChIP-seq): This technique identifies the genomic locations of specific histone modifications (e.g., H3K27ac) or transcription factors by using antibodies to pull down cross-linked protein-DNA complexes, which are then sequenced [34].
- Multi-Omics Integration: Advanced studies now combine datasets from genomics, transcriptomics, epigenomics, and connectomics (brain connectivity mapping) to build a more holistic model of addiction pathophysiology and identify convergent biomarkers [39].
Epigenome Editing
To move from correlation to causation, epigenome editing tools allow for the direct manipulation of epigenetic marks at specific genomic loci.
- CRISPR-dCas9 Systems: A catalytically "dead" Cas9 (dCas9) is fused to epigenetic "writer" or "eraser" enzymes (e.g., DNMT3A for methylation, TET1 for demethylation, p300 for acetylation). Guided by a single-guide RNA (sgRNA), the complex can be targeted to a specific gene to add or remove an epigenetic mark and directly test its effect on gene expression and behavior [34].
- Other Editing Platforms: Transcription activator-like effector nucleases (TALENs) and zinc-finger nucleases (ZFNs) can also be fused to epigenetic modifiers for targeted epigenome editing, though they are often less easily programmable than the CRISPR-dCas9 system [34].
Table 2: Key Research Reagents and Their Applications
| Research Reagent / Tool | Category | Primary Function in Research |
|---|---|---|
| Adeno-associated Virus (AAV) Vectors | Viral Tool | Gene delivery and retrograde tracing of neural circuits [36] |
| Monosynaptic Rabies Virus | Viral Tool | High-resolution mapping of direct synaptic inputs to a target cell population [36] |
| DREADDs (Chemogenetics) | Neuromodulator | Remote, long-timescale control of neuronal activity via systemic ligand injection [37] |
| Channelrhodopsin (Optogenetics) | Neuromodulator | Millisecond-precise activation of neurons with light [36] |
| CRISPR-dCas9 Epigenetic Systems | Epigenome Editor | Targeted addition or removal of DNA methylation/histone modifications at specific genes [34] |
| Bisulfite Conversion Reagents | Epigenetic Analysis | Chemical treatment to distinguish methylated from unmethylated cytosine for sequencing [33] |
The following diagram summarizes the key signaling pathways and molecular interactions in addiction circuits that are investigated using these methodologies.
Figure 2: Molecular Signaling in Addiction Pathophysiology
Experimental Protocols for Key Experiments
Protocol: Circuit-Specific Analysis of Epigenetic States
This protocol details the process for isolating a specific neural circuit and analyzing its cell-type-specific epigenome.
- Circuit Targeting: Inject a retrograde AAV (e.g., AAV2-retro) expressing a fluorescent reporter into a target region (e.g., Basolateral Amygdala, BLA) to label neurons projecting to that site [36] [38].
- Tissue Collection and Cell Sorting: After a suitable expression period, euthanize the animal and microdissect the source region (e.g., Insular Cortex). Prepare a single-cell suspension and use Fluorescence-Activated Cell Sorting (FACS) to isolate fluorescently-labeled (projection-specific) neurons from unlabeled neurons.
- Nuclei Isolation and Sorting (Alternative): For assays requiring intact chromatin, homogenize the brain region and isolate nuclei. Use FACS to sort nuclei based on the fluorescent reporter.
- Downstream Molecular Analysis:
- DNA Methylation: Extract genomic DNA from sorted cells/nuclei and perform bisulfite conversion followed by sequencing (WGBS or RRBS) [34] [35].
- Histone Modifications: Use ChIP-seq on fixed chromatin from sorted cells/nuclei with antibodies against specific histone marks (e.g., H3K27ac) [34].
- Transcriptomics: Extract RNA from sorted cells for RNA-sequencing to correlate epigenetic changes with gene expression [39].
Protocol: Validating a Candidate Epigenetic Target via In Vivo Editing
This protocol tests the causal role of a specific epigenetic mark at a candidate gene in regulating addictive behavior.
- Vector Design: Create an AAV vector expressing:
- dCas9-DNMT3A (to induce methylation) or dCas9-TET1 (to induce demethylation).
- A sgRNA designed to target the regulatory region (e.g., promoter) of the candidate gene (e.g., BDNF in the NAc).
- A cell-type-specific promoter (e.g., CaMKIIa for excitatory neurons) to restrict expression.
- Stereotaxic Surgery: Inject the AAV vectors bilaterally into the target brain region (e.g., NAc) of adult mice.
- Behavioral Validation: After allowing for expression, subject mice to a relevant behavioral paradigm (e.g., conditioned place preference for a drug, or operant self-administration) to assess the effect of the epigenetic manipulation on addiction-related behaviors [34].
- Ex Vivo Verification: After behavior, verify:
- Targeting Efficiency: Quantify methylation changes at the candidate gene's promoter using bisulfite pyrosequencing on microdissected tissue.
- Functional Impact: Measure changes in candidate gene mRNA and protein levels using qRT-PCR and immunohistochemistry/Western blot.
Discussion and Future Directions
The integration of advanced circuit mapping with genetic and epigenetic methodologies is driving a shift toward precision neuromedicine in addiction treatment [36]. The convergence of data from omics technologies—genomics, epigenomics, transcriptomics, and connectomics—is facilitating the development of a dimensional understanding of addiction, as seen in frameworks like the Addictions Neuroclinical Assessment (ANA) [40] [39]. This approach moves beyond mere substance exposure to define addiction by core neurobiological domains such as incentive salience, negative emotionality, and executive function, each with its own biomarkers and potential molecular targets [40].
Future research must address several key challenges. The cell-type and temporal specificity of epigenetic changes require more refined tools to dissect the contributions of specific neuronal and non-neuronal populations across different stages of the addiction cycle [35]. Furthermore, the translatability of epigenetic biomarkers from rodent models to humans needs rigorous validation. The emergence of epidrugs—medications designed to target epigenetic machinery—and the refinement of epigenome editing strategies present promising avenues for novel therapeutics [34]. However, delivering these tools with the necessary specificity to defined neural circuits in humans remains a significant hurdle. By continuing to refine these methodologies and integrate multidimensional data, the field is poised to identify clinically actionable biomarkers and develop more effective, personalized interventions for substance use disorders.
Understanding the neural circuitry of addiction requires tools that can not only monitor but also precisely manipulate specific brain pathways to establish causal relationships. Electrophysiological and optogenetic techniques have emerged as cornerstone methodologies in modern neuroscience for probing the function of neural circuits involving the basal ganglia, extended amygdala, and prefrontal cortex—key regions in addiction pathology [7]. These approaches enable researchers to move beyond correlation to establish causality in circuit function, particularly in mapping the neural substrates underlying compulsive drug-seeking, craving, and relapse [41] [42].
Electrophysiology provides direct measurement of neuronal activity with millisecond temporal resolution, capturing the fundamental electrical signals that constitute neural communication [41]. Optogenetics complements this by enabling precise temporal control of genetically defined neuronal populations using light, allowing researchers to test causal hypotheses about specific circuit elements [42]. When integrated, these techniques form a powerful framework for deconstructing the complex neural dynamics of addiction-related behaviors across the binge-intoxication, withdrawal-negative affect, and preoccupation-anticipation stages that characterize substance use disorders [43].
Fundamental Principles of Circuit Probing Techniques
Electrophysiological Approaches
Electrophysiological techniques measure the electrical activity of neurons, providing direct insight into neural signaling dynamics. Local field potentials (LFPs) represent the summed synaptic activity of neuronal populations and are particularly valuable for identifying oscillatory patterns that correlate with behavioral states [41]. In addiction research, LFPs recorded from the nucleus accumbens (NAc), a central hub of the reward circuit, have revealed potential biomarkers for craving states [41]. For example, specific oscillatory patterns in the NAc have been correlated with cue-induced craving, offering potential readouts for therapeutic interventions.
Single-unit recording extends this approach by capturing the action potential activity of individual neurons, allowing researchers to characterize how specific cells encode information during addiction-related behaviors. This technique has revealed that certain neuronal ensembles in the prefrontal cortex and basal ganglia fire selectively during drug anticipation, consumption, or withdrawal [43]. The exceptional temporal resolution of electrophysiological methods (on the millisecond timescale) makes them ideal for capturing the rapid sequence of neural events that underlie decision-making and habit formation in addiction.
Optogenetic Approaches
Optogenetics enables unprecedented causal interrogation of neural circuits by combining genetic targeting with precise light activation. The fundamental principle involves expressing light-sensitive microbial opsins (e.g., channelrhodopsin for excitation, halorhodopsin for inhibition) in specific neuronal populations, allowing researchers to control their activity with millisecond precision [42]. This technique has revolutionized addiction research by enabling investigators to test whether specific circuit elements are sufficient or necessary for particular behaviors.
The experimental workflow typically begins with identifying a genetically defined neuronal population of interest (e.g., dopamine neurons in the ventral tegmental area or specific prefrontal cortex projection neurons). Researchers then introduce opsin genes using viral vector delivery systems, often with cell-type-specific promoters for targeting [42]. After allowing time for opsin expression, implanted optical fibers deliver light to the target region while behavioral measures are recorded. This approach has been used to map addiction-relevant circuits, demonstrating that activation of the pathway from the nucleus accumbens to the hypothalamus can induce compulsive behaviors, while modulation of medial prefrontal cortex projections to the NAc can suppress drug-seeking [44] [43].
Technical Methodologies and Experimental Protocols
Integrating Optogenetics with Electrophysiology
Combining optogenetic manipulation with simultaneous electrophysiological recording creates a powerful closed-loop system for probing neural circuits. This integrated approach allows researchers to not only control neuronal activity but also monitor the immediate consequences of that manipulation across the network. The experimental setup typically involves:
Viral-mediated opsin delivery: Recombinant adeno-associated viruses (rAAV) encoding light-sensitive opsins are stereotaxically injected into target brain regions (e.g., medial prefrontal cortex, nucleus accumbens, or ventral tegmental area) using cell-type-specific promoters [42].
Optrode implantation: Custom-built optrodes (combined optical fibers and electrodes) are implanted above or within the target region to allow simultaneous light delivery and electrical recording.
Real-time triggering: Software systems like Bonsai or WaveSurfer are used to detect specific neural signatures (e.g., oscillatory patterns) and trigger optogenetic stimulation with minimal latency, enabling closed-loop circuit control.
This methodology has been applied to study the ventral tegmental area dopamine projection to the amygdala, revealing its necessity for cocaine withdrawal-induced anxiety and reinstatement of drug-seeking behavior [45]. By recording neural responses while optically manipulating this pathway, researchers established its causal role in specific addiction phenotypes.
Advanced Circuit Mapping Techniques
Comprehensive circuit analysis requires detailed mapping of connectivity between brain regions. The monosynaptic rabies virus tracing system has become a gold standard for delineating direct inputs to specific neuronal populations [45]. The protocol involves:
Helper virus injection: An AAV expressing TVA receptor and rabies glycoprotein is injected into the target region.
Modified rabies virus injection: After 3-4 weeks, EnvA-pseudotyped, G-deleted rabies virus encoding a fluorescent reporter is injected at the same site.
Input neuron identification: The rabies virus infects only TVA-expressing starter cells, then spreads retrogradely to directly presynaptic partners, which can be quantified and characterized.
This approach revealed that bed nucleus of the stria terminalis GABAergic neurons (BNST(_{GABA})) show elevated activity after cocaine exposure and project to midbrain dopamine cells, forming a circuit that controls cocaine-induced anxiety but not reward sensitization [45].
Table 1: Quantitative Neural Oscillations Associated with Addiction States
| Brain Region | Oscillation Type | Behavioral Correlation | Measurement Technique |
|---|---|---|---|
| Nucleus Accumbens | Gamma-band (30-100 Hz) | Cue-induced craving intensity | Local Field Potential Recording [41] |
| Prefrontal Cortex | Theta-band (4-12 Hz) | Compulsive drug-seeking | Depth Electrode Recording [43] |
| Basal Ganglia | Beta-band (13-30 Hz) | Habit formation | Single-unit & LFP Recording [46] |
Circuit-Specific Insights in Addiction Neuroscience
Prefrontal Cortex Circuits in Stage-Specific Addiction
The medial prefrontal cortex (mPFC) serves as a critical hub in addiction pathology, with distinct subregions contributing to different stages of the addiction cycle [43]. The prelimbic cortex (PL) → nucleus accumbens core pathway promotes cocaine-seeking behavior, while the infralimbic cortex (IL) → nucleus accumbens shell pathway suppresses relapse [43]. These opposing functions highlight the complex top-down control exerted by prefrontal regions over subcortical reward areas.
Optogenetic studies have revealed that drug experience induces lasting synaptic plasticity in these circuits, including alterations in the balance between excitatory and inhibitory transmission and changes in dendritic spine morphology [43]. These adaptations are thought to underlie the persistent vulnerability to relapse that characterizes addiction. Furthermore, different neurotransmitter systems in the mPFC (dopaminergic, glutamatergic, and GABAergic) show stage-specific imbalances that contribute to pathological addiction states [43].
Extended Amygdala-Midbrain Circuits in Negative Affect
The extended amygdala, particularly the bed nucleus of the stria terminalis (BNST), forms critical circuits with midbrain dopamine regions that mediate the negative emotional states associated with drug withdrawal [45]. Using a combination of rabies virus circuit mapping, fiber photometry, and chemogenetic approaches, researchers have demonstrated that the BNST(_{GABA})→VTADA→amygdala pathway is selectively necessary for cocaine-induced anxiety but not for cocaine reward or sensitization [45].
This circuit shows persistent elevation in spontaneous and task-related activity after even a single cocaine exposure, suggesting early neuroadaptations that may contribute to the development of addiction. Silencing this projection prevents the development of anxiety during protracted withdrawal, highlighting its potential as a therapeutic target [45]. Furthermore, VTADA→amygdala cells are strongly activated after a challenge exposure to cocaine and are necessary and sufficient for reinstatement of cocaine place preference, directly linking this circuit to relapse vulnerability.
Basal Ganglia Loops in Habit Formation
The basal ganglia plays a central role in the progression from voluntary drug use to compulsive drug-seeking habits [46] [7]. Animal models of Alcohol Use Disorder (AUD) have revealed dysfunction in both associative and sensorimotor basal ganglia loops that impair action selection and promote compulsive alcohol seeking despite negative consequences [46].
The basal ganglia forms a key node of the brain's "reward circuit," which is over-activated by drugs of abuse, producing intense euphoria [7]. With repeated exposure, this circuit adapts to the presence of the drug, diminishing its sensitivity and making it hard to feel pleasure from natural rewards. Simultaneously, the shift from prefrontal cortical control to striatal control over drug-seeking behavior represents a transition from goal-directed to habitual responding [7]. Optogenetic studies have identified specific striosomal pathways from the nucleus accumbens to the hypothalamus that drive stereotyped behaviors through an aversive hypothalamic-habenula circuit, potentially explaining the compulsive nature of drug-seeking [44].
Table 2: Optogenetic Approaches for Circuit Manipulation in Addiction Models
| Target Circuit | Optogenetic Intervention | Behavioral Effect | Key Findings |
|---|---|---|---|
| PL→NAc core [43] | Channelrhodopsin stimulation | Increased drug-seeking | Sufficient to reinstate extinguished drug-seeking behavior |
| IL→NAc shell [43] | Channelrhodopsin stimulation | Suppressed relapse | Promotes extinction of drug-seeking responses |
| NAc→Hypothalamus [44] | Channelrhodopsin stimulation | Induced compulsive behaviors | Drives repetitive actions even without reward |
| BNST(_{GABA})→VTADA [45] | Halorhodopsin inhibition | Reduced anxiety | Prevents development of withdrawal-induced anxiety |
Research Reagent Solutions
Table 3: Essential Research Reagents for Circuit Neuroscience in Addiction
| Reagent / Tool | Function | Example Application |
|---|---|---|
| Adeno-associated Virus (AAV) Vectors [42] | Gene delivery for opsin expression | Targeted expression of channelrhodopsin in specific neuronal populations |
| Monosynaptic Rabies Virus [45] | Retrograde tracing of direct inputs | Mapping connectivity to specific cell types in addiction circuits |
| Channelrhodopsin-2 (ChR2) [42] | Light-activated cation channel for neuronal excitation | Testing sufficiency of specific circuit activation in drug-seeking |
| Halorhodopsin (NpHR) [42] | Light-activated chloride pump for neuronal inhibition | Testing necessity of specific circuits in addiction behaviors |
| Archaerhodopsin (ArchT) [42] | Light-activated proton pump for neuronal silencing | Prolonged inhibition of specific pathways during behavioral assays |
| DREADDs (Designer Receptors Exclusively Activated by Designer Drugs) [36] | Chemogenetic control of neuronal activity | Modulating circuit activity over longer timescales (hours) |
| Cre-recombinase Mouse Lines [43] | Cell-type-specific targeting | Selective access to genetically defined neuronal populations |
| Fiber Photometry Systems [41] | Recording neural activity in behaving animals | Monitoring calcium or neurotransmitter dynamics during behavior |
| Tetracysteine Display of Optogenetic Elements (Tetro-DOpE) [36] | Real-time monitoring and modification of neuronal populations | Simultaneous manipulation and observation of circuit function |
Signaling Pathways and Workflows
Neural Circuit Mapping and Manipulation Workflow
Diagram 1: Experimental workflow for circuit manipulation studies
Extended Amygdala-Midbrain Circuit in Cocaine Withdrawal
Diagram 2: Circuit mechanism of cocaine withdrawal behaviors
The integration of electrophysiological and optogenetic techniques has fundamentally advanced our understanding of the neural circuitry underlying addiction, providing unprecedented spatial and temporal precision for probing circuit-specific function and establishing causality. These approaches have revealed stage-specific roles for prefrontal-basal ganglia-amygdala circuits in different aspects of addiction, from initial drug reward to compulsive drug-seeking and relapse [43] [45]. The continued refinement of these tools, including the development of more specific opsins, improved neural recording technologies, and sophisticated computational approaches for data analysis, promises to further elucidate the complex neural dynamics of addiction [47] [36].
Future directions in the field include the development of closed-loop systems that can detect pathological neural states and deliver precisely timed interventions to normalize circuit function [41]. Additionally, combining circuit-specific manipulations with transcriptomic and proteomic analyses will help bridge the gap between neural activity and molecular adaptations in addiction [42]. These advances will not only deepen our fundamental understanding of addiction neuroscience but also contribute to the development of targeted neuromodulation therapies for substance use disorders, such as deep brain stimulation informed by craving-related biomarkers [41] and transcranial magnetic stimulation tailored to specific circuit dysfunctions [36].
The development of effective treatments for substance use disorders necessitates a deep understanding of the underlying neurocircuitry. This whitepaper outlines a comprehensive framework for integrating cross-species data to build a consolidated model of addiction neurocircuitry. By synthesizing findings from rodent models, human brain imaging, and advanced in vitro human neuronal systems, we present a unified view of dysregulation across the basal ganglia, extended amygdala, and prefrontal cortex. The document provides detailed experimental protocols for key methodologies, visualizes core signaling pathways, and catalogues essential research tools to empower researchers and drug development professionals in validating and exploiting this integrated model for therapeutic discovery.
Addiction is a chronic brain disorder characterized by a compulsive cycle of bingeing, withdrawal, and relapse, driven by progressive dysregulation of key brain circuits [2]. Research spanning decades has established that this cycle is orchestrated by three primary brain regions: the basal ganglia, central to reward and habit formation; the extended amygdala, central to stress and negative affect; and the prefrontal cortex, central to executive control and relapse [3] [7] [2]. The development of a predictive, consolidated neurocircuitry model requires the integration of diverse, cross-species data. Animal studies, particularly in rodents, allow for precise causal manipulations under controlled conditions, while human neuroimaging and post-mortem studies provide essential validation and context [3]. The challenge lies in reconciling these data sources to create a model that is not only anatomically precise but also functionally predictive for the human condition. This guide details the methodologies and frameworks for achieving this integration, with a specific focus on the neural circuitry of addiction.
Core Neurocircuitry Framework: A Three-Stage Cycle
The addiction cycle is heuristically framed as a three-stage process—binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation—each mediated by distinct yet interacting neurocircuits [2].
Table 1: Key Brain Regions in the Addiction Cycle
| Addiction Stage | Primary Brain Circuit | Key Neurotransmitters | Behavioral Output |
|---|---|---|---|
| Binge/Intoxication | Basal Ganglia (reward circuit) | Dopamine, Opioid Peptides | Reward, Pleasure, Habit Formation |
| Withdrawal/Negative Affect | Extended Amygdala | CRF, Dynorphin, Norepinephrine | Anxiety, Irritability, Dysphoria |
| Preoccupation/Anticipation | Prefrontal Cortex (to Basal Ganglia & Extended Amygdala) | Glutamate, Dopamine | Craving, Relapse, Loss of Executive Control |
Stage 1: Binge/Intoxication
The basal ganglia, particularly the nucleus accumbens, are critical for processing reward. All addictive substances directly or indirectly produce large, aberrant surges of dopamine in this circuit, powerfully reinforcing drug-taking behavior and facilitating the transition from voluntary use to compulsive habit [7] [2]. This stage is characterized by the assignment of excessive incentive salience to drug-associated cues.
Stage 2: Withdrawal/Negative Affect
The extended amygdala is a key structure in the brain's stress system. As addiction progresses, counter-adaptations occur, including a decrease in the function of the reward system and a recruitment of brain stress neurotransmitters like corticotropin-releasing factor (CRF) and dynorphin [2]. This creates a negative emotional state—including anxiety, irritability, and malaise—that defines the withdrawal stage and motivates drug use to achieve temporary relief (negative reinforcement).
Stage 3: Preoccupation/Anticipation
The prefrontal cortex is essential for executive function: decision-making, self-regulation, and impulse control. In addiction, this region becomes profoundly dysregulated, leading to compulsive drug-seeking despite negative consequences [7]. This stage involves disrupted glutamate projections from the prefrontal cortex and insula back to the basal ganglia and extended amygdala, mediating craving and relapse [2].
The following diagram illustrates the interplay between these brain regions and the corresponding behavioral stages of addiction.
Quantitative Cross-Species Data Integration
Building a consolidated model requires the systematic quantification and alignment of data across species. The table below summarizes key quantitative findings from rodent and human studies that inform the model's parameters, focusing on molecular and genetic changes.
Table 2: Cross-Species Quantitative Data for Model Parameters
| Parameter Category | Specific Target / Finding | Rodent Model Data | Human / Clinical Correlation | Integrated Model Inference |
|---|---|---|---|---|
| Gene Expression | Vdac1, Add2, Syt2, Syt4 | Significantly correlated with pain phenotypes across 8 brain regions in BXD mice [48] | PheWAS linked 53 module genes to pain-related GWAS traits; NCAM1, VAMP2 top candidates [48] | Prioritized regulator genes for synaptic dysfunction in addiction |
| Neuroplasticity | cAMP & PKA Signaling | Chronic drug exposure upregulates cAMP formation & PKA activity in reward circuits [2] | Altered prefrontal glutamate function linked to craving & relapse [2] | Key transduction pathway for long-term synaptic changes |
| Circuit Connectivity | Local Clustering & Hub Neurons | Model of rat somatosensory cortex shows local clusters tied by L5 hub neurons [49] | fMRI reveals disrupted functional connectivity between PFC, basal ganglia, and amygdala [2] | Hub-based architecture for information flow; targeted for dysregulation |
| Inhibitory Circuitry | PV+, Sst+, VIP+ Interneurons | Anatomy predicts Sst+ targeting; PV+/VIP+ require additional specificity [49] | Altered cortical inhibition observed in imaging & post-mortem studies of addiction | Model incorporates cell-type-specific connection rules |
Experimental Protocols for Cross-Species Integration
Protocol: Cross-Species Transcriptomic Analysis for Candidate Gene Discovery
This protocol identifies evolutionarily conserved pain and addiction-related gene regulators through integrated bioinformatics.
Primary Workflow:
- Rodent RNA-Seq: Generate transcriptomic data from relevant brain regions (e.g., dorsal horn of lumbar spinal cord) in a neuropathic pain model (e.g., Spared Nerve Injury - SNI) versus control mice [48].
- Methods: Total RNA extraction with Trizol, mRNA enrichment via Oligo(dT) beads, library prep with NEBNext Ultra RNA Library Prep Kit, sequencing on Illumina Novaseq6000 (~50 million 150bp paired-end reads per sample).
- Human DRG Co-Expression Analysis: Perform Weighted Gene Co-expression Network Analysis (WGCNA) on microarray data from >200 human dorsal root ganglion samples to identify co-expressed gene modules [48].
- Module Integration: Select a human gene module significantly over-enriched with a "pain reference set" of known genes.
- Cross-Species Validation:
- Perform Protein-Protein Interaction (PPI) network analysis on the human "pain module" genes.
- Overlap the network genes with differentially expressed genes from the rodent RNA-seq.
- Conduct PheWAS (Phenome-Wide Association Study) to link candidate genes to pain/addiction-related GWAS traits in humans.
- In BXD GRP mice, correlate candidate gene expression with pain phenotypes across multiple brain regions.
Final Output: A prioritized list of cross-species validated candidate genes (e.g., Vdac1, Add2, Syt2, NCAM1, VAMP2) for functional validation [48].
Protocol: Functional Validation in Compartmentalized Human Neurocircuits
This protocol tests the functional role of candidate genes in a simplified, human-specific neurocircuitry model.
Primary Workflow:
- Neuronal Generation: Generate excitatory (glutamatergic), inhibitory (GABAergic), and dopaminergic induced neurons (iNs) from human induced pluripotent stem cells (iPSCs) [50].
- Microfluidic Culture: Seed different neuronal subtypes into a custom 5-compartment polydimethylsiloxane (PDMS) microfluidic device. The device features a large central chamber surrounded by four outer chambers, connected by microchannels (3μm high, 10μm wide) that permit axon penetration but restrict cell bodies, creating a compartmentalized system [50].
- Genetic Manipulation: Use lentiviral vectors to introduce optogenetic actuators (e.g., Channelrhodopsin) or to knock down/overexpress candidate genes in specific neuronal subtypes.
- Functional Assay:
- Morphological: Confirm synaptic contacts between chambers via immunostaining for synapsin.
- Physiological: Use patch-clamp electrophysiology in the central chamber to record excitatory postsynaptic currents (EPSCs) evoked by optogenetic stimulation of neurons in an outer chamber [50].
- Behavioral Correlation: Relate circuit dysfunction to addiction-relevant phenotypes (e.g., altered network synchrony, response to drug application).
Final Output: Causal evidence for a candidate gene's role in regulating human synaptic function and microcircuit activity relevant to addiction pathways.
The following diagram maps the key stages of this cross-species data integration workflow.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents for Neurocircuitry Studies
| Reagent / Tool | Function / Application | Example Use in Addiction Research |
|---|---|---|
| BXD Genetic Reference Population | A panel of ~150 inbred mouse strains for precise mapping of complex traits linking genetics to omics and phenome data [48]. | Identify genetic loci and candidate genes underlying variability in addiction-related behaviors and neurobiology [48]. |
| Optogenetic Tools (e.g., Channelrhodopsin, Halorhodopsin) | Genetically targeted, light-activated ion channels for millisecond-precise excitation or inhibition of specific neuronal populations [51]. | Causally test the role of specific projections (e.g., PFC to nucleus accumbens) in drug-seeking and relapse behavior. |
| Chemogenetic Tools (DREADDs) | Designer Receptors Exclusively Activated by Designer Drugs; allow modulatory, G-protein coupled receptor-based control of neuronal activity via systemic drug injection [51]. | Manipulate activity of specific cell types in addiction circuits over longer timeframes (hours) to model chronic adaptations. |
| Compartmentalized Microfluidic Devices | PDMS culture devices with microchannels that enable spatial separation of different neuronal subtypes while allowing axonal projection and synaptic connection formation [50]. | Model afferent/efferent connections between different brain regions (e.g., VTA DA neurons to NAc) using human iNs. |
| Lentiviral Vectors | Genetically modified viruses for stable gene delivery (overexpression, knockdown, reporter expression) to specific brain regions or cell types [50] [51]. | Deliver optogenetic constructs, DREADDs, or shRNAs against candidate genes into defined neurocircuits in rodent models or human iNs. |
The path to revolutionizing addiction therapeutics lies in moving beyond a siloed view of data from individual species or levels of analysis. The framework presented here provides a robust, actionable guide for building a consolidated neurocircuitry model of addiction. By systematically integrating quantitative data from rodent models, human genetics, and advanced in vitro human systems, and by employing a standardized toolkit of modern neuroscience techniques, researchers can achieve an unprecedented, high-fidelity understanding of the dysregulated neural circuits that drive addiction. This consolidated model will serve as a powerful platform for identifying and validating novel, circuit-based therapeutic targets, ultimately leading to more effective treatments for substance use disorders.
Challenges and Neuroadaptations: Addressing Tolerance, Withdrawal, and Relapse
Drug addiction is recognized as a chronically relapsing disorder characterized by a compulsive pattern of drug seeking and taking, loss of control over intake, and emergence of a negative emotional state during withdrawal [10]. The transition from controlled, recreational use to pathological addiction represents a dramatic dysregulation of brain motivational circuits, driven by progressively worsening neurobiological adaptations [52] [10]. This process can be understood through the conceptual framework of allostasis—a process of achieving stability through physiological change during chronic challenge, distinct from homeostasis which maintains stability within a narrow range [52]. With repeated drug exposure, the brain's reward and stress systems establish a new, pathological equilibrium (allostatic state) through cumulative neuroadaptations. The long-term cost of maintaining this altered state is termed allostatic load, which manifests as a persistent deficit in reward function and heightened stress sensitivity that drives escalating drug use despite adverse consequences [52].
The neurobiology of addiction centers on three primary brain circuits: the basal ganglia (reward and habit formation), the extended amygdala (stress and negative affect), and the prefrontal cortex (executive control and decision-making) [4] [18] [10]. This technical review examines how allostatic processes within and between these circuits create reward deficits that fuel the escalating addiction cycle, providing drug development professionals with a comprehensive framework for targeted therapeutic interventions.
The Addiction Cycle: Neurocircuitry and Neurotransmitter Dynamics
Addiction progresses through a three-stage cycle that worsens over time and involves specific neurocircuitry and neuroplastic changes [4] [11] [18]. The table below summarizes the primary brain regions, neurotransmitter changes, and behavioral manifestations associated with each stage.
Table 1: Neurobiological Characteristics of the Three-Stage Addiction Cycle
| Stage | Key Brain Regions | Primary Neurotransmitter Changes | Behavioral Manifestation |
|---|---|---|---|
| Binge/Intoxication | Basal ganglia (ventral striatum, nucleus accumbens), ventral tegmental area | ↑ Dopamine, ↑ Opioid peptides, ↑ GABA [10] | Euphoria, incentive salience, positive reinforcement |
| Withdrawal/Negative Affect | Extended amygdala (central nucleus of amygdala, bed nucleus of stria terminalis) | ↓ Dopamine, ↑ Corticotropin-releasing factor (CRF), ↑ Dynorphin [52] [10] | Dysphoria, anxiety, irritability, negative reinforcement |
| Preoccupation/Anticipation | Prefrontal cortex (orbitofrontal, dorsolateral), basolateral amygdala, hippocampus | ↑ Glutamate, ↑ CRF, Altered dopamine [10] | Craving, impaired executive function, compulsivity |
The progression through these stages involves a shift from impulsivity (early stage, driven by positive reinforcement) to compulsivity (late stage, driven by negative reinforcement) [10]. This transition is marked by two key neurobiological processes: within-system adaptations (changes in the same neural systems that mediate acute drug reward) and between-system adaptations (recruitment of alternative neural systems not initially involved in drug reward) [52]. The allostatic load develops as these adaptations accumulate, creating a persistent deficit state that becomes increasingly difficult to reverse.
Figure 1: The Three-Stage Addiction Cycle and Associated Neurocircuitry. This diagram illustrates the interconnected stages of addiction, their primary neural substrates, and key neurotransmitter changes. BNST = bed nucleus of stria terminalis; CeA = central nucleus of amygdala; NAcc = nucleus accumbens; OFC = orbitofrontal cortex; dlPFC = dorsolateral prefrontal cortex; CRF = corticotropin-releasing factor.
Molecular Mechanisms of Allostatic Load in Reward Circuits
Dopamine System Dysregulation: The Within-System Adaptation
The mesolimbic dopamine system, originating in the ventral tegmental area (VTA) and projecting to the nucleus accumbens and other forebrain regions, constitutes the primary reward circuit initially activated by drugs of abuse [7] [52]. All drugs of abuse, with the possible exception of opioids, acutely increase dopamine release in the nucleus accumbens, producing euphoria and reinforcing drug-taking behavior [52]. However, with repeated administration, within-system adaptations occur that diminish dopamine function:
- Reduced tonic dopamine levels: Chronic drug exposure leads to a decrease in basal dopamine release in the nucleus accumbens, reducing the sensitivity to natural rewards and creating an anhedonic state [52] [18].
- Altered receptor sensitivity: The brain compensates for excessive dopamine stimulation by reducing dopamine D2 receptor availability in the striatum, further diminishing reward sensitivity [7].
- Shift from reward to cue response: Dopamine firing patterns transform from responding to the drug reward itself to anticipating reward-related stimuli (incentive salience), explaining cravings triggered by drug-associated cues [18].
These changes are quantified through intracranial self-stimulation (ICSS) thresholds in animal models, where escalating drug use produces a compensatory increase in reward thresholds that reflects a hedonic allostasis—the brain's attempt to counteract chronic reward system activation [52]. This resetting of the reward system creates a persistent deficit state wherein individuals require the drug merely to function at a normal emotional baseline, a key marker of allostatic load.
Stress System Recruitment: The Between-System Adaptation
Concurrent with dopamine system dysregulation, between-system adaptations recruit brain stress circuits not initially involved in drug reward [52]. The extended amygdala, particularly through its bed nucleus of the stria terminalis (BNST) and central nucleus of the amygdala (CeA), becomes hyperactive during withdrawal and mediates the negative emotional state that drives negative reinforcement [52] [18] [10]. Key molecular players in this between-system adaptation include:
- Corticotropin-releasing factor (CRF): Extrahypothalamic CRF systems in the extended amygdala become hyperactive during withdrawal, producing anxiety-like and stress-like responses [52] [10].
- Dynorphin systems: This endogenous κ-opioid receptor ligand is upregulated in the nucleus accumbens during withdrawal, producing dysphoric effects that counter dopamine-mediated reward [10].
- Noradrenergic systems: Locus coeruleus norepinephrine activity increases during withdrawal, contributing to anxiety, arousal, and stress sensitivity [10].
These stress systems are normally buffered by anti-reward neurotransmitters including neuropeptide Y, nociceptin, and endocannabinoids [18] [10]. In addiction, this buffering capacity is diminished, creating an imbalance that favors stress surfeits over reward. The resulting negative emotional state becomes a powerful driver of escalated drug use through negative reinforcement—taking drugs to relieve the dysphoria and anxiety of withdrawal [52] [10].
Table 2: Key Neurotransmitter Systems in Allostatic Load Development
| System | Change in Addiction | Brain Regions Involved | Functional Consequences |
|---|---|---|---|
| Dopamine | Decreased basal release, altered phasic release | VTA, nucleus accumbens, prefrontal cortex | Reduced reward sensitivity, anhedonia, enhanced cue reactivity |
| CRF | Increased extrahypothalamic activity | Extended amygdala (CeA, BNST) | Anxiety, stress sensitivity, negative affect |
| Dynorphin | Increased activity | Nucleus accumbens, VTA, extended amygdala | Dysphoria, aversion, counter-modulation of dopamine |
| Glutamate | Altered prefrontal regulation | Prefrontal cortex, nucleus accumbens, amygdala | Executive dysfunction, impaired control, enhanced craving |
| Endocannabinoids | Decreased signaling | Prefrontal cortex, amygdala, striatum | Reduced buffering of stress, enhanced vulnerability |
Genetic Vulnerability and Research Methodologies
Genetic Vulnerability to Allostatic Load
Individual differences in vulnerability to addiction reflect genetic variations that influence how neural circuits respond to drugs and adapt to chronic exposure. Genome-wide association studies (GWAS) have identified specific risk loci associated with substance use disorders:
- CHRNA2 and cannabis use disorder: A GWAS of cannabis use disorder (CUD) identified a genome-wide significant risk locus (rs56372821) that is a strong expression quantitative trait locus (eQTL) for CHRNA2, which encodes the neuronal acetylcholine receptor alpha-2 subunit [53]. Reduced expression of CHRNA2 in cerebellar tissue is associated with CUD, suggesting a potential target for therapeutic intervention [53].
- Nicotinic receptor genes: Evidence of natural selection has been identified in nicotinic receptor gene clusters (CHRNs) on chromosomes 8 and 15, which are associated with nicotine, alcohol, and cocaine dependence [54]. These genes may have been selected for their effects on cognitive function, with incidental effects on addiction vulnerability.
- Opioid receptor polymorphisms: Studies of opioid receptor genes (OPRD1, OPRK1, OPRM1) have yielded mixed results, with some populations showing no significant associations between specific polymorphisms and opioid use disorder, highlighting the importance of demographic factors and population-specific genetic architecture [55].
These genetic findings support the concept that individual variations in addiction vulnerability have a strong neurobiological basis, which may inform the development of personalized treatment approaches targeting specific molecular pathways.
Experimental Approaches for Studying Allostatic Mechanisms
Research on allostatic load in addiction employs sophisticated methodological approaches across multiple levels of analysis. The table below outlines key experimental paradigms and their applications in studying reward deficits and allostatic load.
Table 3: Experimental Approaches for Studying Allostatic Load in Addiction
| Methodology | Key Measures | Applications in Allostasis Research | Technical Considerations |
|---|---|---|---|
| Intracranial Self-Stimulation (ICSS) | Brain reward thresholds | Quantifies hedonic set point changes; elevated thresholds indicate reward deficits [52] | Requires specialized equipment and surgical expertise |
| In vivo Microdialysis | Extracellular neurotransmitter levels | Measures dynamic changes in dopamine, glutamate, CRF during addiction cycle [10] | Limited temporal and spatial resolution |
| Fast-Scan Cyclic Voltammetry | Rapid dopamine transients | Captures phasic dopamine signaling to drugs and cues [10] | Excellent temporal resolution but limited to few neurotransmitters |
| Animal Self-Administration Models | Escalation of intake, motivation (progressive ratio), resistance to punishment | Models transition from controlled to compulsive use [52] [10] | Requires extensive training periods; species differences matter |
| Brain Imaging (fMRI, PET) | Regional brain activation, receptor availability, connectivity | Translates animal findings to humans; identifies neural correlates of craving and withdrawal [10] | Expensive; limited molecular specificity |
Figure 2: Experimental Workflow for Studying Allostatic Load in Addiction. This diagram outlines a multi-method research approach for investigating allostatic mechanisms, integrating genetic, behavioral, neurochemical, and circuit-level analyses to identify therapeutic targets. BG = basal ganglia; EA = extended amygdala; PFC = prefrontal cortex; CRF = corticotropin-releasing factor.
Table 4: Key Research Reagents for Investigating Allostatic Mechanisms in Addiction
| Reagent/Resource | Primary Application | Research Utility |
|---|---|---|
| Dopamine Receptor Ligands (e.g., SCH23390 for D1, raclopride for D2) | Receptor binding and localization studies | Quantifying receptor changes in allostatic state [10] |
| CRF Receptor Antagonists (e.g., antalarmin, R121919) | Stress system manipulation | Testing CRF involvement in withdrawal-negative affect stage [52] [10] |
| κ-Opioid Receptor Agonists/Antagonists (e.g., U50488, nor-BNI) | Dynorphin system modulation | Probing dysphoria mechanisms in reward deficits [10] |
| DREADDs (Designer Receptors Exclusively Activated by Designer Drugs) | Circuit-specific neuronal manipulation | Establishing causal role of specific pathways in addiction cycle [11] |
| CRISPR/Cas9 Systems | Gene editing in specific cell types | Validating candidate genes from human genetic studies [53] |
| Fast-Acting Opioid Sensors (e.g., FLASHO) | Real-time opioid signaling monitoring | Tracking endogenous opioid dynamics during addiction stages |
| Transgenic Animal Models | Gene- and circuit-specific studies | Modeling human genetic vulnerabilities in addiction [54] |
Implications for Therapeutic Development
Understanding addiction through the lens of allostatic load and reward deficits provides a framework for developing targeted therapies that address specific stages of the addiction cycle and their underlying neuroadaptations [4]. Potential therapeutic approaches include:
- Dopamine stabilization: Medications that normalize dopamine function without producing abuse liability, such as partial dopamine agonists or drugs that enhance tonic versus phasic dopamine release [10].
- Stress system targets: CRF receptor antagonists, κ-opioid receptor antagonists, and neuropeptide Y enhancers to mitigate the negative emotional state of withdrawal [52] [18] [10].
- Glutamate modulators: Drugs that restore prefrontal cortex control over drug-seeking behavior, such as metabotropic glutamate receptor modulators or N-acetylcysteine [10].
- Genetic-based interventions: Personalized medicine approaches that target specific genetic vulnerabilities identified through GWAS and other genomic studies [53] [4].
The allostatic model emphasizes that addiction creates persistent neuroadaptations that remain long after detoxification, explaining high relapse rates and highlighting the need for long-term management strategies similar to other chronic diseases [4]. This perspective also reduces stigma by framing addiction as a neurobiological disorder rather than a moral failing, potentially improving treatment engagement and social support [4] [18].
The transition from controlled drug use to addiction represents a profound dysregulation of brain reward and stress systems, characterized by allostatic load that manifests as persistent reward deficits and enhanced stress sensitivity. This pathological state emerges through within-system adaptations in dopamine circuits and between-system recruitment of stress circuits, primarily involving the basal ganglia, extended amygdala, and prefrontal cortex [52] [10]. The resulting cycle of worsening motivation and emotional regulation creates a self-perpetuating pattern of compulsive drug seeking and taking that becomes increasingly difficult to reverse.
Future research directions should focus on: (1) identifying specific molecular mechanisms that mediate transition through addiction stages; (2) developing biomarkers to quantify allostatic load in humans; (3) creating circuit-specific interventions that target distinct addiction stages; and (4) exploring epigenetic mechanisms that mediate gene-environment interactions in addiction vulnerability [4] [18]. By targeting the specific neuroadaptations that drive allostatic load in addiction, researchers and drug development professionals can develop more effective therapeutic strategies that address the core neurobiology of this debilitating disorder.
Substance use disorder (SUD) represents a dramatic dysregulation of motivational circuits, characterized by a transition from voluntary, goal-directed drug use to compulsive drug-seeking behaviors that persist despite adverse consequences [10]. This transition is underpinned by long-lasting neuroadaptations within specific brain circuitry, particularly the corticostriatal loops that interconnect the prefrontal cortex, basal ganglia, and extended amygdala [56] [57]. Research over the past two decades has demonstrated that addiction is fundamentally a disorder of neuroplasticity, where drugs of abuse co-opt molecular mechanisms normally involved in learning and memory to create powerful, pathological associations [58] [56]. The resulting changes strengthen corticostriatal synapses in a manner that promotes compulsive behavior while diminishing cognitive control, creating a cycle of addiction that involves three interrelated stages: binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation (craving) [3] [10].
The neuroplasticity observed in addiction shares remarkable similarities with the mechanisms underlying long-term memory formation [56]. Steven Hyman aptly noted that "memory disorders are often thought of as conditions involving memory loss, but what if the brain remembers too much or too powerfully records pathological associations?" [56]. This perspective frames addiction as a pathological learning process in which drugs produce exceptionally powerful memories that hijack normal reward circuits [56]. The corticostriatal pathways, which integrate cognitive, motivational, and sensory information to guide behavioral output, undergo profound plasticity during this transition, resulting in a shift from prefrontal cortical control to striatal-dominated habitual responding [59] [10].
Molecular Mechanisms of Neuroplasticity in Corticostriatal Circuits
Synaptic Plasticity Mechanisms: LTP, LTD, and Homeostatic Imbalances
At the synaptic level, drugs of abuse induce plasticity through mechanisms that mirror those involved in normal learning and memory, primarily through the modulation of glutamatergic signaling and dopaminergic neurotransmission [56]. Long-term potentiation (LTP) and long-term depression (LTD), the primary cellular models for learning and memory, are profoundly altered by repeated drug exposure [56]. These processes typically involve N-methyl-D-aspartate (NMDA) receptor-mediated trafficking of α-amino-3-hydroxyl-5-methyl-4-isoxazole-propionate (AMPA) receptors to and from the cell surface [56].
A pivotal discovery in addiction neuroscience revealed that a single exposure to cocaine causes enhancement of synaptic strength at excitatory synapses on dopamine neurons in the ventral tegmental area (VTA), measured as an increased AMPA/NMDA ratio [56] [60]. This drug-evoked potentiation has since been demonstrated with virtually all major drugs of abuse, including amphetamine, morphine, ethanol, nicotine, and benzodiazepines, but not with psychoactive substances lacking abuse potential [56]. This convergence of cellular responses provides a neural mechanism by which initial neuroadaptations underlying addiction are triggered [56].
Table 1: Key Synaptic Plasticity Changes in Corticostriatal Circuits in Addiction
| Brain Region | Plasticity Change | Functional Consequence | Experimental Evidence |
|---|---|---|---|
| Ventral Tegmental Area (VTA) | Increased AMPA/NMDA ratio at excitatory synapses on DA neurons | Enhanced responsiveness to drug-associated stimuli; initiation of pathological reward learning | Observed 24h after single cocaine exposure; occludes subsequent LTP [56] |
| Nucleus Accumbens (NAc) | Increased dendritic spine density in medium spiny neurons | Persistent changes facilitating drug-seeking behavior | Observed in rats self-administering cocaine; persists during abstinence [58] |
| Orbitofrontal Cortex to Dorsal Striatum pathway | Increased AMPA/NMDA ratio | Compulsive reward-seeking despite punishment | Selectively observed in mice that persevered when seeking was punished [61] |
| Prefrontal Cortex to NAc pathway | Imbalance in glutamate homeostasis; reduced basal extracellular glutamate | Potentiated release of synaptic glutamate during drug-seeking | Found after withdrawal from chronic cocaine; contributes to relapse [58] |
In the striatum, chronic drug exposure leads to imbalances in glutamate homeostasis that profoundly affect synaptic plasticity [58]. Following withdrawal from chronic cocaine, researchers have observed a marked disruption in glutamate regulation, with both cystine-glutamate exchange and glutamate uptake being reduced in the nucleus accumbens [58]. This imbalance is associated with reduced basal extracellular glutamate levels alongside a potentiated release of synaptic glutamate during drug-seeking behavior [58]. Additionally, there is a basal increase in the AMPA to NMDA current ratio and a loss of both LTP and LTD, indicating a fundamental disruption in the brain's ability to regulate synaptic strength appropriately [58].
Intracellular Signaling Cascades and Transcriptional Regulation
The synaptic changes described above are mediated and maintained by intricate intracellular signaling cascades that ultimately lead to alterations in gene expression [59]. Combined signaling of dopamine and glutamate systems, particularly through dopamine D1 receptors and NMDA receptors, triggers critical intracellular pathways that lead to changes in chromatin structure, gene expression, synaptic plasticity, and ultimately behavior [59]. A key pathway involves NMDA receptor activation leading to calcium influx, which activates multiple signaling mechanisms, several of which converge on Extracellular Receptor signaling Kinase (ERK) [59]. ERK regulates a variety of transcription factors that coordinate the formation and stabilization of long-term memories [59].
DeltaFosB, a particularly stable transcription factor, accumulates in dopamine terminals in the cortex and striatum following repeated exposure to all drugs of abuse tested [58]. This transcription factor appears to be involved in the development of motivated behaviors, and disruption of this process blocks the development of drug-associated plasticity such as behavioral sensitization [58]. Additionally, the brain-derived neurotrophic factor (BDNF) has been implicated in the incubation of drug craving, with levels increasing in the ventral tegmental area and nucleus accumbens during withdrawal from cocaine, correlating with enhanced relapse susceptibility [58].
Figure 1: Intracellular Signaling Cascade in Addiction. This diagram illustrates the key molecular pathways through which drugs of abuse trigger neuroplasticity changes that lead to compulsive drug-seeking behavior. The convergence of dopamine and glutamate signaling activates intracellular cascades that ultimately alter gene expression and synaptic function.
Key Brain Regions and Neurocircuitry in the Transition to Compulsivity
The Corticostriatal Network: Basal Ganglia, Prefrontal Cortex, and Extended Amygdala
The transition from goal-directed to compulsive drug seeking involves coordinated dysregulation across three key brain networks: the basal ganglia, responsible for reward and habit formation; the prefrontal cortex, governing executive function and behavioral control; and the extended amygdala, mediating stress and negative affect [3] [10]. These regions form interconnected loops that normally work in concert to regulate motivated behavior, but become dysregulated in addiction [10].
The basal ganglia, particularly the nucleus accumbens (NAc) and dorsal striatum, play crucial roles in the binge/intoxication stage of addiction [3] [10]. The NAc serves as a key interface for integrating sensory, reward, and motor information [59], while the dorsal striatum becomes increasingly involved as drug use progresses from voluntary to habitual [10]. Research has demonstrated that NMDAR blockade in the NAc impairs initial operant learning but does not affect subsequent performance, highlighting its role in the acquisition phase of drug-seeking behavior [59].
The prefrontal cortex (PFC), including the orbitofrontal cortex (OFC) and dorsolateral prefrontal cortex (DLPFC), is crucial for executive functions such as decision-making, inhibitory control, and behavioral flexibility [3] [57]. In addiction, PFC dysfunction contributes to the loss of control over drug intake and the inability to resist drug-related cues [57]. Imaging studies have consistently shown that addicted individuals exhibit reduced metabolic activity in the ACC, OFC, and DLPFC, which correlates with impairments in self-regulation [57]. Recent research has specifically implicated the orbitofrontal cortex to dorsal striatum pathway in compulsive reward-seeking, with increased AMPA/NMDA ratio observed selectively at these synapses in individuals who persist in drug-seeking despite punishment [61].
The extended amygdala plays a pivotal role in the withdrawal/negative affect stage of addiction, driving the negative emotional state that emerges when drug use is discontinued [10]. This region is involved in stress responses and contributes to the dysphoria, anxiety, and irritability that characterize drug withdrawal [10]. As addiction progresses, the motivation for drug use shifts from positive reinforcement (seeking pleasure) to negative reinforcement (relief from distress) [10].
Table 2: Brain Regions Implicated in the Transition to Compulsive Drug Seeking
| Brain Region | Primary Function in Addiction | Key Neuroadaptations | Associated Behavioral Manifestation |
|---|---|---|---|
| Nucleus Accumbens | Integration of reward, motor, and sensory information | Increased spine density; altered glutamate homeostasis | Incentive salience; initial drug reinforcement |
| Dorsal Striatum | Habit formation and compulsive drug-seeking | Increased AMPA/NMDA ratio at OFC inputs; strengthened corticostriatal synapses | Loss of control; habitual drug use |
| Prefrontal Cortex | Executive control; decision-making | Reduced metabolic activity; decreased D2 receptors; impaired glutamate signaling | Poor decision-making; failure of inhibitory control |
| Orbitofrontal Cortex | Value representation; outcome expectancy | Altered connectivity with striatum; synaptic changes | Compulsive drug-seeking despite negative consequences |
| Extended Amygdala | Stress and negative affect | Increased CRF and dynorphin; decreased reward function | Negative emotional state; negative reinforcement |
| Ventral Tegmental Area | Dopamine signaling; reward processing | Increased AMPA/NMDA ratio; enhanced synaptic strength | Enhanced drug reward; sensitization |
Neurocircuitry Model of Addiction
The transition to compulsivity can be understood through a three-stage cycle model of addiction that worsens over time and involves all three brain regions [10]. In the binge/intoxication stage, drugs activate dopamine and opioid systems in the basal ganglia, reinforcing drug use and promoting the formation of habits [10]. During the withdrawal/negative affect stage, decreased activity in reward systems combined with recruitment of brain stress systems in the extended amygdala creates a negative emotional state [10]. In the preoccupation/anticipation stage, dysregulation of prefrontal cortex circuits and disrupted glutamate signaling lead to craving and deficits in executive function that promote relapse [10].
This neurocircuitry model helps explain the shift from impulsivity to compulsivity that characterizes addiction [10]. Initially, drug use is driven by positive reinforcement and impulsive behavior seeking immediate gratification [10]. As neuroadaptations accumulate across the corticostriatal loops, drug use becomes increasingly compulsive—perseverative, repetitive actions that are excessive and inappropriate, often performed to reduce the tension or anxiety associated with withdrawal [10].
Figure 2: Corticostriatal Neurocircuitry in Addiction. This diagram illustrates the key brain circuits involved in the transition to compulsive drug-seeking, highlighting the interactions between prefrontal control regions and subcortical reward and habit centers that become dysregulated in addiction.
Experimental Models and Methodological Approaches
Behavioral Paradigms for Studying Compulsivity
Animal models have been essential for elucidating the neurobiological mechanisms underlying the transition to compulsive drug seeking [10]. These models attempt to capture specific dimensions of addiction, with the most relevant focusing on the compulsive aspects of drug use [10]. Several key behavioral paradigms have been developed to model different aspects of addiction:
Operant self-administration is a fundamental procedure where animals are trained to perform an instrumental response (e.g., lever press or nose poke) to receive a drug infusion [56]. This model allows researchers to study drug-taking behavior in a controlled setting and can be modified to examine progression to compulsivity by introducing punishment contingencies or progressive ratio schedules [61] [10].
The extinction/reinstatement model is widely used to study relapse-like behavior [56]. After animals learn to self-administer a drug, the drug is removed and the operant behavior is extinguished [56]. Reinstatement of drug-seeking can then be triggered by various factors known to precipitate relapse in humans, including drug priming, exposure to drug-associated cues, or stress [56]. This model has demonstrated that the tendency to relapse actually increases over time, a phenomenon termed "incubation" that is associated with increasing levels of BDNF in the reward circuitry [58].
Conditioned place preference (CPP) is an indirect measure of drug reward based on classical conditioning principles [56]. In this paradigm, one environment is paired with drug administration while another is paired with vehicle [56]. A preference for the drug-paired environment is taken as a measure of the rewarding properties of the drug [56].
To specifically model compulsivity, researchers have developed punished seeking paradigms where animals continue to seek drugs despite adverse consequences [61]. For example, in a seek-take chain schedule, animals must complete a seeking response before gaining access to a taking response that delivers the drug [61]. Compulsivity is measured by persistence in the seeking response when it is paired with punishment [61].
The Scientist's Toolkit: Key Research Reagents and Methodologies
Table 3: Essential Research Reagents and Methods for Studying Corticostriatal Plasticity
| Reagent/Method | Primary Application | Key Findings Enabled | Technical Considerations |
|---|---|---|---|
| NMDA receptor antagonists (e.g., AP-5) | Block NMDA receptors to study their role in plasticity | Demonstrated necessity of NMDAR activation for initial operant learning [59] | Site-specific microinjections required; timing critical for differentiating acquisition vs performance |
| Viral vector systems (optogenetics/chemogenetics) | Circuit-specific manipulation of neuronal activity | Identified OFC to DS pathway as critical for compulsive seeking [61] | Requires precise targeting and validation; off-target effects possible |
| Microdialysis | Measure extracellular neurotransmitter levels | Revealed glutamate homeostasis disruptions in NAc during withdrawal [58] | Limited temporal resolution; technically challenging in small rodents |
| Electrophysiology (patch-clamp recordings) | Measure synaptic strength (AMPA/NMDA ratios) | Discovered drug-evoked potentiation at VTA synapses [56] [60] | Requires brain slice preparation; technically demanding |
| Fiber photometry | Record neural activity in behaving animals | Detected activity peaks in DS at reward availability cue [61] | Limited to population-level activity; calibration critical |
| DREADDs (Designer Receptors Exclusively Activated by Designer Drugs) | Chemogenetic control of specific neuronal populations | Allowed temporal control of OFC neurons to establish causal role in compulsivity [61] | Ligand pharmacokinetics important; potential off-target effects |
| N-acetylcysteine | Restore glutamate homeostasis via cystine-glutamate exchange | Demonstrated reduced cocaine seeking in rats and humans [58] | Systemic administration; potential peripheral effects |
| Positron Emission Tomography (PET) | Measure receptor availability (e.g., D2 receptors) in humans | Revealed D2 receptor decreases in striatum of addicted individuals [57] | Limited spatial resolution; radiation exposure |
| Functional Magnetic Resonance Imaging (fMRI) | Measure brain activity and connectivity | Identified abnormal cortico-striatal connectivity in addiction [57] | Indirect measure of neural activity; motion artifacts problematic |
Technical Protocols for Key Experiments
Protocol: Measuring Synaptic Plasticity at Corticostriatal Synapses Using Electrophysiology
This protocol outlines the procedure for measuring drug-induced synaptic plasticity at corticostriatal synapses using brain slice electrophysiology, based on methods that have revealed fundamental mechanisms in addiction neuroscience [56] [60].
Materials and Reagents:
- Artificial cerebrospinal fluid (aCSF) containing (in mM): 126 NaCl, 2.5 KCl, 1.2 MgCl2, 2.4 CaCl2, 1.2 NaH2PO4, 21.4 NaHCO3, and 11.1 glucose
- Brain slicing solution (sucrose-aCSF) containing (in mM): 210 sucrose, 2.5 KCl, 1.2 NaH2PO4, 25 NaHCO3, 0.5 CaCl2, 7 MgCl2, and 7 glucose
- CNQX (AMPA receptor antagonist), 20 μM
- AP-5 (NMDA receptor antagonist), 50 μM
- Picrotoxin (GABA_A receptor antagonist), 100 μM
- Patch pipettes (3-5 MΩ resistance) filled with internal solution containing (in mM): 130 cesium methanesulfonate, 5 NaCl, 1 MgCl2, 0.02 EGTA, 10 HEPES, 2 Mg-ATP, 0.1 GTP, and 5 QX-314
Procedure:
- Prepare acute brain slices (300 μm thickness) from drug-treated and control animals using a vibrating microtome
- Maintain slices in oxygenated (95% O2/5% CO2) aCSF at 32°C for at least 1 hour before recording
- Identify corticostriatal neurons in the dorsal striatum using infrared differential interference contrast microscopy
- Obtain whole-cell voltage-clamp recordings at -70 mV to isolate AMPA receptor-mediated currents
- Electrically stimulate cortical afferents to the striatum at 0.1 Hz
- Record AMPA receptor-mediated excitatory postsynaptic currents (EPSCs)
- Change holding potential to +40 mV and record in presence of CNQX (20 μM) to isolate NMDA receptor-mediated EPSCs
- Calculate AMPA/NMDA ratio by dividing the peak amplitude of AMPA EPSCs (-70 mV) by the amplitude of NMDA EPSCs (+40 mV) measured 50 ms after the stimulus
- Analyze paired-pulse ratio by delivering two stimuli at 50 ms interval to assess presynaptic function
- For LTP induction, apply high-frequency stimulation (100 Hz for 1 second) while clamping the cell at 0 mV
Data Interpretation: An increased AMPA/NMDA ratio indicates enhanced synaptic strength at corticostriatal synapses, suggesting strengthening of these connections following drug exposure [61] [56]. This measure has been particularly informative for identifying specific pathway alterations in compulsive individuals [61].
Protocol: Assessing Compulsive Drug-Seeking Using a Seek-Take Chain Schedule
This protocol details the procedure for evaluating the transition to compulsive reward-seeking using an optogenetic self-stimulation paradigm in mice, adapted from methods that have successfully identified neural correlates of compulsivity [61].
Materials and Reagents:
- Operant conditioning chambers with two retractable levers/response manipulanda
- Optical fibers and laser system for optogenetic stimulation
- AAV vectors encoding Channelrhodopsin-2 (ChR2) under control of a dopamine neuron-specific promoter (e.g., TH-Cre)
- Programmable punishment delivery system (e.g., footshock or air puff)
- Behavioral software for data collection and analysis
Procedure:
- Surgically implant mice with AAV-TH-ChR2-EYFP or control virus in the ventral tegmental area (VTA)
- Allow 3-4 weeks for viral expression before behavioral testing
- Habituate mice to operant chambers and establish baseline activity
- Train mice on a seek-take chain schedule where:
- Completion of the "seek" response (e.g., nose poke) results in presentation of the "take" lever
- Completion of the "take" response results in delivery of optogenetic stimulation of VTA dopamine neurons
- Establish stable seeking and taking behavior (typically 10-14 daily sessions)
- Introduce punishment contingencies to the seeking component:
- Gradually increase probability of mild footshock (0.3 mA, 0.5 s) upon seek response completion
- Continue until punishment probability reaches 100%
- Classify mice as "compulsive" or "non-compulsive" based on persistence of seeking behavior under punishment
- For neural activity recording, implant fiber optic cannulas above the dorsal striatum
- Use fiber photometry to record calcium signals during seek and take components
- For inhibition experiments, use optogenetic hyperpolarization of spiny projection neurons time-locked to the cue predicting reward availability
Data Analysis: Compare the number of completed seek-take sequences between punishment conditions and between experimental groups. Compulsive mice will persist in seeking despite punishment, whereas non-compulsive mice will suppress seeking behavior [61]. Neural activity peaks in the dorsal striatum at the moment of signaled reward availability are predictive of compulsive phenotypes [61].
Figure 3: Experimental Workflow for Studying Compulsive Drug-Seeking. This diagram outlines the key steps in a comprehensive approach to investigate the neural mechanisms underlying compulsive drug-seeking behavior, incorporating behavioral assessment, neural recording, and causal manipulation.
Therapeutic Implications and Future Directions
The understanding that addiction involves specific neuroplastic changes in corticostriatal circuits has opened promising avenues for therapeutic intervention [58] [57]. As addiction is increasingly viewed as a chronic brain disorder with a strong neurobiological basis, treatments have shifted from simple detoxification to approaches that target the underlying neural adaptations [3] [58].
One promising approach involves restoring glutamate homeostasis in the nucleus accumbens [58]. Research has demonstrated that administration of N-acetylcysteine, which promotes cystine-glutamate exchange, can normalize glutamate levels and reduce drug-seeking in animal models [58]. Clinical trials have shown that N-acetylcysteine reduces desire for cocaine in human addicts and may also be effective for pathological gambling and cigarette smoking [58]. Similarly, the antibiotic ceftriaxone has been shown to increase glutamate uptake and prevent reinstatement of cocaine seeking in animal models [58].
Another innovative approach involves real-time functional magnetic resonance imaging biofeedback of brain activity [58]. This method allows addicted individuals to learn to activate frontal control mechanisms to inhibit drug craving, potentially strengthening compromised executive control circuits [58]. Early studies suggest that providing feedback of frontal activation can help patients learn to activate inhibitory structures and suppress drug craving [58].
Deep brain stimulation (DBS) and repeated transcranial magnetic stimulation (rTMS) represent more direct methods for modulating corticostriatal circuits [57]. These techniques allow targeted manipulation of specific brain regions and have shown promise in treating refractory cases of addiction, though further research is needed to optimize parameters and identify ideal candidates [57].
Future research directions include developing more specific pharmacological agents that target the molecular pathways involved in drug-induced neuroplasticity without disrupting normal learning and memory [56]. The advent of optogenetics and chemogenetics has enabled unprecedented precision in manipulating specific neural circuits, allowing researchers to establish causal relationships between circuit activity and behavior [61]. These technologies, combined with increasingly sophisticated animal models that better capture the complexity of addiction, hold great promise for identifying novel treatment targets [61] [10].
As research progresses, the focus is shifting toward personalized medicine approaches that account for individual differences in the specific neuroadaptations underlying addiction [10]. Understanding how genetic and environmental factors influence vulnerability to these neuroplastic changes will be crucial for developing more effective, individualized treatments for substance use disorders [56] [10].
Substance use disorders are chronic, relapsing conditions characterized by a compulsion to seek and take drugs, loss of control over intake, and emergence of a negative emotional state when access to the drug is prevented [10] [11]. The disorder follows a recurrent cycle of binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation (craving) that worsens over time and involves fundamental neuroplastic changes in brain reward, stress, and executive systems [3] [10]. A core challenge in treatment remains the persistent vulnerability to relapse, with more than 60% of individuals treated for substance use disorders experiencing relapse within the first year after discharge from treatment, and many remaining at elevated risk for years afterward [3]. This persistence is driven by enduring neuroadaptations in specific brain circuits that become hypersensitive to drug-associated cues and contribute to craving long after acute withdrawal has subsided.
Revolutionary advances in neurobiology have transformed our understanding of addiction from a moral failing to a chronic brain disease with identifiable circuitry dysregulations [3]. Research has consistently identified three key brain regions as central to the pathology of addiction: the basal ganglia (reward and habit formation), the extended amygdala (stress and negative affect), and the prefrontal cortex (executive control and regulation) [3] [11]. This whitepaper examines the circuit mechanisms underlying cue-reactivity and long-term relapse vulnerability, synthesizing current evidence from neurobiological, neuroimaging, and behavioral research to provide a comprehensive framework for researchers and drug development professionals.
The Addiction Cycle: A Three-Stage Framework
The addiction cycle can be conceptualized as a three-stage, recurring pattern that progressively worsens, driven by neuroplastic changes in distinct but interconnected brain systems. The following table summarizes the core features, neural substrates, and key neurotransmitters for each stage:
Table 1: The Three-Stage Cycle of Addiction
| Stage | Core Features | Primary Neural Substrates | Key Neurotransmitter Changes |
|---|---|---|---|
| Binge/Intoxication | Reward, pleasure, initial use | Ventral tegmental area (VTA), nucleus accumbens (ventral striatum) | ↑ Dopamine, ↑ opioid peptides, ↑ serotonin [10] |
| Withdrawal/Negative Affect | Dysphoria, anxiety, irritability | Extended amygdala (central nucleus, bed nucleus of stria terminalis) | ↑ Corticotropin-releasing factor (CRF), ↑ dynorphin, ↑ norepinephrine; ↓ dopamine, ↓ serotonin [10] [62] |
| Preoccupation/Anticipation (Craving) | Craving, drug-seeking, relapse | Prefrontal cortex (orbitofrontal, anterior cingulate, dorsolateral), basolateral amygdala, hippocampus | ↑ Glutamate, ↑ corticotropin-releasing factor, ↑ dopamine in prefrontal regions [10] [11] |
This framework provides a heuristic for understanding how substance use progresses from controlled intake to compulsive addiction, with each stage reinforcing the next in a self-perpetuating cycle [10] [11]. The transition through these stages involves a shift from positive reinforcement (seeking the pleasurable effects of the drug) to negative reinforcement (seeking relief from the negative emotional state of withdrawal) [10]. As individuals continue to misuse substances, progressive neuroadaptations occur in the structure and function of the brain that compromise normal cognitive and emotional functioning and drive the transition from occasional, controlled use to chronic misuse that is difficult to control [3].
Neurocircuitry of Craving and Relapse Vulnerability
The Basal Ganglia: Reward Processing and Habit Formation
The basal ganglia, particularly the ventral striatum (including the nucleus accumbens) and dorsal striatum, play pivotal roles in the binge/intoxication stage of addiction by processing drug reward and facilitating the formation of habitual drug-seeking behaviors [3] [17]. All addictive substances directly or indirectly increase dopamine transmission in the nucleus accumbens, producing their initial rewarding effects and motivating repeated use [3] [17]. Human imaging studies using positron emission tomography (PET) have shown that intoxicating doses of alcohol and drugs release dopamine and opioid peptides into the ventral striatum, with fast and steep dopamine release associated with the subjective sensation of being "high" [10].
The striatum is functionally organized in a ventral-to-dorsal spiral gradient that facilitates the transition from goal-directed to habitual drug use [17]. The nucleus accumbens in the ventral striatum serves as the initial interface where reward information is processed, while progressive engagement of the dorsomedial striatum and eventually the dorsolateral striatum underlies the development of compulsive drug-seeking habits that are resistant to devaluation [17]. This progression is facilitated by dopaminergic transmission within striato-nigro-striatal (SNS) ascending spirals that connect the ventral striatum with more dorsal regions [17]. Research demonstrates that the dorsolateral striatum is critical for the operation of Pavlovian instrumental transfer - the process by which drug-associated cues alter motivational salience and trigger drug-seeking behavior - highlighting its role in cue-reactivity [17].
The Extended Amygdala: Stress and Negative Affect
The extended amygdala, comprising the bed nucleus of the stria terminalis, central amygdala, and possibly a transition zone in the nucleus accumbens shell, represents the core stress system of the brain and plays a fundamental role in the withdrawal/negative affect stage of addiction [3] [11] [62]. This region is activated during acute drug withdrawal and protracted abstinence, generating the negative emotional state that drives drug-seeking through negative reinforcement mechanisms [62]. Two key neurotransmitter systems in the extended amygdala - corticotropin-releasing factor (CRF) and norepinephrine - are critically involved in the motivational aspects of dependence and relapse [62].
During withdrawal from all major drugs of abuse, extracellular CRF increases in the central nucleus of the amygdala, creating a feedforward cycle that promotes anxiety-like responses and escalated drug self-administration [62]. Pharmacological blockade of CRF receptors or norepinephrine receptors in the extended amygdala reduces both the anxiety-like behaviors associated with withdrawal and the increased drug-seeking that occurs during abstinence [62]. These findings highlight the critical role of brain stress systems in the extended amygdala in the negative reinforcement processes that perpetuate addiction once dependence has developed.
The Prefrontal Cortex: Executive Control and Craving
The prefrontal cortex (PFC), particularly the orbitofrontal cortex (OFC), anterior cingulate cortex (ACC), and dorsolateral prefrontal cortex (DLPFC), is integral to the preoccupation/anticipation stage of addiction, mediating craving, executive function, and self-regulation [11] [16]. In addiction, the PFC undergoes significant functional disruptions that result in a syndrome of impaired response inhibition and salience attribution (iRISA) [16]. This syndrome is characterized by attributing excessive salience to drugs and drug-related cues, decreased sensitivity to non-drug reinforcers, and decreased ability to inhibit maladaptive behaviors [16].
The following table summarizes specific PFC dysfunctions in addiction:
Table 2: Prefrontal Cortex Dysfunction in Addiction
| PFC Subregion | Normal Function | Dysfunction in Addiction |
|---|---|---|
| Orbitofrontal Cortex (OFC) | Value representation, outcome expectation, decision-making | Drug-related anticipation, choice of immediate reward, inaccurate predictions [16] |
| Anterior Cingulate Cortex (ACC) | Conflict monitoring, error detection, attention | Attention bias toward drug cues, inflexibility in goals to procure drugs [16] |
| Dorsolateral PFC (DLPFC) | Working memory, behavioral control, planning | Impaired self-monitoring, formation of memory biased toward drug-related stimuli [16] |
Neuroimaging studies have consistently shown that drug cues activate the OFC and ACC in addicted individuals but not in controls, and the magnitude of this activation correlates with self-reported craving [16]. Additionally, the DLPFC shows reduced activity during tasks requiring inhibitory control, contributing to impulsivity and inability to suppress drug-seeking behaviors even in the face of negative consequences [16]. These PFC disruptions are thought to result from both the neurotoxic effects of chronic drug exposure and the neuroadaptations that occur in response to repeated drug administration.
Cue-Reactivity and Relapse Vulnerability: Mechanisms and Evidence
The Role of Conditioned Cues in Relapse
Drug-associated cues (people, places, paraphernalia, internal states) that are repeatedly paired with drug use acquire conditioned incentive properties through Pavlovian conditioning mechanisms [63] [64]. These cues can subsequently trigger powerful conditioned responses, including physiological reactions, craving, and drug-seeking behavior, even after extended periods of abstinence [63]. In learning-based models of behavior, drug effects serve as unconditioned responses, while cues regularly present during drug use become conditioned stimuli that elicit conditioned responses [65]. Clinical studies have consistently demonstrated that humans respond to drug-associated cues with craving along with conditioned sympathetic activation, dopamine release, and associated neural activity in regions including the ventral striatum [65].
A comprehensive meta-analysis of 237 studies representing 51,788 participants found a significant prospective association between drug cues and craving and subsequent drug use and relapse, with an odds ratio of 2.05 (95% CI, 1.94-2.15) [65]. This indicates that cue-reactivity and craving more than double the odds of future drug use or relapse. Moderator analyses revealed that some of the strongest associations were found for cue-induced craving, real cues or images, and studies using ecological momentary assessment [65]. These findings provide compelling evidence that drug cue-reactivity and craving are core mechanisms underlying drug use and relapse.
Neurobiology of Cue-Induced Craving
Cue-induced craving involves distributed neural systems that process reward, emotion, memory, and executive control. Functional magnetic resonance imaging (fMRI) studies have consistently identified activation in the ventral striatum, orbitofrontal cortex, anterior cingulate cortex, amygdala, and prefrontal cortex when addicted individuals are exposed to drug-related cues [63] [66]. The mesolimbic dopamine system, originating in the ventral tegmental area and projecting to the nucleus accumbens, is critically involved in attributing incentive salience to drug cues, making them powerful triggers for motivation and action [10] [11].
Additionally, the insula appears to play a specialized role in craving by integrating interoceptive signals (bodily states) with emotional and motivational information to create conscious drug urges [11]. Patients with damage to the insula have been reported to experience a complete disappearance of addiction, suggesting this region is necessary for maintaining craving and drug-seeking behavior [11]. The persistence of neural responses to drug cues, even after extended abstinence, helps explain the long-term vulnerability to relapse that characterizes addiction.
Experimental Models and Methodologies
Animal Models of Relapse
Animal models have been instrumental in elucidating the neurobiological mechanisms underlying relapse vulnerability. The most widely used model is the reinstatement procedure, where animals are trained to self-administer drugs, subjected to extinction (where responses no longer produce drug), and then tested for renewal of drug-seeking behavior following exposure to various triggers [64]. Three primary stimuli reliably reinstate drug-seeking in this model: (1) exposure to drug-associated cues, (2) stress, and (3) priming doses of the drug itself [64]. This model has demonstrated strong predictive validity for understanding human relapse phenomena.
Other important animal models include:
- Conditioned place preference: Measures the rewarding properties of drugs by assessing an animal's preference for environments paired with drug effects [64]
- Incubation of craving model: Demonstrates that cue-induced craving actually increases rather than decreases during the first months of abstinence [64]
- Extended access models: Animals with extended access to drugs (6+ hours daily) more rapidly develop escalation of intake and compulsive-like patterns, mimicking the transition to addiction in humans [11]
These models permit investigations of specific neuroadaptations at molecular, cellular, and circuit levels that mediate the transition from occasional substance use to loss of control and chronic addiction [10].
Human Neuroimaging Approaches
Human studies have benefited greatly from brain-imaging technologies, particularly functional magnetic resonance imaging (fMRI) and positron emission tomography (PET), which allow researchers to non-invasively investigate biochemical, functional, and structural changes in the living human brain [3] [63]. The Addiction Craving and Cue Reactivity Initiative Network (ACRIN) has emerged as a collaborative framework to improve methodological rigor and reporting standards for addiction craving and cue reactivity research [66]. Key methodological approaches include:
- fMRI drug cue-reactivity (FDCR): Presents drug-related cues while measuring brain activation patterns [66]
- PET imaging: Measures neurotransmitter dynamics (dopamine, opioid) during cue exposure or drug administration [10] [63]
- Ecological momentary assessment (EMA): Collects real-time data on craving and cue exposure in natural environments [65]
These approaches have consistently identified the neural circuitry underlying cue-reactivity, including the ventral striatum, extended amygdala, and prefrontal regions, with increased activation in these areas predicting elevated relapse risk [63] [16].
Research Reagents and Methodological Toolkit
Table 3: Essential Research Reagents and Methodologies for Studying Cue-Reactivity and Relapse
| Category | Specific Tools/Assays | Research Application |
|---|---|---|
| Animal Models | Reinstatement model, conditioned place preference, incubation of craving, extended access self-administration | Modeling relapse phenomena, transition to addiction [64] |
| Neuroimaging Approaches | fMRI cue-reactivity paradigms, PET with dopamine/opioid radioligands, ecological momentary assessment | Measuring neural responses to cues, neurotransmitter dynamics, real-world craving [65] [63] [66] |
| Pharmacological Probes | CRF receptor antagonists, dopamine receptor agonists/antagonists, opioid receptor ligands, norepinephrine modulators | Testing neurochemical mechanisms, potential treatments [10] [62] |
| Genetic Tools | CRISPR-mediated gene editing, transgenic animals, viral vector-mediated gene manipulation (optogenetics, chemogenetics) | Identifying molecular mechanisms, circuit-specific functions [10] |
| Cue Reactivity Paradigms | Standardized drug cue databases, craving self-reports (single-item and multi-item scales), physiological measures | Assessing cue-induced craving, standardized measurement across studies [66] |
Neuroadaptations and Molecular Mechanisms
Chronic drug use induces enduring neuroadaptations at molecular, cellular, and circuit levels that persist long after substance use stops and contribute to the persistence of craving and relapse vulnerability [3] [11]. Key neuroadaptations include:
- Dopamine system dysregulation: Reduced dopamine D2 receptor availability and blunted dopamine responsiveness to natural rewards, creating an reward deficit state [11] [16]
- Glutamatergic plasticity: Alterations in AMPA and NMDA receptor function and distribution, particularly in the nucleus accumbens and prefrontal cortex, that enhance responsiveness to drug cues [11]
- CRF system activation: Persistent upregulation of CRF signaling in the extended amygdala that maintains a negative emotional state and stress reactivity [62]
- Transcriptional and epigenetic changes: Alterations in gene expression profiles through mechanisms such as ΔFosB accumulation, histone modifications, and DNA methylation that maintain addictive states [10]
These neuroadaptations collectively create a brain state characterized by heightened sensitivity to drug cues, reduced sensitivity to natural rewards, elevated stress reactivity, and compromised executive control - a combination that powerfully perpetuates the addiction cycle and maintains vulnerability to relapse [3] [11] [16].
Visualizing the Core Neurocircuitry of Addiction
The following Graphviz diagram illustrates the primary neural circuits and their interactions in the addiction cycle:
Implications for Treatment Development
Understanding the persistent neuroadaptations in the basal ganglia, extended amygdala, and prefrontal cortex provides critical insights for developing more effective treatments for addiction. Current evidence suggests that successful interventions must address multiple aspects of the addiction cycle:
- Medications: Development of compounds that target specific components of the addiction cycle, including dopamine partial agonists, CRF antagonists, glutamate modulators, and opioid system modulators [10] [62]
- Neuromodulation approaches: Transcranial magnetic stimulation (TMS) and direct current stimulation (tDCS) targeting prefrontal regions to enhance executive control and reduce craving [66]
- Behavioral interventions: Cue exposure therapy, contingency management, and cognitive training designed to reverse maladaptive learning and strengthen cognitive control [63]
- Combined approaches: Simultaneously targeting multiple systems (reward, stress, executive control) to address the comprehensive circuitry dysfunction in addiction [11] [16]
The identification of specific circuit dysfunctions also enables the development of biomarkers for predicting treatment response and relapse risk. For example, fMRI drug cue-reactivity has shown promise as a biomarker for treatment development, though it has not yet received FDA approval [66]. Future research focusing on cross-species validation of cue-reactivity measures and large-scale consortium studies (such as ENIGMA-ACRI) will be crucial for advancing biomarker development and personalized treatment approaches [66].
The persistence of cravings and long-term relapse vulnerability in addiction is mediated by enduring neuroadaptations in interconnected brain circuits involving the basal ganglia, extended amygdala, and prefrontal cortex. The transition through the addiction cycle - from binge/intoxication to withdrawal/negative affect to preoccupation/anticipation - involves a cascade of neuroplastic changes that progressively strengthen drug-seeking habits while weakening executive control and enhancing stress reactivity. Cue-reactivity emerges as a core mechanism through which drug-associated stimuli acquire powerful control over behavior, with compelling evidence demonstrating that drug cues and craving significantly predict future drug use and relapse.
Future research directions should focus on: (1) elucidating the molecular mechanisms that maintain long-term neuroadaptations, (2) developing cross-species biomarkers for cue-reactivity and relapse vulnerability, (3) optimizing neuromodulation and pharmacological approaches that target specific circuit dysfunctions, and (4) implementing early intervention strategies that prevent the progression from initial drug use to addiction. By building on our current understanding of the neurocircuitry of addiction, researchers and drug development professionals can work toward more effective strategies for addressing the persistent challenge of relapse in substance use disorders.
Drug addiction is a chronic relapsing disorder characterized by a compulsive drive to seek and take drugs despite adverse consequences. This whitepaper explores the neurobiological framework of hedonic allostasis, a process wherein chronic drug use leads to persistent alterations in brain reward circuits. Through within-system and between-system neuroadaptations, the reward system establishes a new, pathological set point characterized by a decreased sensitivity to natural rewards and an elevated emotional baseline. This review details the triple-stage addiction cycle—binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation—and its underlying neurocircuitry, focusing on the basal ganglia, extended amygdala, and prefrontal cortex. We present quantitative data from key studies, detailed experimental protocols, and essential research tools to facilitate the development of novel therapeutic strategies.
The allostatic model of addiction posits that chronic drug consumption induces a persistent dysregulation of brain reward pathways, leading to a new, maladaptive equilibrium [67] [68]. Unlike homeostasis, which maintains stability through negative feedback mechanisms to a fixed set point, allostasis achieves stability through change, establishing a new operating level via feed-forward mechanisms [68]. In addiction, this results in a chronic deviation of the hedonic set point [69]. The addict's physiological state is maintained functional through this affective adaptation, rather than by a return to the original homeostatic balance. This framework explains the transition from initial drug use for pleasure ("feeling high") to later use to relieve the distress of a negative emotional state ("feeling normal") [70] [69]. This allostatic state is fueled by dysregulation of both reward circuits and the activation of brain stress systems, manifesting as compulsive drug-taking and a loss of control over intake [67].
Neurocircuitry of the Addiction Cycle
The addiction cycle is a spiraling process that involves three core stages, each mediated by distinct but overlapping neurocircuits [3] [11] [10]. The progression through this cycle involves a shift from positive reinforcement (driven by the pleasurable effects of the drug) to negative reinforcement (driven by the relief of withdrawal) [10].
Binge/Intoxication Stage: This initial stage is centered on the acute rewarding and reinforcing effects of drugs. The basal ganglia, particularly the nucleus accumbens (NAc) in the ventral striatum, is the key neurocircuit. All major drugs of abuse directly or indirectly increase dopamine levels in the NAc, reinforcing drug-taking behavior and establishing incentive salience [3] [10]. With repeated use, control over drug-seeking shifts from the ventral to the dorsal striatum, promoting the formation of compulsive habits [11] [10].
Withdrawal/Negative Affect Stage: When drug access is prevented, a negative emotional state emerges (e.g., dysphoria, anxiety, irritability). This stage is mediated by the extended amygdala and its stress systems. Key neuroadaptations include a decrease in reward system function (e.g., reduced dopamine signaling) and a recruitment of brain stress neurotransmitters, notably corticotropin-releasing factor (CRF) and dynorphin [70] [10] [68]. This combination produces the hypernegative emotional state that drives negative reinforcement.
Preoccupation/Anticipation Stage: This stage involves craving and deficits in executive function that lead to relapse. The prefrontal cortex (PFC), including the orbitofrontal cortex and anterior cingulate cortex, is critically involved. Dysregulation of glutamate projections from the PFC to the basal ganglia and extended amygdala compromises top-down control, impairing decision-making and promoting impulsivity [11] [10]. The basolateral amygdala and hippocampus are also involved in cue-induced craving and context-driven relapse.
Core Mechanisms: Hedonic Dysregulation and Set-Point Change
The transition to addiction is marked by two key categories of neuroadaptation that drive hedonic allostasis.
Within-System Neuroadaptations
Within-system adaptations occur within the very brain reward circuits that are initially activated by the drug. The primary response is a counter-adaptation to chronic drug exposure. For example, repeated surges in dopamine lead to a compensatory reduction in baseline dopamine function and a blunted response to the drug itself and to natural rewards [70] [69]. This manifests as tolerance to the drug's pleasurable effects and a generalized hedonic hyposensitivity, or anhedonia.
Between-System Neuroadaptations
Between-system adaptations occur when chronic drug exposure recruits other brain systems that oppose the reward function of the drug. The primary between-system change is the engagement of brain stress systems, primarily within the extended amygdala [69] [68]. The key mediators are CRF and the κ-opioid receptor agonist dynorphin. These systems are activated during withdrawal, producing anxiety-like and dysphoric states that contribute to the negative emotional state and drive negative reinforcement [70] [68].
Quantitative Data from Key Preclinical Studies
The following tables summarize critical quantitative findings from animal studies that form the empirical foundation for the hedonic allostasis model.
Table 1: Escalation of Drug Intake and Increased Motivation in Extended Access Models
| Drug | Access Paradigm | Key Behavioral Outcome | Reported Effect Size | Citation |
|---|---|---|---|---|
| Cocaine | 1 hr vs 6 hr daily | Escalation of daily intake | ~150% increase over 1 hr baseline | [70] |
| Cocaine | Short vs Long Access | Breakpoint on Progressive-Ratio | Significant increase (higher motivation) | [70] |
| Heroin | Extended Access | Persistently increased motivation | Lasting increase post-escalation | [67] |
| Alcohol | Dependence-Induced | Alcohol self-administration | ~50-100% increase in dependent rats | [70] |
Table 2: Neurobiological Markers of Hedonic Allostasis in Dependent Animals
| Measured Parameter | Technique | Brain Region | Change in Dependence | Citation |
|---|---|---|---|---|
| Brain Reward Threshold | Intracranial Self-Stimulation (ICSS) | Medial Forebrain Bundle | Significant Increase (Anhedonia) | [70] |
| Dopamine D2 Receptor | Receptor Binding | Ventral Striatum | Decrease | [10] |
| Extracellular CRF | Microdialysis | Central Nucleus of Amygdala | Significant Increase | [70] [68] |
| Anxiety-like Response | Elevated Plus Maze | - | Significant Increase | [70] |
Detailed Experimental Protocols
To investigate hedonic allostasis, researchers employ sophisticated behavioral paradigms paired with neurobiological measures. Below are detailed protocols for two cornerstone experiments.
Protocol: Escalation of Self-Administration
This model demonstrates the transition from controlled to compulsive drug use [70] [67].
- Subjects: Laboratory rats (e.g., Long-Evans or Wistar), surgically implanted with a chronic intravenous catheter into the jugular vein.
- Apparatus: Standard operant conditioning chambers equipped with two levers (active and inactive), a cue light, and a syringe pump for drug infusion.
- Training:
- Animals are trained to self-administer the drug (e.g., cocaine, 0.25-1.0 mg/kg/infusion) on a Fixed-Ratio 1 (FR1) schedule of reinforcement.
- Each active lever press results in a drug infusion paired with a discrete cue light.
- Sessions last for 1-2 hours per day until stable intake is established (variability < 10% over 3 consecutive days).
- Experimental Manipulation:
- Short Access (ShA) Group: Continues with 1-hour daily sessions.
- Long Access (LxA) Group: Switched to extended daily sessions (e.g., 6 hours).
- Key Dependent Variables:
- Primary: Number of infusions earned per session.
- Secondary: The temporal pattern of intake within the session.
- Expected Outcome: The LxA group will show a progressive escalation in daily drug intake over approximately 10-14 days, while the ShA group will maintain stable, low intake.
Protocol: Brain Stimulation Reward (ICSS) Threshold Measurement
This procedure directly measures the hedonic set point by quantifying the reward threshold [70] [68].
- Subjects: Rats implanted with a bipolar stimulating electrode targeting the medial forebrain bundle at the level of the lateral hypothalamus.
- Apparatus: Operant chamber with a single lever connected to a constant-current stimulator.
- Habilitation & Training:
- Animals recover from surgery and are habituated to the chamber.
- They are trained to lever press for sinusoidal electrical stimulation (e.g., 100-200 µA, 0.1-0.3 s, 60-100 Hz).
- Threshold Determination (Discrete-Trials Current-Threshold Procedure):
- Each trial begins with a non-contingent stimulus.
- The animal has a limited time (e.g., 7.5 s) to respond (lever press) to receive a second, contingent stimulus identical to the first.
- A series of descending and ascending current intensities are presented.
- The threshold for a given series is defined as the midpoint between the current level that supports responding and the level that does not.
- Experimental Manipulation:
- Baseline reward thresholds are established over several days.
- Animals are then subjected to a chronic drug regimen (e.g., chronic escalating-dose cocaine, or alcohol vapor to induce dependence).
- ICSS thresholds are measured at specified time points during intoxication, acute withdrawal (e.g., 8-12 hours post-drug), and protracted abstinence (days to weeks).
- Key Dependent Variable: The current intensity threshold (in µA) required to maintain responding.
- Expected Outcome: Chronic drug administration and withdrawal will produce a significant and persistent elevation in brain reward thresholds, reflecting a decrease in reward function and an increase in the hedonic set point.
Visualization of Key Signaling Pathways
The following diagram summarizes the primary neurotransmitter systems dysregulated across the three stages of the addiction cycle, highlighting the shift from dopamine-centric reward to CRF/dynorphin-centric stress.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents for Investigating Hedonic Allostasis
| Reagent / Tool | Category | Primary Function in Research | Example Application |
|---|---|---|---|
| SCH 23390 | Pharmacological | Selective dopamine D1 receptor antagonist. | To block the reinforcing effects of cocaine and assess D1 receptor contribution to reward [67] [11]. |
| Corticotropin-Releasing Factor (CRF) | Neuropeptide | Agonist of the CRF system. | To directly induce stress-like and anxiogenic effects in behavioral assays [68]. |
| CRF Receptor Antagonists (e.g., Antalarmin) | Pharmacological | Block CRF type 1 (CRF1) receptors. | To reverse dependence-induced increases in drug self-administration and anxiety-like behavior [70] [68]. |
| Nor-Binaltorphimine (nor-BNI) | Pharmacological | Selective kappa-opioid receptor (KOR) antagonist. | To block the dysphoric effects of dynorphin, reducing the negative affect stage of withdrawal [10]. |
| Intracranial Self-Stimulation (ICSS) | Technical Assay | Directly measures brain reward function. | To quantify changes in reward thresholds (hedonic set-point) during drug withdrawal [70] [68]. |
| Microdialysis | Technical Assay | Measures extracellular neurotransmitter levels in vivo. | To document elevated CRF levels in the amygdala during withdrawal [70]. |
| Viral Vector Systems (e.g., DREADDs) | Molecular Tool | Allows chemogenetic manipulation of specific neuronal populations. | To causally test the role of defined neural circuits in drug-seeking and relapse [10]. |
The hedonic allostasis model provides a powerful framework for understanding addiction as a chronic brain disease characterized by a pathologically reset reward set-point. The core pathology involves dysregulations across three interacting neurocircuits—the basal ganglia, extended amygdala, and prefrontal cortex—driven by sequential within-system and between-system neuroadaptations. This leads to a self-perpetuating cycle where drug use is compulsively driven to alleviate the negative emotional state of withdrawal, rather than to achieve pleasure.
Future research must focus on translating these preclinical findings into novel therapeutics. Promising targets include:
- CRF1 Receptor Antagonists: To mitigate the powerful stress component that drives negative reinforcement.
- Dynorphin/KOR System Modulators: To alleviate the dysphoric state associated with withdrawal.
- Glutamate System Modulators: To restore cognitive control and weaken maladaptive drug-related memories.
A deep understanding of the allostatic neuroadaptations detailed in this review is paramount for the rational design of the next generation of addiction treatments, moving beyond simple detoxification to address the persistent vulnerability to relapse that defines this disorder.
Drug addiction represents a chronically relapsing disorder characterized by compulsion to seek and take drugs, loss of control over intake, and emergence of a negative emotional state during withdrawal [10] [11]. Despite significant advances in neurobiology, the translation of preclinical findings to effective clinical treatments has been hampered by substantial methodological challenges. The profound heterogeneity in addiction symptomatology and treatment response reflects complex interactions within neural circuitry that are difficult to capture in laboratory settings [71] [72]. This technical guide addresses the core methodological hurdles in modeling addiction phenotypes, with specific focus on the neurocircuitry framework encompassing basal ganglia, extended amygdala, and prefrontal cortex systems.
The addiction cycle can be conceptualized as a three-stage, recurring pattern—binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation (craving)—that worsens over time and involves distinct but interacting neuroplastic changes [10] [11]. Each stage involves specific neural substrates: the ventral tegmental area and ventral striatum for the binge/intoxication stage, the extended amygdala for the withdrawal/negative affect stage, and the prefrontal cortex, orbitofrontal cortex, dorsal striatum, and basolateral amygdala for the preoccupation/anticipation stage [11]. Methodologically, this requires developing models that not only capture these distinct phases but also their transitions and interactions, while accounting for the individual differences that characterize addiction vulnerability and expression [71].
Theoretical Framework: Neural Circuitry of Addiction
Key Neurocircuits in Addiction Pathology
Addiction pathophysiology involves progressive dysregulation of three primary brain circuits: the basal ganglia reward system, the extended amygdala stress system, and the prefrontal cortex executive system [10]. These systems do not operate in isolation but rather form an integrated network whose disruption leads to the characteristic symptoms of addiction. The transition from voluntary drug use to compulsive addiction involves a shift from impulsive to compulsive behavior mediated by neuroadaptations in these circuits [11] [73].
The basal ganglia play a critical role in reward processing and habit formation, with a progressive ventral to dorsal striatal shift underlying the transition from goal-directed to habitual drug seeking [74]. The extended amygdala (including central amygdala, bed nucleus of stria terminalis, and shell of nucleus accumbens) mediates the negative emotional state of withdrawal through brain stress systems such as corticotropin-releasing factor (CRF) and norepinephrine [73]. The prefrontal cortex exhibits compromised executive function that contributes to impaired response inhibition and salience attribution (iRISA syndrome) [16].
Neurotransmitter Systems Across the Addiction Cycle
Table 1: Key Neurotransmitter Changes Across Addiction Stages
| Addiction Stage | Neurotransmitter | Direction of Change | Primary Brain Regions |
|---|---|---|---|
| Binge/Intoxication | Dopamine | Increase | Ventral striatum, ventral tegmental area |
| Opioid peptides | Increase | Ventral striatum | |
| γ-aminobutyric acid (GABA) | Increase | Ventral tegmental area, nucleus accumbens | |
| Withdrawal/Negative Affect | Corticotropin-releasing factor (CRF) | Increase | Extended amygdala |
| Dynorphin | Increase | Extended amygdala | |
| Dopamine | Decrease | Ventral striatum | |
| Neuropeptide Y | Decrease | Extended amygdala | |
| Preoccupation/Anticipation | Glutamate | Increase | Prefrontal cortex to nucleus accumbens |
| Dopamine | Increase | Prefrontal cortex | |
| Corticotropin-releasing factor (CRF) | Increase | Extended amygdala |
The neurochemical dysregulation across the addiction cycle illustrates the complex methodological challenge of modeling temporal dynamics in laboratory settings [10]. Different neurotransmitter systems predominate at different stages, requiring careful temporal consideration in experimental design.
Figure 1: Core Neurocircuitry in the Three-Stage Addiction Cycle
Methodological Framework: The Addictions Neuroclinical Assessment (ANA) Approach
Conceptual Foundation
The Addictions Neuroclinical Assessment (ANA) framework has been proposed as a clinical approach for addressing heterogeneity in addiction by focusing on three core neurofunctional domains: incentive salience, negative emotionality, and executive (dys)function [71]. This framework provides a structured methodology for modeling complex addiction phenotypes by focusing on fundamental behavioral processes rather than surface-level symptoms. The ANA leverages deep phenotyping with factor analytic methods to construct core neurofunctional domains that can be applied across different substances and populations [71].
The ANA domains align with the three-stage addiction cycle and corresponding neurocircuitry: incentive salience maps to the binge/intoxication stage and basal ganglia function; negative emotionality maps to the withdrawal/negative affect stage and extended amygdala function; and executive function maps to the preoccupation/anticipation stage and prefrontal cortex function [71]. This alignment provides a theoretical bridge between laboratory models and clinical manifestations.
Experimental Paradigms for Domain Assessment
Table 2: Experimental Paradigms for Assessing ANA Domains
| ANA Domain | Behavioral Assay | Physiological/Neural Measures | Translational Validation |
|---|---|---|---|
| Incentive Salience | Pavlovian Instrumental Transfer | fMRI in ventral striatum, dopamine release | Correlation with drug cue reactivity |
| Conditioned Place Preference | Neuronal activation in VTA-NAc pathway | Predictive of relapse vulnerability | |
| Negative Emotionality | Elevated Plus Maze | CRF release in extended amygdala | Association with stress-induced craving |
| Defensive burying test | Heart rate variability, cortisol response | Correlation with negative reinforcement motives | |
| Executive Function | Stop Signal Reaction Time | fMRI in prefrontal cortex, DLPFC activation | Association with treatment retention |
| Probabilistic Reversal Learning | Theta band oscillations in anterior cingulate | Predictive of behavioral flexibility in recovery |
Technical Approaches: Modeling Circuit-Based Addiction Phenotypes
Modeling the Ventral to Dorsal Striatal Progression
The transition from goal-directed to habitual drug seeking represents a core process in addiction that can be modeled through serial connectivity between ventral and dorsal striatal regions [74]. The striatonigrostriatal (SNS) spiral circuitry provides an anatomical substrate for this progression, with dopaminergic transmission facilitating the shift from action-outcome to stimulus-response processing [17].
Experimental Protocol: Serial Connectivity in Striatal Pathways
- Objective: To assess the progression from ventral to dorsal striatal control in drug-seeking behavior
- Subjects: Rodents with targeted genetic manipulations or human subjects with stratified addiction severity
- Apparatus: Operant conditioning chambers with cue presentation systems
- Procedure:
- Train subjects on variable interval schedules for drug reinforcement
- Conduct outcome devaluation tests to determine goal-directed vs. habitual behavior
- Use chemogenetics or fMRI to map functional connectivity between NAc and DLS
- Assess dopamine release dynamics across striatal subregions using microdialysis or PET
- Measures: Response persistence following devaluation, neural activation patterns, dopamine transients
This protocol captures the critical shift from ventral striatum-mediated positive reinforcement to dorsal striatum-mediated habitual responding that characterizes addiction [17] [74].
Modeling Extended Amygdala Stress Systems
The extended amygdala plays a key role in the negative emotional state of withdrawal through recruitment of brain stress neurotransmitters such as CRF, dynorphin, and norepinephrine [73]. Modeling this domain requires assays that capture both the physiological and affective components of withdrawal.
Experimental Protocol: Brain Stress Response Assessment
- Objective: To quantify extended amygdala hyperactivity during withdrawal
- Subjects: Dependent subjects following chronic drug administration
- Apparatus: Behavioral test chambers with capacity for physiological monitoring
- Procedure:
- Establish dependence through chronic drug administration
- Measure anxiety-like behaviors during acute withdrawal (e.g., social interaction, startle response)
- Administer CRF antagonists and α2-adrenergic agonists to reverse withdrawal phenotypes
- Use microdialysis to measure CRF and norepinephrine release in extended amygdala
- Measures: Behavioral anxiety indices, stress neurochemical levels, autonomic responses
This approach models the negative reinforcement processes that become increasingly important as addiction progresses [73].
Figure 2: Comprehensive Workflow for Addiction Phenotyping
Modeling Prefrontal Cortex Dysfunction
The prefrontal cortex (PFC) exhibits compromised function across multiple domains in addiction, including impaired response inhibition, salience attribution, and executive control [16]. The iRISA (impaired Response Inhibition and Salience Attribution) syndrome provides a framework for modeling these deficits.
Experimental Protocol: Prefrontal Cortex Executive Function
- Objective: To assess PFC-dependent cognitive control in addiction
- Subjects: Human substance users or rodents with PFC-targeted manipulations
- Apparatus: fMRI-compatible tasks or operant systems with cognitive testing capabilities
- Procedure:
- Administer Go/No-Go or Stop Signal Task to measure response inhibition
- Conduct monetary incentive delay task to assess salience attribution
- Use fMRI to measure PFC activation patterns during task performance
- Apply transcranial magnetic stimulation (TMS) or optogenetics to modulate PFC activity
- Measures: Response inhibition accuracy, BOLD signal in PFC subregions, behavioral flexibility
This protocol models the compromised executive function that contributes to relapse vulnerability [16].
Research Reagents and Technical Tools
Table 3: Essential Research Reagents for Addiction Circuitry Studies
| Reagent/Tool | Application | Mechanism/Function | Example Use |
|---|---|---|---|
| DREADDs (Designer Receptors Exclusively Activated by Designer Drugs) | Circuit-specific manipulation | Chemogenetic control of neural activity | Targeting ventral vs. dorsal striatum pathways |
| CRF Receptor Antagonists | Stress system modulation | Block CRF1 receptors in extended amygdala | Reducing withdrawal-induced drug seeking |
| Dopamine Receptor Ligands (e.g., SCH 23390, raclopride) | Dopamine system interrogation | D1/D2 receptor antagonism | Determining receptor contributions to drug reward |
| Fiber Photometry Systems | Neural activity monitoring | Real-time calcium or neurotransmitter imaging | Measuring dopamine dynamics during drug seeking |
| Fast-Scan Cyclic Voltammetry | Dopamine release measurement | Electrochemical detection of dopamine transients | Quantifying phasic dopamine signals |
| Optogenetic Constructs (e.g., Channelrhodopsin) | Precise neural circuit control | Light-activated ion channels | Establishing causal circuit relationships |
Computational Modeling Approaches
Computational approaches provide powerful methods for understanding intersubject variability in addiction by modeling the interaction between ventral and dorsal cortico-striatal circuits [72]. Reinforcement learning frameworks can simulate how imbalances between model-based (goal-directed) and model-free (habitual) control systems contribute to addiction phenotypes.
Implementation Protocol: Computational Modeling of Circuit Interactions
- Objective: To simulate endophenotypic differences in addiction vulnerability
- Framework: Dual-system reinforcement learning model with model-based and model-free components
- Parameters:
- Learning rates for drug vs. natural rewards
- Balance parameter between model-based and model-free systems
- Discounting factors for delayed outcomes
- Simulation Conditions: Varying degrees of circuit dominance (ventral vs. dorsal striatum)
- Output Measures: Addiction development likelihood, symptom severity, treatment response
This approach reveals that endophenotypic differences in the balance between circuit systems result in an inverted-U shape in optimal choice behavior, with greater unbalance leading to higher addiction vulnerability [72].
Integration and Future Directions
Overcoming methodological hurdles in addiction modeling requires integrated approaches that span multiple levels of analysis—from molecular and circuit mechanisms to behavior and computation. The future of addiction modeling lies in developing approaches that can capture individual differences in circuit dysfunction while maintaining translational relevance to the human condition. This includes incorporating developmental stage, sex differences, environmental factors, and polysubstance use into existing models to better reflect the complexity of addiction in human populations.
The methodological framework presented here—centered on the ANA domains and their underlying neural circuits—provides a structured approach for advancing our understanding and treatment of addiction. By employing circuit-specific assays, targeted manipulations, and computational modeling, researchers can deconstruct the complexity of addiction into tractable experimental approaches while maintaining sight of the integrated nature of this disorder.
Validation and Comparative Analysis: Shared Circuitry in Behavioral Addictions and Comorbidities
Addiction, whether to substances or behaviors, is a chronically relapsing disorder characterized by compulsion, loss of control, and emergence of negative emotional states upon cessation. Contemporary neuroscience research reveals that both drug and behavioral addictions involve shared neurocircuitry, primarily encompassing the basal ganglia, extended amygdala, and prefrontal cortex. This review provides an in-depth analysis of the neurobiological overlaps and divergences between these addiction forms, synthesizing evidence from functional neuroimaging, molecular studies, and behavioral models. We detail the specific roles of key brain regions across the binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation stages of the addiction cycle. The data support a model wherein drugs of abuse and compulsive natural rewards "hijack" common reward and motivation pathways, inducing similar neuroplastic adaptations that drive the transition from controlled to compulsive engagement.
Addiction is increasingly conceptualized as a disorder of brain circuits that transcends the traditional substance-based classification. Non-drug addictions—including pathological gambling, compulsive eating, sexual behavior, shopping, and excessive internet use—manifest behavioral patterns strikingly similar to substance use disorders, including craving, impaired control, tolerance, withdrawal, and high relapse rates [75]. Prevalence estimates indicate these behavioral addictions are common, affecting 1-2% for pathological gambling, 5% for compulsive sexual behavior, 2.8% for binge-eating disorder, and 5-6% for compulsive buying in the United States [75].
The neurocircuitry of addiction provides a unified framework for understanding both drug and behavioral addictions. This framework centers on three core brain regions—the basal ganglia, extended amygdala, and prefrontal cortex—that form dynamic networks responsible for reward processing, stress response, and executive control [10] [11] [4]. The present review examines the specific neuroadaptations within these circuits across different addiction types, highlighting both convergent and divergent mechanisms through a comparative circuitry analysis.
Neuroanatomy of Addiction: Core Circuits and Functions
Key Brain Regions in Addiction Pathology
Table 1: Core Neural Circuits in Drug and Behavioral Addictions
| Brain Region | Primary Functions in Addiction | Associated Behavioral Manifestations |
|---|---|---|
| Basal Ganglia (particularly ventral striatum/nucleus accumbens) | Processes reward effects; forms habitual substance taking; part of brain's "reward circuit" [4]; mediates incentive salience [10] | Euphoria of drug high; diminished sensitivity to natural rewards; habitual compulsive behaviors |
| Extended Amygdala | Mediates stress responses; produces feelings of unease, anxiety, and irritability during withdrawal [4]; key substrate for negative reinforcement [10] | Negative emotional states in withdrawal; using drugs/behaviors for temporary relief from discomfort |
| Prefrontal Cortex | Mediates executive function (organizing thoughts, prioritizing tasks, managing time, decision-making) [4]; exerts control over substance taking [10] | Compulsive drug/behavior seeking with reduced impulse control; impaired decision-making |
The Three-Stage Addiction Cycle
Addiction can be conceptualized as a repeating three-stage cycle—binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation—that worsens over time and involves specific neuroplastic changes in brain reward, stress, and executive systems [10] [11] [4]. This framework applies to both substance and behavioral addictions, with individuals typically moving from impulsivity to compulsivity as the disorder progresses [10].
Figure 1: The Three-Stage Addiction Cycle and Associated Brain Regions
Comparative Neurocircuitry Analysis
Binge/Intoxication Stage: Shared Reward Pathways
The binge/intoxication stage is primarily associated with the acute rewarding or pleasurable effects of substances or behaviors, heavily involving the basal ganglia and its "reward circuit" [4]. Both drugs of abuse and non-drug rewards activate the mesocorticolimbic dopamine system, with the ventral tegmental area (VTA) and nucleus accumbens as focal points [11].
Neurotransmitter Dynamics: Drugs of abuse produce fast, steep increases in dopamine release in the ventral striatum, activating low-affinity dopamine D1 receptors necessary for rewarding effects [10]. Similar dopamine release patterns occur with non-drug rewards, though the magnitude and temporal dynamics may differ. Functional neuroimaging studies in humans show that gambling, shopping, orgasm, playing video games, and viewing appetizing food activate many of the same mesocorticolimbic regions as drugs of abuse [75] [76].
Table 2: Neurotransmitter Changes Across the Addiction Cycle
| Neurotransmitter/Neuromodulator | Binge/Intoxication Stage | Withdrawal/Negative Affect Stage | Preoccupation/Anticipation Stage |
|---|---|---|---|
| Dopamine | Increase [10] | Decrease [10] | Increase [10] |
| Opioid Peptides | Increase [10] | Decrease (receptors) [10] | - |
| Glutamate | - | - | Increase [10] |
| Corticotropin-Releasing Factor (CRF) | - | Increase [10] | Increase [10] |
| Dynorphin | - | Increase [10] | - |
| Endocannabinoids | - | Decrease [10] | - |
Withdrawal/Negative Affect Stage: The Role of the Extended Amygdala
The withdrawal/negative affect stage involves a negative emotional state (dysphoria, anxiety, irritability) when access to the drug or behavior is prevented. This stage primarily involves the extended amygdala, which becomes increasingly sensitive with repeated engagement [4].
During this stage, decreased function of the dopamine reward system combines with recruitment of brain stress neurotransmitters, including CRF and dynorphin, in the extended amygdala neurocircuitry [10]. This creates a powerful negative reinforcement mechanism, where the addicted individual seeks the substance or behavior not primarily for pleasure but to obtain temporary relief from this discomfort [4]. Both substance and behavioral addictions demonstrate similar activation patterns in the extended amygdala during withdrawal states [75] [76].
Preoccupation/Anticipation Stage: Executive Function Dysregulation
The preoccupation/anticipation (craving) stage involves seeking the substance or behavior again after a period of abstinence, engaging a distributed network including the prefrontal cortex, orbitofrontal cortex, dorsal striatum, basolateral amygdala, hippocampus, and insula [11]. This stage represents the complex interplay between craving and disrupted inhibitory control.
The prefrontal cortex is critical for executive function—organizing thoughts, prioritizing tasks, managing time, and making decisions—including exerting control over engagement with addictive substances or behaviors [4]. In addiction, this region becomes dysregulated, resulting in compromised executive function that manifests as impaired decision-making and reduced impulse control [10]. The shift in balance between prefrontal cortical circuits and those of the basal ganglia and extended amygdala makes an individual seek the drug or behavior compulsively [4].
Neuroplasticity Mechanisms: Molecular and Cellular Adaptations
Synaptic and Neurochemical Plasticity
Repeated exposure to both drugs of abuse and non-drug rewards induces neuroplastic adaptations in addiction-related circuits. These changes range from altered neurotransmitter levels to modified cell morphology and changes in transcriptional activity [75] [76].
The incentive-sensitization theory posits that repeated exposure leads to sensitization of the incentive-motivational properties of drugs and drug-related cues, mediated by sensitized nucleus accumbens dopamine release upon exposure to these cues [75] [76]. This mechanism appears to translate between drug and non-drug rewards, as evidenced by dopamine dysregulation syndrome in patients taking dopaminergic medications, characterized by compulsive engagement in non-drug rewards such as gambling, shopping, or sex [75].
The opponent process theory provides another framework, suggesting two processes engaged during repeated experiences: an initial affective or hedonic response followed by an opposing process that counteracts this initial state [75] [76]. With repeated exposure, the opposing process strengthens, contributing to the negative emotional state during withdrawal.
Circuit-Level Reorganization
The transition to addiction involves neuroplasticity across all key addiction structures, beginning with changes in the mesolimbic dopamine system and progressing through a cascade of neuroadaptations from the ventral striatum to dorsal striatum and orbitofrontal cortex, eventually dysregulating the prefrontal cortex, cingulate gyrus, and extended amygdala [11]. This progression helps explain the shift from voluntary, controlled engagement to compulsive behavior that characterizes both substance and behavioral addictions.
Figure 2: Neuroplasticity Cascade in Addiction Development
Experimental Approaches and Methodologies
Research Models and Paradigms
The study of addiction neurocircuitry employs diverse experimental approaches, including animal models, human laboratory studies, and neuroimaging techniques. Animal models permit investigations of specific signs or symptoms associated with the psychopathological condition, particularly the transition from controlled engagement to addiction [10].
Recent models incorporate individual and strain diversity in responses, complex environments with access to alternative reinforcers, and effects of stressful stimuli, allowing investigation of neurobiological processes underlying addiction risk and environmental factors providing resilience [10]. These models also explore developmental stage and sex influences, recognizing the greater vulnerability during adolescence and distinct trajectories in men and women [10].
The Scientist's Toolkit: Key Research Reagents and Methodologies
Table 3: Essential Research Tools for Addiction Circuitry Studies
| Research Tool/Methodology | Primary Application | Technical Function |
|---|---|---|
| Functional Magnetic Resonance Imaging (fMRI) | Human neuroimaging studies | Measures brain activity by detecting changes associated with blood flow; identifies regions activated during craving, intoxication, or withdrawal states [75] |
| Positron Emission Tomography (PET) | Human neuropharmacology studies | Uses radioligands to measure neurotransmitter release, receptor occupancy, and metabolic activity; documents dopamine release in ventral striatum [10] |
| Optogenetics | Animal circuit manipulation | Uses light to control neurons genetically modified to express light-sensitive ion channels; establishes causal relationships between specific circuits and behaviors [77] |
| Fast-Scan Cyclic Voltammetry | Animal neurotransmitter dynamics | Measures real-time neurotransmitter release (particularly dopamine) with high temporal resolution; characterizes phasic dopamine signaling during reward processing [75] |
| Electrophysiological Recordings | Animal synaptic plasticity studies | Measures changes in neuronal excitability and synaptic transmission; quantifies alterations in AMPA/NMDA ratio following drug or reward exposure [75] |
| Molecular Biological Assays | Animal and postmortem human studies | Measures changes in gene expression, protein levels, and epigenetic modifications; identifies transcriptional factors mediating addiction vulnerability [10] |
Implications for Therapeutic Development
Understanding the shared neurocircuitry between drug and behavioral addictions enables the development of targeted interventions. The brain disease model of addiction has contributed significantly to current views of substance use disorders, with understanding of neurobiological changes enabling development of novel intervention and prevention methods [4].
Identification of neural systems and structures changed by both drug and non-drug reward seeking allows for development of different medication and non-medication-based treatments targeting these vulnerable brain areas [4]. Research on genetics and epigenetics has helped establish biomarkers that directly or indirectly affect neurobiological processes involved in developing addictions, refining potential treatments and prevention strategies [4].
Drug and behavioral addictions share fundamental neural substrates centered on the basal ganglia, extended amygdala, and prefrontal cortex. These regions form dynamic networks that undergo specific neuroadaptations across the binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation stages of the addiction cycle. While drugs of abuse and natural rewards initially engage these circuits through different mechanisms, the resulting neuroplasticity reveals remarkable convergence, particularly in the transition from controlled to compulsive engagement.
Future research should continue to elucidate the precise molecular and cellular adaptations within these circuits, with particular emphasis on individual differences in vulnerability and resilience. This comparative circuitry approach promises to yield novel therapeutic strategies that target shared mechanisms across addiction subtypes, ultimately advancing treatment for both substance and behavioral addictions.
Addiction, now clinically termed Substance Use Disorder (SUD), is defined by the American Society of Addiction Medicine (ASAM) as a treatable, chronic medical disease involving complex interactions among brain circuits, genetics, the environment, and an individual's life experiences [78]. This whitepaper delineates the validation of this definition through the well-established chronic disease analogy with Type 2 Diabetes Mellitus (T2DM) and Hypertension, framing this comparison within the context of modern neuroscience research on the basal ganglia, extended amygdala, and prefrontal cortex. For the research and drug development community, we present quantitative epidemiological data, detailed experimental protocols for validating this analogy, and essential research toolkits to advance the discovery of novel therapeutic interventions.
Neurocircuitry of Addiction: The Tripartite Cycle
The brain disease model of addiction conceptualizes it as a chronic relapsing disorder characterized by a three-stage cycle—binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation (craving). Each stage is mediated by specific neurocircuits [11] [4].
Stage 1: Binge/Intoxication. This stage is centered on the basal ganglia, particularly the ventral striatum (including the nucleus accumbens), a key node of the brain's "reward circuit." Drugs of abuse produce intense euphoria by triggering massive surges of neurotransmitters, especially dopamine, far exceeding those produced by natural rewards. This over-activation "teaches" the brain to repeat the behavior, powerfully reinforcing the drug-taking habit. With repeated exposure, this circuit adapts, diminishing its sensitivity and reducing the individual's ability to feel pleasure from anything but the drug [7] [4].
Stage 2: Withdrawal/Negative Affect. The extended amygdala is the central structure in this stage. When drug use ceases, the extended amygdala becomes hyperactive, leading to a negative emotional state—dysphoria, anxiety, irritability, and stress—that characterizes withdrawal. This creates a powerful motivational drive to seek the drug again not to get high, but to find temporary relief from this discomfort. This circuit becomes increasingly sensitive with continued drug use [11] [4].
Stage 3: Preoccupation/Anticipation (Craving). This stage involves a widespread network with a key role for the prefrontal cortex. The prefrontal cortex is responsible for executive functions: organizing thoughts, prioritizing tasks, decision-making, and exerting self-control over impulses. In addiction, this region becomes dysregulated. The balance shifts between the prefrontal cortex (impulse control) and the circuits of the basal ganglia (habit) and extended amygdala (negative affect), leading to compulsive drug seeking with reduced inhibitory control. Other areas, like the orbitofrontal cortex, dorsal striatum, hippocampus, and insula, are also involved in craving and the learned "reflex" of drug-seeking triggered by environmental cues [7] [11].
The transition to addiction involves neuroplasticity across all these structures, progressing from changes in the mesolimbic dopamine system to a cascade of neuroadaptations that dysregulate prefrontal control and heighten the reactivity of the extended amygdala [11].
Quantitative Evidence: Epidemiological Overlap of SUD, Diabetes, and Hypertension
Robust epidemiological data demonstrate that SUD significantly increases the risk for and exacerbates the complications of other chronic conditions like T2DM and Hypertension, underscoring shared pathophysiological and healthcare management challenges.
Table 1: Risk of Incident Hypertension and Diabetes Associated with Alcohol Use Disorder (AUD)
| Outcome | AUD Cohort Incidence (per 1,000 person-years) | Non-AUD Cohort Incidence (per 1,000 person-years) | Adjusted Hazard Ratio (aHR) | 95% Confidence Interval |
|---|---|---|---|---|
| Hypertension | 35.1 | 20.7 | 1.72 | 1.68 - 1.76 |
| Diabetes | 20.2 | 9.36 | 2.18 | 2.11 - 2.24 |
| Both Diabetes & Hypertension | 10.3 | 5.38 | 2.02 | 1.94 - 2.10 |
Source: Taiwan insurance claims data follow-up study (N=60,590 per cohort) [79].
Table 2: Risk of T2DM Complications in Patients with Co-occurring T2DM, Hypertension, and SUD
| Substance Use Disorder (SUD) | Cerebrovascular Accident (aOR) | Diabetic Neuropathy (aOR) | Diabetic Renal Disease (aOR) | Myocardial Infarction (aOR) | All-Cause Mortality (aOR) |
|---|---|---|---|---|---|
| Tobacco Use Disorder (TUD) | 1.79 | 1.47 | 1.25 | 1.96 | 1.15 |
| Opioid Use Disorder (OUD) | 1.94 | NS | 1.34 | 2.01 | NS |
| Cocaine Use Disorder | 2.67 | 1.35 | NS | 2.68 | 1.61 |
| Cannabis Use Disorder (CUD) | NS | NS | NS | 2.48 | 1.49 |
| Alcohol Use Disorder (AUD) | NS | 1.27 | NS | 1.42 | 1.35 |
Source: Analysis of IBM Watson Health Explorys EHR data; NS = Not Significant [80].
Furthermore, patients with SUD face significant disparities in diagnosis. A cross-sectional study found that patients with SUD had 1.81 times greater odds of undiagnosed hypertension and 1.93 times greater odds of undiagnosed diabetes compared to those without SUD, highlighting critical gaps in the quality of primary care they receive [81].
Experimental Protocols: Validating the Analogy through EHR Data Analysis
The following provides a detailed methodology for conducting retrospective cohort studies to investigate the association between SUD and chronic medical conditions like T2DM and hypertension, based on established protocols from recent literature.
Protocol: Retrospective Cohort Study Using Electronic Health Records (EHR)
Objective: To assess the risk of developing hypertension and/or diabetes in a cohort with a diagnosed Substance Use Disorder compared to a matched control cohort without SUD.
1. Data Source and Setting:
- Utilize a large-scale, integrated EHR platform (e.g., IBM Watson Health Explorys or similar) that aggregates data from multiple healthcare organizations, including inpatient, outpatient, and billing systems [80].
- Ensure the dataset is de-identified and that the study is approved by an Institutional Review Board (IRB) with a waiver of informed consent due to the use of pre-existing, non-identified data [80].
2. Study Population and Cohort Definition:
- Inclusion Criteria: Identify adult patients (≥18 years) with at least one encounter diagnosis code for T2DM and hypertension. Use ICD-9-CM and ICD-10-CM codes for accurate phenotyping (e.g., ICD-9: 250.* for diabetes; 401-405 for hypertension) [80] [79].
- SUD Cohort: Identify patients with at least one encounter diagnosis code for abuse or dependence (e.g., ICD-9: 291, 303, 305.0; ICD-10: F10.1-F10.2) for specific substances: Tobacco, Opioids, Cocaine, Cannabis, and Alcohol. Use only abuse/dependence codes to define the SUD group, aligning with DSM-5 criteria [80].
- Comparison Cohort: For each SUD patient, select one or more controls from the general patient population without a diagnosis of the target SUD. Use propensity score matching to minimize confounding. Key matching variables should include:
3. Outcome Measures:
- The primary outcomes are the first recorded diagnosis of hypertension and/or diabetes after the index date (first SUD diagnosis for the SUD cohort, or a matched date for controls).
- Ensure the outcome diagnosis occurs after the index date for T2DM, hypertension, and SUD to establish temporality. Patients with outcome diagnoses prior to these dates should be excluded or coded as "missing" for that outcome [80].
- Undiagnosed cases can be defined using clinical values without diagnosis codes (e.g., ≥2 BP readings ≥140/90 without a hypertension diagnosis; HbA1c ≥6.5% without a diabetes diagnosis) [81].
4. Statistical Analysis:
- Calculate incidence rates for each outcome per 1,000 person-years of follow-up.
- Use Cox proportional hazards regression to estimate hazard ratios (HRs) and 95% confidence intervals (CIs) for the risk of each outcome in the SUD cohort compared to the non-SUD cohort.
- Perform multivariate analysis adjusting for any residual confounding factors after matching (e.g., CCI score, specific co-morbidities) to generate adjusted HRs (aHRs) [80] [79].
- Use the Kaplan-Meier method to plot cumulative incidence curves and the log-rank test to assess differences between cohorts [79].
Table 3: Essential Research Reagents and Resources for Addiction Neurobiology
| Resource/Reagent | Function/Application | Specific Example / Notes |
|---|---|---|
| Animal Models | Study behavioral stages of addiction (self-administration, conditioned place preference) and neuroplasticity. | Rodent models with extended access to drugs to observe escalation of intake and compulsive seeking [11]. |
| Human Brain Imaging (fMRI, PET) | Map functional and neurochemical changes in human brain circuits in vivo. | Used to identify hypoactivity in the prefrontal cortex and hyperactivity in the extended amygdala in individuals with SUD [11] [4]. |
| Genome-Wide Association Studies (GWAS) | Identify specific genetic loci and variants associated with SUD vulnerability. | Identified a locus on chromosome 8 controlling CHRNA2 expression linked to Cannabis Use Disorder [4]. |
| ICD-9/10-CM Code Sets | Phenotyping patients in EHR-based research; ensuring accurate cohort definition. | ICD-9: 291, 303, 305.0; ICD-10: F10.1-F10.2 for Alcohol Use Disorder. Critical for reproducible retrospective studies [80] [79]. |
| Propensity Score Matching Algorithms | Reduce confounding in observational studies by creating balanced comparison groups. | SAS macros using Mahalanobis distance within propensity score calipers (e.g., Ohio Valley Node programs) [80]. |
| Charlson Comorbidity Index (CCI) | Quantify patient burden of co-existing diseases for risk adjustment in clinical studies. | Weighted index of 19 conditions; predicts risk of mortality and resource use [79]. |
The evidence validating addiction as a chronic medical illness analogous to diabetes and hypertension is compelling and multi-faceted. It rests on a foundation of shared chronicity, high relapse rates (40-60% for addiction, comparable to 30-70% for diabetes and hypertension), and strong genetic and environmental determinants [82]. The critical differentiator is the neurobiological basis of addiction, rooted in the dysfunctional plasticity of the basal ganglia, extended amygdala, and prefrontal cortex.
For researchers and drug development professionals, this analogy provides a powerful framework. It mandates a shift from seeking ephemeral "cures" to developing long-term management strategies that target specific stages of the addiction cycle and specific underlying neurocircuits. Future efforts must focus on:
- Circuit-Targeted Therapeutics: Designing small molecules, biologics, and neuromodulation technologies that specifically normalize function in the reward, stress, and executive control circuits.
- Precision Medicine: Utilizing genetic (e.g., GWAS-identified risk alleles like CHRNA2) and neuroimaging biomarkers to stratify patients and predict treatment response [4].
- Integrated Care Models: Acknowledging and addressing the high comorbidity between SUD and other chronic conditions like T2DM and hypertension in both clinical trial design and real-world treatment delivery.
Embracing this chronic disease model is essential for reducing stigma, guiding public policy, and ultimately, for discovering the next generation of effective treatments for substance use disorders.
Adolescence is a critical neurodevelopmental period characterized by the maturation of neural circuits that govern reward, emotion, and executive control. This phase entails a dynamic interplay between the prefrontal cortex (PFC), basal ganglia, and extended amygdala, rendering adolescents uniquely susceptible to circuit dysregulation and the onset of substance use disorders (SUDs) [83] [3]. This whitepaper synthesizes current research on the neurobiological mechanisms underlying adolescent vulnerability, providing a technical guide for researchers and drug development professionals. We present quantitative data, experimental methodologies, and visual tools to advance the study of addiction-related neural circuitry.
Neurodevelopmental Basis of Adolescent Vulnerability
The adolescent brain undergoes protracted structural and functional changes, creating a mismatch between early-maturing limbic systems (e.g., amygdala) and late-maturing prefrontal regulatory regions [83]. Key processes include:
- Prefrontal Cortex Maturation: The PFC, involved in executive functions like impulse control and decision-making, is the last brain region to mature, with synaptic pruning and myelination continuing into the mid-20s [83] [7]. This delayed maturation limits top-down regulation of emotion and reward-seeking behaviors.
- Limbic System Hyperreactivity: Subcortical regions, including the amygdala and ventral striatum (a key component of the basal ganglia), exhibit heightened reactivity to emotional and rewarding stimuli [83] [84]. This imbalance predisposes adolescents to risk-taking and emotional dysregulation.
- Network-Level Changes: Functional connectivity between the PFC and limbic regions is refined during adolescence, and disruptions in this connectivity are linked to psychopathology and SUDs [85] [86].
Key Neural Circuits in Addiction and Adolescent Vulnerability
Addiction involves a three-stage cycle—binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation—each mediated by specific neural circuits [3] [7]. The table below summarizes the roles of these circuits and their adolescent-specific vulnerabilities.
Table 1: Neural Circuits in Addiction and Adolescent Vulnerabilities
| Brain Circuit | Primary Functions | Adolescent Vulnerabilities |
|---|---|---|
| Basal Ganglia | Reward processing, habit formation (via dopamine surges) [7]. | Hyperactivation to rewards (e.g., social, drug-related cues); heightened dopamine signaling [83] [7]. |
| Extended Amygdala | Stress response, negative affect (via corticotropin-releasing hormone) [3]. | Elevated stress reactivity; sensitization during withdrawal [3] [86]. |
| Prefrontal Cortex (PFC) | Executive control, impulse regulation [83]. | Immature connectivity to limbic regions; reduced inhibitory control [83] [7]. |
Quantitative Data on Adolescent vs. Adult Circuit Dysregulation
Studies using fMRI, EEG, and behavioral assays reveal distinct differences between adolescents and adults in circuit function. The table below consolidates key metrics from experimental research.
Table 2: Comparative Metrics of Circuit Dysregulation in Adolescence vs. Adulthood
| Metric | Adolescent Response | Adult Response | Experimental Method |
|---|---|---|---|
| Amygdala Reactivity | Elevated activation to negative stimuli [83] [86]. | Attenuated activation; greater PFC modulation [83]. | fMRI during emotional tasks [84]. |
| Dopamine Release | Exaggerated in ventral striatum to rewards [7]. | Moderate release; better regulation [7]. | PET imaging with reward cues [3]. |
| PFC-Limbic Connectivity | Weaker functional connectivity [86]. | Stronger anticorrelated activity [83]. | Resting-state fMRI [85] [86]. |
| Risk of Relapse | Higher relapse post-abstinence (e.g., 59–90% in year one) [87]. | Lower relapse rates with MAT [87]. | Clinical trials (e.g., MAT vs. abstinence) [87]. |
Experimental Protocols for Studying Circuit Dysregulation
fMRI-Based Functional Connectivity Analysis
Objective: Assess PFC-amygdala connectivity during emotion regulation. Workflow:
- Participant Recruitment: Adolescents (ages 12–17) and adults (25+), matched for demographics.
- Task Design: Use an emotional Go/No-Go paradigm with neutral and affective stimuli.
- Image Acquisition: Collect T2*-weighted BOLD signals at 3T (TR = 2 s, voxel size = 3 mm³). Preprocess data using AFNI: motion correction, spatial smoothing (6 mm FWHM), and bandpass filtering (0.01–0.1 Hz) [85].
- Analysis:
- Compute functional connectivity (seed-based: amygdala to PFC).
- Apply generalized linear models (GLMs) to compare group differences.
- Control for motion (Framewise Displacement < 0.5 mm) [85].
EEG-Based Emotion Dysregulation Monitoring
Objective: Identify neural correlates of emotional dysregulation using spectral analysis. Workflow:
- EEG Recording: 64-channel system during resting state and emotion-induction tasks.
- Feature Extraction: Calculate fractional amplitude of low-frequency fluctuations (fALFF) and regional homogeneity (ReHo) [84].
- Machine Learning: Train convolutional neural networks (CNNs) on EEG features to classify dysregulation states (accuracy: >87% in recent studies) [84].
- Validation: Cross-validate with self-report measures (e.g., Difficulties in Emotion Regulation Scale) [84].
Molecular Assays for Dopaminergic Signaling
Objective: Quantify dopamine receptor density and function in adolescent models. Workflow:
- Animal Models: Use adolescent rodents (e.g., postnatal days 28–42) for microdialysis in the nucleus accumbens.
- Pharmacological Probes: Administer dopamine agonists (e.g., quinpirole) and antagonists (e.g., haloperidol).
- Analysis: Measure dopamine via HPLC and receptor binding via autoradiography.
Diagram: Neural Circuit Interactions in Adolescent Vulnerability
Title: Circuit Dysregulation in Adolescence
The Scientist’s Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Neural Circuit Research
| Reagent/Tool | Function | Example Application |
|---|---|---|
| AFNI Software | fMRI data preprocessing and analysis [85]. | Motion correction, functional connectivity mapping [85]. |
| Kendall’s Coefficient of Concordance (KCC) | Measures local connectivity (ReHo) in fMRI [85]. | Assessing neural homogeneity in PFC networks [85]. |
| fALFF (fractional Amplitude of Low-Frequency Fluctuations) | Quantifies spontaneous neural activity in fMRI [85]. | Identifying resting-state biomarkers of dysregulation [85]. |
| Dopamine D2 Receptor Antagonists | Blocks dopamine signaling; tests reward circuit function. | Microdialysis in rodent models of addiction [7]. |
| Eigenvector Centrality Mapping | Graphs network hubs in fMRI data [85]. | Identifying key nodes in PFC-amygdala pathways [85]. |
Implications for Drug Development
- Targeting Circuit Imbalances: Medications that enhance PFC function (e.g., norepinephrine reuptake inhibitors) or dampen amygdala hyperactivity (e.g., CRF antagonists) show promise [3] [4].
- Personalized Interventions: Biomarkers like fALFF and ReHo can stratify patients for targeted therapies [85] [84].
- Neuroplasticity-Based Strategies: Cognitive training and neurofeedback may strengthen PFC-limbic connectivity [84] [88].
Adolescent brain development is marked by a vulnerable imbalance between reward-driven limbic circuits and immature regulatory systems. Understanding these mechanisms through integrated fMRI, EEG, and molecular protocols enables the development of circuit-specific treatments for SUDs. Future work should focus on longitudinal studies and machine learning models to predict individual risk trajectories.
The high prevalence of psychiatric comorbidity, where individuals meet diagnostic criteria for multiple mental disorders, suggests shared neurobiological origins across different psychopathologies [89]. Emerging evidence now supports the existence of a general psychopathology factor (the "p factor") that underlies higher vulnerability for various psychiatric conditions [89]. This framework fundamentally shifts our understanding of mental disorders from distinct categorical entities to manifestations of shared neural system dysregulations. The critical neural circuits implicated in this transdiagnostic model center on the basal ganglia, extended amygdala, and prefrontal cortex—the very same circuits identified in addiction research [7] [4] [18]. This convergence provides a compelling neurobiological basis for the frequent clinical observation of comorbidities among substance use, mood, anxiety, and impulse control disorders.
Recent large-scale neuroimaging studies have identified a neuropsychopathological (NP) factor across externalizing and internalizing symptoms using multitask connectomes [89] [90]. This NP factor represents a unified, genetically determined phenomenon characterized by delayed development of the prefrontal cortex that leads to poor executive function [89]. The identification of this shared neural basis bridges multidimensional evidence from behavioral, neuroimaging, and genetic substrates, offering new avenues for therapeutic interventions targeting psychiatric comorbidities rather than single disorders [89].
Core Shared Neurocircuitry
The Tripartite Neural Network Model
Decades of neuroscience research have consistently identified three key brain regions and their interconnected circuits that are dysregulated across multiple psychiatric disorders: the basal ganglia, extended amygdala, and prefrontal cortex. These regions form dynamic networks that coordinate motivation, emotional processing, and behavioral control [7] [4].
Table 1: Core Neural Circuits and Their Functions in Psychopathology
| Brain Region | Primary Functions | Role in Psychiatric Disorders |
|---|---|---|
| Basal Ganglia | Reward processing, habit formation, positive motivation [7] [4] | Overactive in binge/intoxication stage; mediates incentive salience [18] |
| Extended Amygdala | Stress response, negative affect, anxiety [7] [4] | Hyperactive in withdrawal/negative affect stage; "anti-reward" system [18] |
| Prefrontal Cortex | Executive function, impulse control, decision-making [7] [4] | Dysregulated in preoccupation/anticipation stage; executive dysfunction [18] |
Neural Circuit Interactions and Dysregulation
The relationship between these three core regions creates a delicate balance that becomes disrupted across multiple psychiatric disorders. The prefrontal cortex normally exerts top-down control over the basal ganglia and extended amygdala, but in states of psychopathology, this regulatory capacity is diminished [4]. Simultaneously, the extended amygdala becomes hypersensitive, amplifying stress and negative affect, while the basal ganglia drives compulsive reward-seeking behaviors [18]. This tripartite dysregulation creates a self-reinforcing cycle that manifests across diagnostic boundaries as impaired response control, emotional dysregulation, and compulsive behaviors.
The shared neurocircuitry model explains why comorbidities follow predictable patterns: the same neural systems are dysregulated across disorders. For instance, the overlap between addiction and anxiety disorders emerges from shared dysregulation of the extended amygdala, while comorbid impulse control and mood disorders reflect overlapping disruptions in prefrontal-basal ganglia circuits [91] [92].
Figure 1: Shared Neurocircuitry Dysregulation in Comorbid Psychiatric Disorders. The diagram illustrates the imbalanced interactions between the three core neural circuits, showing diminished prefrontal control with enhanced subcortical drive that characterizes multiple psychiatric disorders.
Quantitative Evidence from Neuroimaging Studies
The Neuropsychopathological (NP) Factor
Groundbreaking research using the large longitudinal IMAGEN cohort has quantitatively demonstrated a shared neural basis underlying psychiatric comorbidity [89]. This study defined an NP factor across externalizing and internalizing symptoms using multitask connectomes, revealing consistent neural patterns that transcend diagnostic categories.
Table 2: Neuropsychopathological Factor Correlations with Symptom Domains
| Symptom Domain | Specific Disorders | Correlation with NP Factor | Key Associated Neural Circuits |
|---|---|---|---|
| Externalizing | ADHD, Conduct Disorder, ODD | r = 0.31 with NP factor [89] | Prefrontal cortex, inhibitory control networks [89] |
| Internalizing | Specific Phobia, GAD, Eating Disorders | r = 0.23 with NP factor [89] | Extended amygdala, emotional reactivity circuits [89] |
| Cross-Disorder | Comorbid externalizing & internalizing | Significant cross-disorder edges in SST & MID tasks [89] | Consensus positive-positive edges (n=136) [89] |
The study identified that only specific task conditions—particularly the stop signal task (SST) and monetary incentive delay (MID) task—contained significantly more cross-disorder edges than random discoveries (stop success nedge = 325, stop failure nedge = 297, positive feedback nedge = 344; reward anticipation nedge = 316; all P < 0.001) [89]. These cross-disorder edges were stratified into three groups: positive-positive consensus edges (n = 136), negative-negative consensus edges (n = 64), and positive-negative dissensus edges (n = 1,032) [89]. The summed functional connectivity strength of the positive-positive consensus edges was significantly associated with both externalizing and internalizing symptoms simultaneously, forming the reproducible NP factor [89].
Neurocircuitry of Specific Disorders
Research examining specific disorders reveals consistent patterns of neural circuit dysregulation that align with the transdiagnostic model:
Anxiety Disorders: Multiple studies report heightened amygdala activation in response to disorder-relevant stimuli in PTSD, social phobia, and specific phobia [92]. The insular cortex shows heightened activation across many anxiety disorders, while PTSD specifically associates with diminished responsivity in the rostral anterior cingulate cortex and adjacent ventral medial prefrontal cortex [92].
Mood Disorders: The system linking the medial prefrontal cortex to the amygdala, ventral striatum, medial thalamus, hypothalamus, and periaqueductal gray is centrally involved in mood disorders [93]. These circuits form the core neural system that regulates emotional processing and shows consistent abnormalities across major depressive and bipolar disorders.
Trauma-Related Disorders: TBI and PTSD show similar impacts on prefrontal and subcortical connectivity and activation, particularly in circuitry subserving emotion regulation [91]. In the immediate aftermath of trauma, glucocorticoids stimulate excess glutamatergic activity in prefrontal cortex-subcortical circuitry, creating long-term system-level functional and structural changes common to both TBI and PTSD [91].
Experimental Approaches and Methodologies
Key Research Protocols
The identification of shared neurocircuitry relies on sophisticated experimental approaches that can map neural networks and their interactions across diagnostic groups.
Connectome-Based Predictive Modeling (CPM): This approach uses multivoxel pattern analysis of functional connectivity to predict individual differences in symptom severity [89]. The protocol involves:
- Data Acquisition: Collecting task-based fMRI data during multiple cognitive domains (e.g., reward processing, inhibitory control, emotional reactivity) [89]
- Feature Selection: Identifying univariately significant functional connectivity features correlated with symptom measures [89]
- Cross-Validation: Using k-fold cross-validation to build models in a training set and test them in held-out validation sets [89]
- Network Identification: Defining networks based on edges that consistently predict symptoms across cross-validation folds [89]
Longitudinal Neuroimaging Cohort Analysis: Large-scale studies like the IMAGEN cohort (N = 1,750 adolescents followed to young adulthood) enable tracking of neural developmental trajectories [89]. This involves:
- Multiwave Data Collection: Assessing participants at multiple time points (e.g., ages 14, 16, 19, 22) [89]
- Multimodal Imaging: Combining structural, functional, and diffusion MRI with genetic and behavioral assessments [89]
- Developmental Trajectory Modeling: Using growth curve models to map neural and behavioral development [89]
- Cross-Cohort Validation: Testing findings in independent samples (e.g., ABCD, HCP) and clinical datasets (e.g., ADHD-200) [89]
Figure 2: Experimental Workflow for Identifying Shared Neurocircuitry. The diagram outlines the key methodological phases in large-scale studies identifying transdiagnostic neural factors, from data collection through validation.
Table 3: Key Research Reagents and Resources for Neurocircuitry Studies
| Resource Category | Specific Examples | Research Application |
|---|---|---|
| Task-Based fMRI Paradigms | Stop Signal Task (SST), Monetary Incentive Delay (MID), Emotional Reactivity Tasks [89] | Mapping circuit-specific neural signatures of cognitive domains affected across disorders |
| Multimodal Imaging Protocols | Structural MRI, resting-state fMRI, diffusion tensor imaging, arterial spin labeling [89] | Comprehensive characterization of brain structure, function, and connectivity |
| Genetic Analysis Tools | Genome-wide association studies, polygenic risk scoring, epigenomic profiling [89] [4] | Identifying shared genetic vulnerabilities and gene-environment interactions |
| Computational Modeling Approaches | Connectome-based predictive modeling, graph theory analysis, machine learning classifiers [89] | Predicting individual differences in symptom severity and treatment response |
Implications for Drug Development and Future Research
Therapeutic Target Opportunities
The shared neurocircuitry model reveals promising new avenues for therapeutic development that target transdiagnostic mechanisms rather than disorder-specific symptoms:
Prefrontal Cortex Targets: Developing interventions that enhance prefrontal regulatory function could address executive dysfunction across multiple disorders [89] [18]. This includes approaches targeting dopaminergic, glutamatergic, and GABAergic systems in the PFC to improve cognitive control and decision-making [18].
Extended Amygdala Targets: Treatments that normalize stress system hyperactivity in the extended amygdala could alleviate negative affect across mood, anxiety, and substance disorders [18]. Promising targets include CRF antagonists, neurokinin antagonists, and neuropeptide Y enhancers [18].
Neuroinflammatory Mechanisms: Emerging evidence implicates neuroinflammation and oxidative stress in shaping neural circuitry involved in reward, motivation, and negative affect across disorders [94]. Targeting these processes may represent a novel approach for treating comorbidities.
Personalized Treatment Approaches
The Addictions Neuroclinical Assessment (ANA) exemplifies how understanding shared neurocircuitry can inform personalized treatment [18]. This clinical instrument translates the three neurobiological stages of addiction into three neurofunctional domains: incentive salience, negative emotionality, and executive dysfunction [18]. Similar approaches could be developed for broader psychiatric comorbidity, allowing clinicians to target specific circuit dysfunctions regardless of primary diagnosis.
Future research directions should include:
- Circuit-Based Taxonomy Development: Creating diagnostic systems based on neural circuit dysfunction rather than symptom clusters [89]
- Circuit-Targeted Interventions: Developing neuromodulation approaches (TMS, DBS) that target specific dysfunctional circuits across disorders [89]
- Developmental Timing Studies: Identifying critical periods for circuit development to optimize preventive interventions [89]
- Multi-Omic Integration: Combining genomics, transcriptomics, and proteomics with neuroimaging to comprehensively map biological pathways [94]
The recognition of shared neurocircuitry across comorbid psychiatric disorders represents a paradigm shift in neuroscience—from seeking unique neural signatures for each diagnostic category to identifying common pathways that cut across traditional diagnostic boundaries. This transdiagnostic approach promises more effective, targeted interventions for the complex patterns of comorbidity that routinely challenge clinical practice.
The brain disease model of addiction (BDMA) has dominated the scientific and clinical understanding of substance use disorders for nearly three decades. Originally articulated by Alan Leshner, then Director of the National Institute on Drug Abuse (NIDA), who asserted that "addiction is a brain disease, and it matters," this framework positioned addiction as a chronic, relapsing condition rooted in identifiable brain pathology [95]. The model was advanced with dual purposes: to provide an etiological theory based on neuroscientific discoveries and to reduce the profound stigma associated with addiction by reconceptualizing it from a moral failing to a medical condition [95] [96]. The BDMA has successfully guided research priorities worldwide, with NIDA funding an estimated 85% of global addiction research during Leshner's tenure [95].
This review examines the BDMA through a critical yet balanced scientific lens, with particular focus on its intersection with the established neurocircuitry of addiction. While the model has catalyzed significant research advances, particularly in elucidating the roles of the basal ganglia, extended amygdala, and prefrontal cortex, it faces substantial theoretical and empirical challenges [3] [11]. We evaluate these critiques while maintaining the perspective that neurobiological factors are fundamental components of addiction, and we provide a framework for integrating this knowledge with psychosocial and environmental determinants in future research.
The Evolving Neurocircuitry Framework of Addiction
Key Neural Networks in the Addiction Cycle
Research conducted within the BDMA framework has identified three primary brain regions and their associated circuits as critical to the development and maintenance of addiction. These circuits form a heuristic three-stage cycle—binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation—that becomes more severe with continued substance use [3] [11].
Table 1: Primary Brain Regions Implicated in Substance Use Disorders
| Brain Region | Key Functions in Addiction | Associated Stage |
|---|---|---|
| Basal Ganglia | Reward processing, habit formation, motivation | Binge/Intoxication |
| Extended Amygdala | Stress response, negative affect, anxiety | Withdrawal/Negative Affect |
| Prefrontal Cortex | Executive control, decision-making, impulse regulation | Preoccupation/Anticipation |
The basal ganglia, particularly the nucleus accumbens, play a crucial role in the rewarding effects of addictive substances and the formation of habitual substance-taking behaviors [3]. These regions are rich in dopamine neurons, which are activated by acute drug use, reinforcing substance-seeking behavior. With repeated use, neuroadaptations occur in these circuits, shifting drug use from a voluntary to a more habitual behavior.
The extended amygdala becomes engaged during the withdrawal/negative affect stage as substance use escalates. This region mediates stress responses and produces the dysphoria, anxiety, and irritability that characterize withdrawal, thereby motivating continued use to alleviate these negative states [11]. The recruitment of brain stress systems, including corticotropin-releasing factor (CRF), contributes to the negative reinforcement that drives addiction.
The prefrontal cortex, especially the orbitofrontal and medial prefrontal regions, is critical for executive function, including the ability to organize thoughts and activities, prioritize tasks, manage time, and make decisions [3]. In addiction, this region shows reduced activity and disrupted connectivity, impairing impulse control and decision-making capabilities while enhancing the salience of drug-related cues [11].
Neurobiological Transitions in Addiction
The transition from occasional, controlled substance use to chronic addiction involves profound neuroplastic changes across these interconnected circuits. Initial drug use primarily activates reward circuits in the ventral striatum (part of the basal ganglia), producing intense pleasure or euphoria. With repeated exposure, the dorsal striatum becomes increasingly involved, facilitating the development of automated habits and compulsive drug-seeking [11]. Concurrently, the extended amygdala undergoes adaptations that heighten sensitivity to stress, while prefrontal control mechanisms become progressively dysregulated, reducing the capacity for inhibitory control [3].
These transitions are supported by a cascade of molecular and cellular adaptations, including changes in gene expression, synaptic plasticity, and glutamatergic signaling. The net effect is a brain circuitry that is increasingly biased toward drug-seeking and drug-taking, even in the face of negative consequences [11].
Methodological Foundations and Research Tools
Key Experimental Approaches
Research elucidating the neurocircuitry of addiction has employed sophisticated methodological approaches in both animal models and human studies. Each approach offers distinct advantages and limitations for investigating different aspects of addiction.
Table 2: Key Methodological Approaches in Addiction Neuroscience
| Method | Key Applications | Strengths | Limitations |
|---|---|---|---|
| Animal Self-Administration | Study of drug-taking behavior, reinforcement, relapse | Controlled environment, mechanistic studies, genetic manipulation | Cannot capture full human subjective experience |
| Human Brain Imaging (fMRI, PET) | Identification of structural/functional brain changes in addiction | Non-invasive, direct observation of human brain | Correlation rather than causation, cost constraints |
| Genetic and Molecular Approaches | Investigation of genetic vulnerability, molecular adaptations | Precise mechanistic insights, potential therapeutic targets | Complex gene-environment interactions |
| Neuropsychological Testing | Assessment of cognitive deficits in addiction | Direct measure of functional impairment | May not reflect real-world decision-making |
Animal studies, particularly self-administration models, have allowed researchers to investigate addiction mechanisms under highly controlled conditions that would be unethical or impractical in humans [3]. These studies have been instrumental in mapping the specific contributions of different neural circuits to various stages of the addiction cycle.
Human neuroimaging studies using techniques such as functional magnetic resonance imaging (fMRI) and positron emission tomography (PET) have complemented animal work by allowing researchers to observe structural and functional changes in the living human brain [3]. These technologies have revealed that addicted individuals show characteristic patterns of brain activation in response to drug cues, stress, and decision-making tasks.
Essential Research Reagents and Tools
Table 3: Key Research Reagent Solutions in Addiction Neuroscience
| Reagent/Tool | Primary Function | Example Applications |
|---|---|---|
| Dopamine Receptor Ligands | Label and quantify dopamine receptors | PET imaging of D1/D2 receptor availability |
| CRF Antagonists | Block corticotropin-releasing factor receptors | Study stress-induced reinstatement of drug-seeking |
| AMPA/NMDA Receptor Modulators | Manipulate glutamatergic transmission | Investigate synaptic plasticity in reward circuits |
| DREADDs (Designer Receptors) | Chemogenetically control specific neuronal populations | Circuit-specific manipulation of addiction behaviors |
| Calcium Indicators (e.g., GCaMP) | Monitor neuronal activity in real-time | Record neural ensemble dynamics during behavior |
The experimental tools and reagents listed in Table 3 have been fundamental to advancing our understanding of addiction neurobiology. For instance, dopamine receptor ligands used in PET imaging have revealed reduced D2 receptor availability in the striatum of addicted individuals, which correlates with reduced prefrontal metabolism and impaired executive function [11]. Similarly, CRF antagonists have been instrumental in demonstrating the role of stress systems in relapse, showing that blocking CRF receptors can attenuate stress-induced reinstatement of drug-seeking in animal models [11].
Core Critiques of the Brain Disease Model
Conceptual and Definitional Ambiguities
A fundamental critique of the BDMA centers on the absence of a clear, consistent definition of what constitutes a "brain disease" in the context of addiction [95]. Philosophical analysis reveals that both narrow and broad views of brain disease coexist and inform addiction research, often implicitly and inconsistently.
The narrow view posits that a mental condition qualifies as a brain disease only if it manifests similarly to paradigmatic brain diseases, such as neurosyphilis or Alzheimer's disease, resulting from known or unknown structural and functional damage [95]. By this standard, addiction faces significant empirical challenges, as no diagnostic or prognostic biomarkers have been identified despite decades of research [95].
The broad view suggests that brain disease status should be granted automatically to mental disorders, as all mental activity resides in the brain [95]. This perspective risks becoming tautological—if all psychopathology is by definition brain pathology, the designation loses its distinctive meaning and utility.
The conceptual ambiguity is further complicated by what critics describe as frequent recourse to concepts with divergent or shifting meanings, which has obstructed productive debate and coherent advance in knowledge [95].
Limitations in Empirical Support
Despite many studies finding neurobiological differences between people with substance use disorders and healthy controls, the empirical evidence supporting the BDMA remains incomplete in several key areas:
- Lack of biomarkers: No diagnostic or prognostic biomarkers have been identified for addiction, despite the BDMA's premise that addiction reflects specific neuropathology [95].
- Limited treatment translation: The BDMA has yet to lead to significantly better or more precisely targeted treatments for most substance use disorders [95]. Most SUDs, including cannabis, stimulant, benzodiazepine, and new psychoactive substance use disorders, still lack FDA-approved medications [95].
- Questionable specificity: The brain changes observed in addiction share similarities with those found in other conditions involving compulsive behavior, raising questions about whether they represent a specific "disease state" rather than general neural adaptations to repeated behavior [95].
Inadequate Account of Recovery and Heterogeneity
Critics note that the BDMA struggles to account for the substantial heterogeneity in addiction trajectories and recovery pathways [96]. Numerous epidemiological studies have documented that many people overcome substance use problems without formal treatment, a phenomenon often termed "natural recovery" or "spontaneous remission" [96].
This pattern appears inconsistent with the characterization of addiction as a chronic, relapsing brain disease that necessarily requires professional intervention. Proponents of the BDMA counter that these criticisms often fail to distinguish between mere cessation of use and true recovery of function, and that the existence of developmental-limited or less severe forms does not negate the reality of severe, chronic cases [96].
Overshadowing of Psychosocial Factors
Perhaps the most significant critique is that by centering on individual neurobiological vulnerability, the BDMA has obscured fundamental psychosocial causes of substance use disorders [95]. These social and environmental factors—including poverty, unemployment, housing instability, discrimination, low educational attainment, and poor access to health care—have been referred to as the "causes of the causes" in literature on addiction development and maintenance [95].
The emphasis on brain mechanisms may inadvertently divert attention and resources away from social, economic, and public health interventions that could potentially have greater population-level impact [95]. This limitation reflects broader debates in mental health sciences about complex, multifactorial causation and the relative contributions of biological, psychological, and social factors [95].
The BDMA and Stigma: Unfulfilled Promises
A primary justification for the BDMA was that reconceptualizing addiction as a brain disease would reduce the stigma associated with what was previously viewed as moral failure or character weakness [95] [96]. However, empirical evidence suggests that this promise has not been fully realized.
Studies indicate that the use of the BDMA has had little effect on reducing stigma associated with substance use disorders [95]. In some cases, it may have promoted new sources of stigma related to reduced perceived agency and pessimism about recovery [95] [3]. This is consistent with research on other conditions showing that disease labels can be highly stigmatizing when they are perceived as indicating permanent, dangerous, or unpredictable characteristics [95].
The limited impact on stigma may reflect several factors, including oversimplified public communication of the model, persistent cultural beliefs about personal responsibility, and the complex nature of stigma itself, which may not be readily addressed by biological redefinition alone [95].
Quantitative Research Limitations in Addiction Science
Research supporting the BDMA relies heavily on quantitative methodologies, which present specific limitations for understanding a phenomenon as complex and multifaceted as addiction.
Table 4: Limitations of Quantitative Research in Addiction Science
| Limitation | Impact on BDMA Research |
|---|---|
| Lacks depth and context | Neuroimaging findings may show statistical differences but lack explanatory context about meaning or mechanisms |
| Limited by predefined questions | Structured approaches may miss emergent or unexpected aspects of addiction experience |
| Difficulty capturing decision-making processes | Reduced ability to understand how drug-related decisions are made in real-world contexts |
| Requires large samples for reliable insights | Challenges in studying severe addiction cases where population bases may be small |
| Expensive and time-consuming | Limits the scope and scale of neurobiological studies, particularly longitudinal designs |
Quantitative approaches, while valuable for identifying statistical patterns and group differences, struggle to capture the subjective experiences, meanings, and contextual factors that shape addiction and recovery [97] [98]. This limitation is particularly relevant given the substantial individual heterogeneity in addiction presentations and trajectories.
Furthermore, quantitative methods in addiction research face challenges related to improper representation of target populations, inability to fully control the research environment, and difficulty in data analysis, particularly for researchers from non-statistical backgrounds [98]. These methodological constraints highlight the need for complementary qualitative approaches to provide a more comprehensive understanding of addiction.
Toward an Integrated Framework: Future Directions
Moving beyond the current limitations of the BDMA requires acknowledging points of convergence among both proponents and critics. There is general agreement on rejecting moralist views of addiction, combating stigma, and recognizing that both neurobiological and psychosocial factors play key roles in addiction development and maintenance [95].
Future research should aim for greater integration across multiple levels of analysis, recognizing that the brain is the biological substrate from which both addiction and the capacity for behavior change arise [96]. This consilience approach would include:
- Intensified neuroscientific study of recovery: Shifting focus from pathology alone to include neurobiological mechanisms of natural recovery and treatment response [96].
- Development of more nuanced models: Recognizing that addiction exists on a continuum of severity and that different explanatory models may apply to different stages or subtypes [95] [96].
- Greater attention to environmental influences: Investigating how social, economic, and environmental factors interact with neurobiological vulnerabilities across the lifespan [95].
- Improved translation between basic and clinical research: Ensuring that neuroscientific discoveries more effectively inform prevention and treatment efforts [95].
Visualizing the Integrated Neurocircuitry of Addiction
The following diagram illustrates the key brain regions and their interactions in the addiction cycle, integrating the neurobiological perspective with critical environmental influences:
Addiction Neurocircuitry and Environmental Interactions
This diagram represents the interconnected neural circuits central to addiction, highlighting the roles of the basal ganglia (reward and habit), extended amygdala (stress and negative affect), and prefrontal cortex (executive control). Crucially, it incorporates environmental factors as modulatory influences on all three neural systems, acknowledging the importance of psychosocial context in the development and maintenance of addictive disorders.
The brain disease model of addiction has made substantial contributions to our understanding of substance use disorders, particularly in elucidating the roles of the basal ganglia, extended amygdala, and prefrontal cortex. However, the model faces significant critiques regarding its conceptual clarity, empirical support, ability to explain recovery heterogeneity, and attention to psychosocial determinants.
A balanced scientific perspective acknowledges the neurobiological foundations of addiction while recognizing that a comprehensive understanding requires integration across multiple levels of analysis—from molecular and cellular mechanisms to individual psychology and sociocultural context. Future research should build on the genuine insights provided by the BDMA while addressing its limitations through more nuanced, integrative approaches that do not reduce addiction solely to brain pathology but understand the brain as the biological substrate through which multiple causal factors operate.
Conclusion
The neural circuitry of addiction, centered on the basal ganglia, extended amygdala, and prefrontal cortex, provides a robust and empirically validated framework for understanding addiction as a chronic brain disorder. The three-stage cycle elucidates the progressive neuroadaptations that drive the transition from impulsivity to compulsivity, underpinned by specific molecular and cellular changes within these circuits. This neurocircuitry analysis not only validates addiction as a medical condition but also opens precise, circuit-based avenues for therapeutic intervention. Future research must focus on developing stage-specific treatments that target the unique dysregulations in incentive salience, negative reinforcement, and executive control. The translation of these findings into clinical practice, including medications that restore allostatic balance and neuromodulation therapies that reset circuit function, holds the promise of fundamentally improving outcomes for this devastating disorder.
References
- 1. of Neurocircuitry addiction [https://pubmed.ncbi.nlm.nih.gov/19710631/]
- 2. Review Neurobiology of addiction: a neurocircuitry analysis [https://www.sciencedirect.com/science/article/abs/pii/S2215036616001048]
- 3. THE NEUROBIOLOGY OF SUBSTANCE USE, MISUSE, AND ... [https://www.ncbi.nlm.nih.gov/books/NBK424849/]
- 4. The Brain Disease Model of Addiction [https://www.hazeldenbettyford.org/research-studies/addiction-research/brain-disease-model]
- 5. mechanisms and related clinical treatment of... Neurobiological [https://pubmed.ncbi.nlm.nih.gov/38665277/]
- 6. 1.5 Neurobiology of Substance Use Disorders – Community and... [https://pressbooks.txst.edu/nursingcph/chapter/1-5-neurobiology-of-substance-use-disorders/]
- 7. Drugs, Brains, and Behavior: The Science of Addiction - NIDA [https://nida.nih.gov/publications/drugs-brains-behavior-science-addiction/drugs-brain]
- 8. 14.4 Neurobiology of Addiction - Introduction to... | OpenStax [https://openstax.org/books/introduction-behavioral-neuroscience/pages/14-4-neurobiology-of-addiction]
- 9. Addictions and risk behaviors in adolescence: a systematic ... [https://www.frontiersin.org/journals/psychology/articles/10.3389/fpsyg.2025.1646746/full]
- 10. Neurobiology of addiction : a neurocircuitry analysis - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6135092/]
- 11. Neurocircuitry of Addiction | Neuropsychopharmacology [https://www.nature.com/articles/npp2009110]
- 12. GLP-1s show promise in treating alcohol and drug addiction [https://www.endocrine.org/news-and-advocacy/news-room/2025/glp1s-show-promise-in-treating-alcohol-and-drug-addiction]
- 13. UT San Antonio researchers develop safer, non-opioid ... [https://news.utsa.edu/2025/09/ut-san-antonio-researchers-are-developing-safer-non-opioid-alternatives-for-pain-relief/]
- 14. Neuroadaptive mechanisms of addiction: studies on the ... [https://www.sciencedirect.com/science/article/abs/pii/S0924977X03001780]
- 15. The Brain in Recovery [https://www.recoveryanswers.org/recovery-101/brain-in-recovery/]
- 16. Dysfunction of the prefrontal cortex in addiction [https://pmc.ncbi.nlm.nih.gov/articles/PMC3462342/]
- 17. Striatonigrostriatal Spirals in Addiction [https://www.frontiersin.org/journals/neural-circuits/articles/10.3389/fncir.2021.803501/full]
- 18. Neurobiology of Addiction - StatPearls - NCBI Bookshelf [https://www.ncbi.nlm.nih.gov/books/NBK597351/]
- 19. The Addiction Cycle Part III: Preoccupation/Anticipation Stage [https://www.aquilarecovery.com/blog/the-addiction-cycle-part-iii-preoccupation-anticipation-stage/]
- 20. Memory and Addiction: Shared Neural Circuitry and ... [https://www.sciencedirect.com/science/article/pii/S0896627304006051]
- 21. Addiction and the brain: the role of neurotransmitters in the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC80880/]
- 22. homeostasis and dopamine Glutamate : implications for... signaling [https://pmc.ncbi.nlm.nih.gov/articles/PMC8489281/]
- 23. Dynorphin–Still an Extraordinarily Potent Opioid Peptide [https://www.sciencedirect.com/science/article/abs/pii/S0026895X24036769]
- 24. Endogenous Opioid Dynorphin Is a Potential Link between ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8978570/]
- 25. Role of Corticotropin-Releasing Factor in Drug Addiction [https://pmc.ncbi.nlm.nih.gov/articles/PMC3273042/]
- 26. Glutamatergic systems and memory mechanisms underlying opioid... [https://research.vu.nl/en/publications/glutamatergic-systems-and-memory-mechanisms-underlying-opioid-add/]
- 27. Animal Models of Substance Abuse and Addiction [https://pmc.ncbi.nlm.nih.gov/articles/PMC2890392/]
- 28. Understanding Addiction Using Animal Models - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6895146/]
- 29. Shaping vulnerability to addiction – the contribution of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5708151/]
- 30. From concepts to treatment: a dialog between a preclinical ... [https://www.nature.com/articles/s41398-022-02177-5]
- 31. Animal models in addiction research: A dimensional ... [https://www.sciencedirect.com/science/article/abs/pii/S0149763418301428]
- 32. Neurobiological intersections of stress and substance use ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2025.1548372/full]
- 33. Decoding Encoded Cravings: Epigenetic Drivers of Addiction [https://pmc.ncbi.nlm.nih.gov/articles/PMC12467654/]
- 34. Drug addiction and treatment: An epigenetic perspective [https://www.sciencedirect.com/science/article/pii/S0753332223017493]
- 35. DNA Epigenetics in Addiction Susceptibility [https://www.frontiersin.org/journals/genetics/articles/10.3389/fgene.2022.806685/full]
- 36. Neural Circuit Mapping and Neurotherapy-Based Strategies [https://pmc.ncbi.nlm.nih.gov/articles/PMC12297084/]
- 37. Distinct Amygdala Neuronal Populations Control Opioid Use ... [https://pubmed.ncbi.nlm.nih.gov/40935226/]
- 38. Advanced Brain Circuit-Mapping Technique Reveals New ... [https://news.weill.cornell.edu/news/2025/01/advanced-brain-circuit-mapping-technique-reveals-new-anxiety-drug-target]
- 39. Application of omics-based biomarkers in substance use ... [https://www.sciencedirect.com/science/article/pii/S2950162823000085]
- 40. Dimensional Approaches to Understanding Addiction - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6233300/]
- 41. Electrophysiology and functional MRI of cue craving [https://www.sciencedirect.com/science/article/pii/S2667174325001764]
- 42. Integrating Optogenetic and Pharmacological Approaches ... [https://www.sciencedirect.com/science/article/abs/pii/S0031699724010378]
- 43. Medial prefrontal cortex circuit dynamics involved in stage- ... [https://pubmed.ncbi.nlm.nih.gov/41307570/]
- 44. Brain circuit identified as key to repetitive and compulsive ... [https://www.news-medical.net/news/20251122/Brain-circuit-identified-as-key-to-repetitive-and-compulsive-behaviors-in-mice.aspx]
- 45. An extended amygdala-midbrain circuit controlling cocaine ... [https://www.sciencedirect.com/science/article/pii/S2211124722005393]
- 46. Circuit dysfunctions of associative and sensorimotor basal ... [https://www.sciencedirect.com/science/article/pii/S2772392522000517]
- 47. Addiction Neural Dynamics [https://med.umn.edu/center-neural-circuits-addiction/addiction-neural-dynamics]
- 48. Discovery of Novel Pain Regulators Through Integration of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11807727/]
- 49. Modeling and Simulation of Neocortical Micro [https://elifesciences.org/reviewed-preprints/99688v1]
- 50. Establishing in vitro models of neurocircuits with human neurons [https://pmc.ncbi.nlm.nih.gov/articles/PMC5541685/]
- 51. “Neurocircuits to Behavior: The New Revolution” - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5347472/]
- 52. Allostasis and Addiction: Role of the Dopamine and... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3288230/]
- 53. Genome-wide association study implicates CHRNA2 in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7596896/]
- 54. Positive Selection on Loci Associated with Drug and Alcohol ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0134393]
- 55. The influence of single-nucleotide polymorphisms in opioid ... [https://www.sciencedirect.com/science/article/abs/pii/S2452014425000536]
- 56. Neuroplasticity in addiction: cellular and transcriptional perspectives - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3495339/]
- 57. Cortico-Striatal Circuits: Novel Therapeutic Targets for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9364041/]
- 58. Neuroplasticity in addictive disorders - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC3181920/]
- 59. The clinical relevance of neuroplasticity in corticostriatal networks during operant learning - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3830626/]
- 60. Neuroplasticity in addiction: cellular and transcriptional ... [https://www.frontiersin.org/journals/molecular-neuroscience/articles/10.3389/fnmol.2012.00099/full]
- 61. Corticostriatal Activity Driving Compulsive Reward Seeking [https://www.sciencedirect.com/science/article/pii/S0006322321015638]
- 62. Brain stress systems in the amygdala and addiction [https://www.sciencedirect.com/science/article/abs/pii/S0006899309006027]
- 63. “Identifying the neural circuitry of alcohol craving and relapse ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2879014/]
- 64. Psychological and neural mechanisms of relapse - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC2607321/]
- 65. Association of Drug Cues and Craving With Drug Use ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9161117/]
- 66. Addiction Craving and Cue Reactivity Initiative Network ... [https://med.umn.edu/addiction/network/acrin]
- 67. Drug Addiction, Dysregulation of Reward, and Allostasis [https://www.nature.com/articles/1395603]
- 68. Addiction and stress: An allostatic view [https://www.sciencedirect.com/science/article/abs/pii/S0149763418302185]
- 69. A Computational Hypothesis for Allostasis: Delineation of ... [https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2013.00167/full]
- 70. Hedonic Homeostatic Dysregulation as a Driver of Drug ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2801885/]
- 71. Translational Models of Addiction Phenotypes to Advance ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10823887/]
- 72. A Multilevel Computational Characterization of ... [https://www.eneuro.org/content/5/4/ENEURO.0151-18.2018]
- 73. Brain stress systems in the amygdala and addiction - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC2774745/]
- 74. (PDF) Parallel and interactive learning processes within the basal ... [https://www.academia.edu/17247521/Parallel_and_interactive_learning_processes_within_the_basal_ganglia_Relevance_for_the_understanding_of_addiction]
- 75. Natural Rewards, Neuroplasticity, and Non-Drug Addictions [https://pmc.ncbi.nlm.nih.gov/articles/PMC3139704/]
- 76. Natural rewards, neuroplasticity, and non-drug addictions [https://www.sciencedirect.com/science/article/abs/pii/S0028390811001195]
- 77. Navigating the Neural Space in Search of the Neural Code [https://www.sciencedirect.com/science/article/pii/S0896627317301034]
- 78. What is the Definition of Addiction [https://www.asam.org/quality-care/definition-of-addiction]
- 79. Risk of diabetes and hypertension in a population with alcohol ... [https://bmcpublichealth.biomedcentral.com/articles/10.1186/s12889-024-18318-y]
- 80. Medical complications associated with substance use ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6626564/]
- 81. Primary Care Patients With Substance Use Disorder More ... [https://www.bu.edu/aodhealth/2024/08/27/primary-care-patients-with-substance-use-disorder-more-likely-to-have-undiagnosed-hypertension-and-diabetes/]
- 82. The key to addressing the addiction epidemic begins with ... [https://www.jax.org/news-and-insights/jax-blog/2016/february/the-key-to-addressing-the-addiction-epidemic-begins-with-science]
- 83. Neurocognitive bases of emotion regulation development in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6989808/]
- 84. Emotion dysregulation as a marker in adolescent mental ... [https://www.nature.com/articles/s41598-025-22067-2]
- 85. The colors of our brain: an integrated approach for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9107439/]
- 86. Balancing act: Neural correlates of affect dysregulation in ... [https://www.sciencedirect.com/science/article/pii/S1878929320300232]
- 87. MAT vs 12-Step: Evidence-Based Addiction Recovery ... [https://faspsych.com/blog/evidence-based-mat-superior-addiction-recovery-over-traditional-methods/]
- 88. Spring 2025 Research Roundup [https://developingadolescent.semel.ucla.edu/steps-for-youth/item/spring-2025-research-roundups]
- 89. A shared neural basis underlying psychiatric comorbidity [https://www.nature.com/articles/s41591-023-02317-4]
- 90. A shared neural basis underlying psychiatric comorbidity [https://pubmed.ncbi.nlm.nih.gov/37095248/]
- 91. Shared Neurocircuitry of Traumatic Brain Injury and Trauma ... [https://pubmed.ncbi.nlm.nih.gov/34561028/]
- 92. The Neurocircuitry of Fear, Stress, and Anxiety Disorders [https://www.nature.com/articles/npp200983]
- 93. Neurocircuitry of Mood Disorders - PMC - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC3055427/]
- 94. Novel neurotherapeutic targets for substance use disorders [https://pubmed.ncbi.nlm.nih.gov/41193380/]
- 95. Reevaluating the brain disease model of addiction [https://www.sciencedirect.com/science/article/abs/pii/S2215036625000604]
- 96. Addiction as a brain disease revised: why it still matters, ... [https://www.nature.com/articles/s41386-020-00950-y]
- 97. Pros and Cons of Quantitative Research - Key Insights [https://heymarvin.com/resources/pros-and-cons-of-quantitative-research/]
- 98. Limitations and weakness of quantitative research methods [https://www.projectguru.in/limitations-quantitative-research/]