Protein-Protein Interaction Assays: A Comprehensive Guide for Signaling Pathway Analysis in Drug Discovery
This article provides a comprehensive overview of protein-protein interaction (PPI) assays and their pivotal role in deciphering cellular signaling pathways for biomedical research and therapeutic development.
Protein-Protein Interaction Assays: A Comprehensive Guide for Signaling Pathway Analysis in Drug Discovery
Abstract
This article provides a comprehensive overview of protein-protein interaction (PPI) assays and their pivotal role in deciphering cellular signaling pathways for biomedical research and therapeutic development. It covers foundational PPI biology, explores established and cutting-edge methodological approaches—from co-immunoprecipitation and yeast two-hybrid to deep learning and novel functional assays like LinkLight. The content offers practical troubleshooting guidance, frameworks for experimental validation, and comparative analysis to help researchers select the optimal techniques. By synthesizing traditional methods with recent computational advances, this guide empowers scientists to reliably map interaction networks, overcome common experimental hurdles, and accelerate drug discovery pipelines.
The Language of Signaling: Understanding Protein-Protein Interaction Fundamentals
Protein-protein interactions (PPIs) are fundamental regulators of cellular function, influencing a multitude of biological processes such as signal transduction, cell cycle regulation, transcriptional regulation, and metabolic pathways [1]. These interactions form an elaborate network, or interactome, that allows proteins to communicate and coordinate complex activities essential for life [2]. PPIs can be categorized based on their nature, temporal characteristics, and functions into several distinct types [1]. Stable interactions typically form large multiprotein complexes that exist for extended periods, such as the nuclear pore complex or the proteasome. In contrast, transient interactions are brief and reversible, often occurring in signaling cascades where they are activated by specific stimuli like post-translational modifications. PPIs can also be obligate, where the proteins are unstable outside the complex, or non-obligate, where the interacting proteins can exist independently. Other classifications include homodimeric (between identical proteins) and heterodimeric (between different proteins) interactions [1]. Understanding these diverse interaction types is crucial for elucidating cellular regulatory mechanisms and identifying potential therapeutic targets in disease pathways.
Classification and Characteristics of PPIs
The following table summarizes the core characteristics, functions, and experimental considerations for the major classes of protein-protein interactions.
Table 1: Classification and Characteristics of Protein-Protein Interactions
| Interaction Type | Stability & Duration | Key Functional Roles | Structural Features | Experimental Considerations |
|---|---|---|---|---|
| Stable Complexes | Long-lived; often obligate | Structural scaffolding (e.g., cytoskeleton), enzymatic complexes (e.g., proteasome) [1] | Large, buried interfaces with complementary surfaces [2] | Co-immunoprecipitation (Co-IP), affinity purification mass spectrometry (AP-MS), native PAGE [1] |
| Transient Signaling | Short-lived, reversible | Signal transduction, phosphorylation cascades, allosteric regulation [1] | Often smaller, shallower interfaces; can be dependent on PTMs [2] | Yeast two-hybrid (Y2H), surface plasmon resonance (SPR), fluorescence resonance energy transfer (FRET) |
| Homo-oligomeric | Between identical subunits | Form symmetric complexes; can regulate activity via cooperativity [1] | Symmetric binding interfaces | Analytical ultracentrifugation, cross-linking studies |
| Hetero-oligomeric | Between different subunits | Form multi-protein machines; integrate different functions [1] | Asymmetric, often modular interfaces | AP-MS, Y2H, protein complementation assays |
A critical structural concept in PPIs is the binding interface, which often contains specific residue combinations and unique architectural layouts forming cooperative "hot spots" [2]. These hot spots are defined as residues whose substitution results in a substantial decrease (ΔΔG ≥ 2 kcal/mol) in the binding free energy of a PPI [2]. The energetic contributions of hot spots stem from their localized networked arrangement within tightly packed "hot" regions, enabling flexibility and the capacity to bind to multiple different partners [2].
Quantitative Analysis of PPI Binding Pockets
Recent structural datasets have enabled a pocket-centric analysis of PPIs. The following table quantifies key characteristics of binding pockets involved in protein-protein interactions and their relationship with ligands, based on a comprehensive analysis of over 23,000 pockets [3].
Table 2: Quantitative Analysis of PPI and Ligand Binding Pockets
| Pocket Metric | Dataset Findings | Significance for Drug Discovery |
|---|---|---|
| Overall Dataset Scale | >23,000 pockets; >3,700 proteins; >500 organisms [3] | Provides a vast resource for structural analysis and machine learning model training |
| Orthosteric Competitive Pockets (PLOC) | Directly compete with protein partner's epitope [3] | Target for inhibitors that directly disrupt the PPI interface |
| Orthosteric Non-Competitive (PLONC) | Ligands bind orthosteric site without direct competition [3] | May influence partner function/conformation without direct steric hindrance |
| Allosteric Pockets (PLA) | Situated near but not overlapping orthosteric site [3] | Enable allosteric modulation of PPIs; often more druggable than flat orthosteric interfaces |
| Protein Family Coverage | >1,700 unique protein families represented [3] | Enables broad comparative studies and identification of cross-family binding motifs |
Experimental Protocols for PPI Analysis
PROPER-seq for Transcriptome-Scale PPI Mapping
PROPER-seq (Protein-Protein Interaction Sequencing) is a high-throughput method for mapping PPIs en masse by converting cellular transcriptomes into barcoded protein libraries [4].
Workflow:
- Library Construction: Convert the transcriptome of input cells (e.g., HEK293, T lymphocytes) into an RNA-barcoded protein library using SMART-display technology.
- Interaction Capture: Incubate the library to allow protein interactions. All interacting protein pairs are captured through proximity-driven barcode ligation.
- Sequencing and Decoding: Record interacting pairs as chimeric DNA sequences via reverse transcription and PCR amplification. Decode interactions massively by high-throughput sequencing and mapping to the reference proteome.
- Validation: Confirm novel interactions using orthogonal methods such as co-immunoprecipitation (coIP) and affinity purification-mass spectrometry (AP-MS).
Applications: PROPER-seq has identified 210,518 human PPIs, including 17,638 previously uncharacterized interactions and 17,000 computationally predicted interactions [4]. It is particularly valuable for identifying synthetic lethal gene pairs and mapping context-specific interactomes in different cell types.
Structural Characterization of PPI Interfaces
This protocol details the steps for characterizing PPI binding pockets from 3D structural data, as used to create large-scale datasets [3].
Workflow:
- Protein Selection and Curation:
- Source structures from the Protein Data Bank (PDB), selecting heterodimer complexes for PPIs and protein-ligand complexes.
- Apply quality filters: resolution ≤ 3.5 Å for X-ray/cryo-EM; R-free - R-factor ≤ 0.07 (X-ray); Fourier shell correlation ≥ 0.143 (cryo-EM).
- Remove structures with atoms in alternative locations at interfaces.
- Structure Preparation:
- Repair incomplete amino acids using FoldX software.
- Remove heteroatoms and water molecules.
- Protonate structures using the OPLS-AA force field in GROMACS.
- Pocket Detection and Classification:
- Detect pockets using VolSite with adjusted parameters to accommodate typically shallower PPI pockets.
- For heterodimers (HD), detect pockets at the interface using one protein as the target and the other as the "ligand," then reverse roles.
- Classify ligand-binding pockets in protein-ligand complexes as Orthosteric Competitive (PLOC), Orthosteric Non-Competitive (PLONC), or Allosteric (PLA) based on their spatial relationship to the PPI interface [3].
Visualizing Signaling Pathways and Experimental Workflows
PPI-Mediated Signaling Pathway
Diagram Title: PPI-Mediated Signal Transduction from Membrane to Nucleus
PROPER-seq Experimental Workflow
Diagram Title: PROPER-seq Workflow for Large-Scale PPI Mapping
PPI Pocket Classification
Diagram Title: Classification of Ligand Binding Pockets in PPIs
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents and Resources for PPI Analysis
| Reagent/Resource | Type | Function in PPI Research | Example Sources/Databases |
|---|---|---|---|
| STRING Database | Bioinformatics Database | Known and predicted PPIs across species; integrates genomic context data [1] | https://string-db.org/ [1] |
| BioGRID | Literature-Curated Database | Manually curated protein/genetic interactions from high-throughput studies [1] | https://thebiogrid.org/ [1] |
| PROPER v.1.0 Database | Experimental PPI Database | Repository of 210,518 human PPIs identified via PROPER-seq technology [4] | https://genemo.ucsd.edu/proper [4] |
| VolSuite | Software Tool | Detects and characterizes binding pockets on protein structures [3] | Cited in Method [3] |
| Co-immunoprecipitation (Co-IP) Kits | Laboratory Reagent | Validates binary PPIs from cell lysates using antibody-mediated pulldown [1] | Commercial suppliers (e.g., Thermo Fisher, Abcam) |
| Fragment Libraries | Chemical Reagents | Low molecular weight compounds for screening binders to PPI hot spots via FBDD [2] | Commercial suppliers (e.g., Maybridge, Enamine) |
| AlphaFold2 | Computational Tool | Predicts protein structures and complexes to model PPI interfaces [1] [2] | https://alphafold.ebi.ac.uk/ |
Therapeutic Targeting of PPIs
Targeting PPIs with small molecules has historically been challenging due to the large, flat nature of many interaction interfaces. However, several strategic approaches have enabled successful drug development [2]:
- Hot Spot Identification: Rational drug design focuses on structural hot spots—residues that contribute disproportionately to binding energy. Computational tools and alanine scanning mutagenesis are used to identify these regions [2].
- Fragment-Based Drug Discovery (FBDD): This approach uses low molecular weight fragments that bind weakly to sub-pockets within the PPI interface. These fragments are then optimized or linked to create high-affinity inhibitors [2].
- Peptidomimetics: These compounds are designed to mimic the secondary structure (e.g., α-helices, β-sheets) of key peptide regions involved in PPIs, thereby disrupting the interaction [2].
- Stabilizers vs. Inhibitors: While most efforts focus on PPI inhibition, stabilizers that enhance endogenous PPIs present a promising but more challenging therapeutic strategy, as they require a profound understanding of PPI thermodynamics [2].
The continued advancement of PPI research technologies, from high-throughput experimental mapping to sophisticated computational predictions, is rapidly expanding the druggable proteome and opening new avenues for therapeutic intervention in cancer, inflammatory diseases, and viral infections [2].
Why PPIs are Central to Signal Transduction and Cellular Decision-Making
Protein-protein interactions (PPIs) form the fundamental framework for cellular communication, acting as the primary mechanism through which cells receive, process, and respond to external and internal signals. These physical interactions between two or more proteins govern a vast array of biological processes, including signal transduction, gene expression regulation, metabolic pathways, and responses to stress [5] [1]. The network of these interactions, known as the interactome, allows proteins to coordinate complex functions essential for life, from structural support to catalyzing biochemical reactions [2]. Within signaling pathways, PPIs are not merely connections; they are dynamic, regulated events that determine the specificity, amplitude, and temporal nature of cellular signals, ultimately leading to critical cellular decisions such as proliferation, differentiation, and apoptosis [1] [2]. Understanding PPIs is therefore crucial for elucidating the molecular basis of cellular behavior and for identifying potential therapeutic targets in drug discovery [5] [2].
The Fundamental Role of PPIs in Signal Transduction
In signal transduction, extracellular signals are converted into intracellular responses through a series of PPIs. These interactions facilitate the relay of information from cell surface receptors to intracellular effectors, ensuring precise control over cellular activities.
- Signal Initiation and Amplification: The binding of a ligand to a cell surface receptor often induces a conformational change that promotes its interaction with downstream adapter proteins. This initial PPI nucleates the formation of larger signaling complexes, amplifying the signal as it propagates through the cell [2].
- Specificity and Regulation: PPIs confer specificity to signaling pathways by ensuring that only the correct proteins interact at the right time and place. This is often mediated by specialized protein domains such as SH2, SH3, and PDZ domains, which recognize specific peptide motifs or post-translational modifications on their binding partners [2]. The transient nature of many PPIs allows for rapid and reversible control of signaling flux.
- Integration of Signals: Cellular decision-making requires the integration of multiple signals. PPIs enable crosstalk between different signaling pathways, allowing the cell to compute a coordinated response to a complex set of stimuli. Dysregulation of these interacting networks is a hallmark of many diseases, including cancer and inflammatory disorders [2].
The following diagram illustrates a generalized signaling pathway driven by sequential PPIs, leading to a specific cellular decision.
Computational Prediction of PPIs: Methods and Data
The experimental characterization of PPIs can be time-consuming and resource-intensive. Computational methods, particularly those powered by machine learning (ML) and deep learning, have emerged as powerful tools for large-scale PPI prediction [5] [1].
The performance of ML models is heavily dependent on the quality and breadth of training data. Key biological databases provide essential ground-truth data for known and predicted interactions.
Table 1: Key Data Sources for PPI Prediction and Analysis
| Database Name | Description | Key Utility in PPI Research |
|---|---|---|
| STRING | Database of known and predicted PPIs derived from experiments, computational methods, and text mining [5] [1]. | Provides a comprehensive global perspective on protein interaction networks across species. |
| BioGRID | A repository of biologically relevant protein and genetic interactions from curated experimental data [5] [1]. | Source of high-quality, experimentally validated PPIs for model training and validation. |
| IntAct | Open-source database system and analysis tools for molecular interaction data [1]. | Provides curated PPI data for developing predictive models. |
| Protein Data Bank (PDB) | Single global archive for 3D structural data of proteins and nucleic acids [1]. | Essential for structure-based feature extraction and docking studies. |
| AlphaFold DB | Database of protein structure predictions from the AlphaFold AI system [5]. | Enables large-scale extraction of structural features for proteins without experimentally solved structures. |
Core Computational Approaches
Computational methods for predicting PPIs can be broadly categorized, each with its own strengths and applications.
Table 2: Core Computational Methods for PPI Prediction
| Method Category | Key Principle | Common Algorithms/Tools |
|---|---|---|
| Homology-Based Methods | Infers interactions based on evolutionary conservation, assuming that orthologous proteins in other species interact ("guilt by association") [2]. | BLAST, INTEROLOGUE mapping. |
| Traditional Machine Learning (ML) | Uses manually engineered features from protein sequences, structures, or genomic context to train classifiers [5] [2]. | Support Vector Machines (SVM), Random Forests (RF). |
| Deep Learning (DL) | Automatically learns hierarchical features and complex patterns from raw data like amino acid sequences or 3D structures [5] [1]. | Graph Neural Networks (GNNs), Convolutional Neural Networks (CNNs), Transformers. |
Deep learning architectures, particularly Graph Neural Networks (GNNs), are highly suited for PPI prediction because they can natively model the network-like structure of interactomes. GNNs, such as Graph Convolutional Networks (GCNs) and Graph Attention Networks (GATs), generate node representations by aggregating information from a protein's neighbors in the network, thereby capturing both local patterns and global relationships [1]. For example, the AG-GATCN framework integrates GAT and temporal convolutional networks to improve robustness against noise in PPI analysis [1].
The workflow for a typical deep learning-based PPI prediction pipeline is illustrated below.
Experimental Protocol: Validating PPIs in a Signaling Pathway
The following protocol provides a detailed methodology for experimentally validating a predicted PPI within a signaling pathway, using Co-Immunoprecipitation (Co-IP) followed by Western Blotting as a gold-standard approach.
Application Note
This protocol is designed to confirm a physical interaction between two proteins (Protein A and Protein B) suspected to interact in a specific signal transduction pathway. Validation is critical after in silico prediction and before functional assays.
Materials and Reagents
Table 3: Research Reagent Solutions for Co-IP Validation
| Reagent/Material | Function | Example/Note |
|---|---|---|
| Specific Antibodies | To capture and detect target proteins. | Anti-Protein A antibody for IP; Anti-Protein B for WB. |
| Cell Lysis Buffer | To solubilize cells and extract proteins while preserving native interactions. | Include non-ionic detergents (e.g., NP-40, Triton X-100) and protease/phosphatase inhibitors. |
| Protein A/G Beads | Solid-phase matrix to bind antibody-protein complexes. | Agarose or magnetic beads conjugated with Protein A/G. |
| Immunoblotting Reagents | For protein separation and detection. | SDS-PAGE gel, PVDF membrane, ECL substrate. |
| Control IgGs | To confirm the specificity of the IP. | Normal mouse/rabbit IgG for negative control. |
Step-by-Step Procedure
Cell Stimulation and Lysis
- Culture cells expressing Protein A and Protein B under appropriate conditions.
- If the PPI is stimulus-dependent (e.g., growth factor-induced), treat cells with the relevant ligand for the required time. Include an unstimulated control.
- Lyse cells in a suitable ice-cold lysis buffer (e.g., RIPA buffer) for 30 minutes. Centrifuge at high speed (14,000 x g) for 15 minutes at 4°C to clear the lysate.
Antibody-Bead Complex Preparation
- Pre-clear the cell lysate by incubating with control beads (e.g., Protein A/G) for 30-60 minutes to reduce non-specific binding.
- Meanwhile, incubate the specific antibody against Protein A (e.g., 1-5 µg) with Protein A/G beads in lysis buffer for at least 1 hour at 4°C on a rotator.
Co-Immunoprecipitation
- Incubate the pre-cleared cell lysate with the antibody-bead complex overnight at 4°C on a rotator.
- Prepare a negative control by incubating lysate with control IgG-bead complex.
- The following day, pellet beads by brief centrifugation and wash 3-4 times with cold lysis buffer to remove non-specifically bound proteins.
Elution and Western Blot Analysis
- Elute bound proteins from the beads by adding 2X Laemmli sample buffer and boiling for 5-10 minutes.
- Separate the eluted proteins and input lysate controls by SDS-PAGE.
- Transfer proteins to a PVDF membrane and perform Western blotting.
- Probe the membrane with an antibody against the putative interacting partner, Protein B, to detect its presence in the immunoprecipitate.
- Re-probe the membrane with an antibody against Protein A to confirm successful IP.
Data Interpretation
A successful Co-IP validation is indicated by a clear signal for Protein B in the lane where Protein A was immunoprecipitated, but not in the negative control IgG lane. This confirms a physical association between Protein A and Protein B under the tested conditions.
PPI Modulators in Drug Discovery and Therapeutic Applications
Given their central role in disease pathways, PPIs represent an attractive class of therapeutic targets. The development of PPI modulators—small molecules or biologics that inhibit or stabilize an interaction—has transitioned from a daunting challenge to a viable drug discovery strategy [2].
Strategies for PPI Modulator Discovery
- High-Throughput Screening (HTS): Utilizes chemically diverse libraries, often enriched with "PPI-friendly" compounds, to identify lead modulators [2].
- Fragment-Based Drug Discovery (FBDD): Particularly suited for targeting the discontinuous "hot spots" on PPI interfaces. Low molecular weight fragments bind to sub-sites and are subsequently linked or optimized into high-affinity inhibitors [2].
- Structure-Based Drug Design: Leverages high-resolution structural information (from X-ray crystallography, Cryo-EM, or AlphaFold2 predictions) of the PPI interface to rationally design inhibitors that mimic key interacting residues [5] [2].
Approved PPI Modulators
Several PPI modulators have received FDA approval, validating the clinical potential of this approach [2]. Key examples include:
- Venetoclax: Inhibits the BCL-2/BAX PPI, restoring apoptosis in cancer cells.
- Sotorasib & Adagrasib: Target the KRAS G12C mutant protein, once considered "undruggable."
- Maraviroc: A CCR5 antagonist that prevents HIV from interacting with the host cell co-receptor.
The diagram below outlines a generalized workflow for the discovery and development of PPI-targeted therapeutics.
Protein-protein interactions are fundamental to cellular signaling, and their specificity is often mediated by modular protein domains. Among the most critical are leucine zippers, SH2 domains, and SH3 domains. These domains facilitate the assembly of complex signaling networks that regulate processes ranging from immune response to cell growth and differentiation. Understanding their structure and function is essential for research in signaling pathway analysis and therapeutic development.
Leucine Zippers are coiled-coil domains that mediate protein dimerization, a key step in the activation of many transcription factors and signaling complexes. They are characterized by a repetitive heptad pattern where leucine residues appear at every seventh position, creating a hydrophobic interface that facilitates dimer stability and specificity [6]. In synthetic biology, engineered leucine zipper pairs (e.g., LZ-EE and LZ-RR) are used to recruit substrates to specific cellular locations with high affinity, enabling the precise control of synthetic signaling pathways [6].
SH2 Domains (Src Homology 2 domains) are approximately 100 amino acids long and specifically recognize and bind to phosphorylated tyrosine (pY) residues on partner proteins [7]. This binding is crucial for tyrosine kinase signaling, as it recruits downstream effector proteins to activated receptors. The human proteome contains roughly 110 proteins with SH2 domains, which are found in enzymes, adaptors, and transcription factors [7]. A key structural feature is a deep pocket within the βB strand that binds the phosphate moiety, involving a highly conserved arginine residue (βB5) that forms a salt bridge with the pY residue [7]. Beyond phosphopeptide binding, nearly 75% of SH2 domains can also interact with membrane lipids like PIP₂ and PIP₃, which helps in membrane recruitment and modulates their signaling activity [7]. Furthermore, SH2 domain-containing proteins like GRB2 and LAT are involved in driving the formation of liquid-liquid phase-separated condensates (LLPS), which enhance signaling efficiency in processes such as T-cell activation [7].
SH3 Domains (Src Homology 3 domains) are smaller modules of about 60 amino acids that typically bind to proline-rich motifs (PRMs) in partner proteins [8]. They fold into a compact β-barrel structure consisting of five β-strands connected by flexible loops (RT, n-Src, and distal loops) [8]. The sequence variation within these loops confers binding specificity for different PRMs. Some SH3 domains, such as those from c-Src, Eps8, and Nck1, can undergo non-canonical 3D domain-swapping, forming intertwined dimers or higher-order oligomers, which may represent a mechanism for amyloid fibril formation or alternative regulation [8].
Table 1: Key Characteristics of Protein Interaction Domains
| Domain | Typical Size | Primary Ligand | Key Structural Features | Main Biological Role |
|---|---|---|---|---|
| Leucine Zipper | Variable (heptad repeats) | Self (dimerization) | Coiled-coil α-helices, hydrophobic interface | Protein dimerization and complex assembly |
| SH2 Domain | ~100 amino acids | Phosphotyrosine (pY) motifs | β-sandwich fold, conserved Arg in pY pocket | Relay of phosphotyrosine signaling |
| SH3 Domain | ~60 amino acids | Proline-rich motifs (PRMs) | β-barrel fold, variable RT/n-Src loops | Recruitment of proline-rich effector proteins |
Quantitative Analysis of Domain Properties
Quantitative data on the biophysical and functional properties of these domains are crucial for experimental design, particularly in biosensor engineering and inhibitor development.
SH2 domains demonstrate a remarkable structural conservation despite low sequence identity (as little as ~15% in some family members), underscoring that their three-dimensional fold is almost exclusively optimized for binding pY-peptide motifs [7]. The binding affinity and specificity of SH2 domains are influenced by the amino acids C-terminal to the phosphotyrosine, typically at the pY+1 to pY+3 positions. The structural basis for this specificity lies in the conformation and sequence of surface loops, such as the EF and BG loops, which vary between different SH2 domains [7].
For leucine zippers, their utility in synthetic systems like the SPN-FLUX platform relies on a high signal-to-noise ratio. In this system, cognate zipper halves (e.g., LZ-EE and LZ-RR) provide high-affinity, specific recruitment, enabling minimal background activity and robust activation upon dimerization [6]. Flow cytometry and microplate reader assays confirmed that receptor-coupled networks using these zippers exhibited low background and significant signal induction upon stimulation [6].
Table 2: Quantitative Functional Data from Domain Applications
| Domain / System | Key Quantitative Metric | Experimental Context | Implication for Research |
|---|---|---|---|
| Engineered Leucine Zippers (in SPN-FLUX) | High signal-to-noise ratio; Significant MFI change post-induction | Mammalian cell biosensor (HEK293) | Enables design of low-background, inducible synthetic receptors [6] |
| SH2 Domain Family | ~75% bind membrane lipids (e.g., PIP2, PIP3) | Analysis of human SH2 proteome | Lipid binding is a major regulatory mechanism beyond pY recognition [7] |
| SH2 Domain Structure | As low as ~15% pairwise sequence identity | Structural genomics | High functional conservation despite low sequence homology [7] |
| c-Src SH3 Domain | Forms 3D domain-swapped dimers/oligomers | Biophysical characterization (pH, temp) | Potential for alternative folding states with pathological implications [8] |
Experimental Protocols and Workflows
Protocol: Probing SH2-pY Interactions with a SPN-FLUX-Based Biosensor
The following protocol details the use of a synthetic phosphorylation network to study SH2 domain recruitment to phosphorylated substrates in live mammalian cells, adapted from the SPN-FLUX platform [6].
Principle: A membrane-bound synthetic receptor is designed to phosphorylate a substrate upon ligand-induced dimerization. The phosphorylated ITAMs on the substrate then recruit a protein-binding (PB) domain containing SH2 domains, which is detected via complementation of a split fluorescent protein.
Reagents and Materials:
- Plasmids: Encoding the following components:
- KC (Kinase-Chain): A membrane-targeted fusion of FRB, CD28 transmembrane domain, and the active kinase domain of ABL1.
- ZC (Zipper-Chain): A membrane-targeted fusion of FKBP, CD28 transmembrane domain, and a leucine zipper half (LZ-EE).
- Substrate: A cytosolic fusion of three CD3ζ-derived ITAM motifs, the cognate leucine zipper half (LZ-RR), and the N-terminal fragment (β-strands 1-10) of mNeonGreen2.
- PB (Protein-Binding Domain): A cytosolic fusion of the tandem SH2 domains from ZAP70 and the C-terminal fragment (β-strand 11) of mNeonGreen2.
- Ligand: Rapamycin to induce FKBP/FRB dimerization.
- Cells: HEK293T cells.
- Instruments: Flow cytometer or fluorescent microplate reader.
Procedure:
- Cell Transfection: Co-transfect HEK293T cells with the four plasmids (KC, ZC, Substrate, PB) using a standard transfection method (e.g., PEI or lipofection).
- Ligand Stimulation: At 24-48 hours post-transfection, treat cells with 100-500 nM rapamycin or a vehicle control (DMSO). Incubate for 1-3 hours.
- Signal Detection:
- Flow Cytometry: Harvest cells, resuspend in PBS, and analyze using a flow cytometer. Excite at 506 nm and detect emission at 517 nm. The mean fluorescence intensity (MFI) of the mNeonGreen2 channel indicates SH2 domain recruitment and complex formation.
- Microplate Reading: Transfer cells to a clear-bottom 96-well plate. Measure fluorescence directly in the plate reader using the same excitation/emission settings.
- Data Analysis: Normalize the MFI of rapamycin-treated samples to the vehicle control to calculate the fold induction. A high signal-to-noise ratio confirms specific, ligand-dependent recruitment of the SH2 domain-containing PB module to the phosphorylated substrate.
Protocol: Analyzing SH3 Domain Interactions and Oligomerization
This protocol outlines the biochemical and biophysical characterization of SH3 domains, including their canonical PRM binding and non-canonical 3D domain-swapping, based on studies of the c-Src SH3 domain [8].
Principle: Wild-type or mutant SH3 domains are expressed, purified, and subjected to biophysical analyses to assess their stability, PRM-binding capability, and propensity to form domain-swapped oligomers.
Reagents and Materials:
- Expression Vector: pHTP1 with an N-terminal 6xHis-tag and TEV cleavage site.
- Cells: E. coli BL21(DE3) competent cells.
- Buffers: Lysis buffer, Ni-NTA binding/wash/elution buffers, size-exclusion chromatography (SEC) buffer.
- Instruments: AKTA FPLC system, SEC column (e.g., Superdex 75), circular dichroism (CD) spectrometer, differential scanning calorimetry (DSC).
Procedure:
- Cloning and Mutagenesis: Clone the gene for the SH3 domain of interest (e.g., c-Src, Abl) into the expression vector. Generate chimeric constructs by swapping loop regions (e.g., RT, n-Src loops) using synthetic gene synthesis or site-directed mutagenesis.
- Protein Expression and Purification:
- Transform the plasmid into E. coli BL21(DE3) cells. Induce expression with IPTG.
- Lyse cells and purify the 6xHis-tagged protein via Ni-NTA affinity chromatography.
- Cleave the His-tag using TEV protease and perform a second Ni-NTA step to remove the tag and protease.
- Further purify the protein using SEC. Analyze the elution profile to identify monomeric and potential oligomeric peaks.
- Biophysical Characterization:
- Thermal Stability: Use CD spectroscopy or DSC to measure the melting temperature (Tm) of the SH3 domain. Compare wild-type and chimeric proteins to assess the impact of loop swaps on stability.
- Oligomerization State: Use analytical SEC or multi-angle light scattering (MALS) under varying conditions (e.g., pH, concentration) to quantify the formation of domain-swapped dimers.
- Ligand Binding: Use isothermal titration calorimetry (ITC) or surface plasmon resonance (SPR) to measure the binding affinity (Kd) for a canonical proline-rich peptide ligand.
The following workflow diagram illustrates the key steps in the SPN-FLUX protocol for analyzing SH2-pY interactions:
Figure 1: Experimental workflow for the SPN-FLUX biosensor assay to study SH2-pY interactions.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents and Tools for Domain Studies
| Reagent / Tool | Function / Description | Example Use Case | Key Feature |
|---|---|---|---|
| SPN-FLUX Platform [6] | A fully post-translational biosensor platform integrating synthetic phosphorylation with split reporters. | Real-time detection of SH2 domain recruitment to phosphorylated ITAMs in live cells. | Rapid response (<1 hour); tunable reporting; modular. |
| CoDIAC Python Package [9] | A comprehensive, structure-based domain interface analysis tool. | Mapping SH2 domain interfaces and identifying regulatory PTMs from structural data. | Reveals coordinated regulation by phosphorylation/acetylation. |
| STRING Database [10] | A public resource of protein-protein associations, including physical and functional interactions. | Placing SH2/SH3 domain-containing proteins into functional pathways and networks. | Integrates experimental, predicted, and curated data; provides confidence scores. |
| Engineered Leucine Zippers (LZ-EE/LZ-RR) [6] | High-affinity, specific peptide pairs for forced protein recruitment. | Recruiting a cytosolic substrate to a membrane receptor in synthetic signaling circuits. | High affinity and specificity; low background. |
| Chimeric SH3 Domains [8] | SH3 domains with swapped loop regions (e.g., RT, n-Src loops). | Elucidating the structural determinants of domain swapping and PRM binding specificity. | Allows dissection of loop-specific functions. |
The following diagram illustrates the molecular architecture of the SPN-FLUX biosensor, showing how its components interact to generate a signal:
Figure 2: Molecular mechanism of the SPN-FLUX biosensor. Rapamycin-induced dimerization brings the kinase close to its substrate. Phosphorylated ITAMs on the substrate are then bound by the SH2 domains of the PB module, bringing the split fluorescent protein fragments together and generating a detectable signal.
Protein-protein interactions (PPIs) are fundamental to cellular signaling, regulating processes from gene expression to metabolic pathway flux [11]. Disruption of these interactions is a cardinal feature of numerous diseases, including cancer and neurodegeneration, making them attractive therapeutic targets [11]. A detailed understanding of the biological consequences of PPIs—specifically altered enzyme kinetics, substrate channeling, and the formation of new binding sites—is therefore critical for both basic research and drug discovery. This application note details practical protocols and analytical frameworks for investigating these phenomena within signaling pathway analysis, providing researchers with methodologies to de-risk early-stage discovery and accelerate program timelines.
Altered Kinetics from Protein-Protein Interactions
Quantitative Analysis of Kinetic Parameters
The formation of transient enzyme assemblies can significantly alter catalytic efficiency ((k{cat}/KM)) and substrate selectivity by reshaping the enzyme's conformational landscape [12] [13]. Table 1 summarizes kinetic parameter changes observed in a computational redesign of aspartate aminotransferase (AAT), where remodeling enriched a reactive conformation and altered function [13].
Table 1: Kinetic Consequences of Remodeling the Conformational Landscape of Aspartate Aminotransferase (AAT) [13]
| Enzyme Variant | (K_M) for l-Aspartate (mM) | (k_{cat}) for l-Aspartate (s^{-1})) | (k{cat}/KM) for l-Aspartate (M^{-1}s^{-1})) | (K_M) for l-Phenylalanine (mM) | (k_{cat}) for l-Phenylalanine (s^{-1})) | (k{cat}/KM) for l-Phenylalanine (M^{-1}s^{-1})) | Selectivity Switch (Fold) |
|---|---|---|---|---|---|---|---|
| Wild-Type (WT) | 0.21 ± 0.03 | 7.8 ± 0.3 | 37,000 ± 5,000 | N.D. | N.D. | 400 ± 100 | (Reference) |
| HEX Mutant | 0.059 ± 0.007 | 1.32 ± 0.03 | 22,000 ± 3,000 | 0.27 ± 0.03 | 9.0 ± 0.2 | 33,000 ± 4,000 | 1.5 |
| VYIY Mutant | 0.09 ± 0.02 | 0.244 ± 0.006 | 2,700 ± 600 | 0.58 ± 0.02 | 20.9 ± 0.2 | 36,000 ± 1,000 | 13 |
Protocol: Measuring Altered Kinetics via Steady-State Analysis
This protocol is adapted from kinetic analyses used to characterize remodeled aminotransferases [13] and standard enzymatic assays for HTS [14].
Key Research Reagents:
- Purified wild-type and mutant enzymes (e.g., AAT variants).
- Native and non-native substrates (e.g., l-Aspartate and l-Phenylalanine).
- Cofactors (e.g., Pyridoxal Phosphate (PLP) for AAT).
- Assay Buffer (e.g., 50 mM HEPES, pH 7.5).
- Stopping reagent compatible with detection method.
- Microplate reader or spectrophotometer.
Procedure:
- Reaction Setup: Prepare a master mix containing assay buffer, cofactor, and a fixed, limiting concentration of enzyme.
- Substrate Titration: Aliquot the master mix into a series of tubes or a microplate well. Initiate the reaction by adding a range of substrate concentrations (e.g., 0.1 x (KM) to 10 x (KM)).
- Incubation: Allow the reaction to proceed at a controlled temperature (e.g., 30°C) for a predetermined time, ensuring less than 10% of the substrate is consumed to maintain initial velocity conditions.
- Reaction Termination: Stop the reaction at precise time points by adding a stopping reagent (e.g., acid) or by rapid heat inactivation.
- Product Quantification: Measure the concentration of the reaction product using a calibrated method (e.g., spectrophotometry, HPLC).
- Data Analysis: Plot the initial velocity (v) against substrate concentration ([S]). Fit the data to the Michaelis-Menten equation ((v = (V{max} * [S]) / (KM + [S]))) using non-linear regression software to determine (KM) and (V{max}). Calculate (k{cat} = V{max} / [E]_{total}).
Data Interpretation: A significant change in (k{cat}/KM) for a non-native substrate, as seen in Table 1, indicates a successful alteration of the enzyme's conformational landscape and selectivity [13].
Signaling Pathway Impact of Altered Kinetics
Altered enzyme kinetics from PPIs can directly modulate signal transduction flux. For instance, in GPCR pathways, differential recruitment of β-arrestin versus G-proteins represents a kinetically distinct branch point that can be profiled using functional cell-based assays [11].
Diagram 1: GPCR signaling branches with distinct kinetic outcomes.
Substrate Channeling in Metabolic Regulation
Mechanisms and Quantitative Evidence
Substrate channeling describes the direct transfer of a metabolic intermediate between consecutive enzymes in a pathway without its release into the bulk cytosol [12]. This phenomenon is a frequent consequence of enzyme assembly and can enhance pathway efficiency, protect labile intermediates, and regulate flux at metabolic branch points [12] [15]. A common misconception is that channeling accelerates steady-state reaction rates; for most enzymes, metabolite diffusion is not rate-limiting [12]. The primary kinetic benefit is often a reduction in the lag phase (transient time) before the pathway reaches steady state [12].
Table 2 contrasts the primary mechanisms of substrate channeling and their functional consequences.
Table 2: Mechanisms and Functional Consequences of Substrate Channeling in Enzyme Assemblies [12] [15]
| Mechanism | Description | Key Functional Consequence | Example Pathway(s) |
|---|---|---|---|
| Tunneling | Intermediate passes through a physical tunnel between active sites. | Sequesters toxic or labile intermediates. | Tryptophan synthase, Polyketide biosynthesis |
| Electrostatic Channeling | Intermediate transfer guided by complementary electrostatic surfaces. | Increases local substrate concentration near the active site. | Glycolysis, Oxidative phosphorylation |
| Swing Arms / Covalent Tethering | Intermediate is covalently attached to a flexible prosthetic group. | Enables direct transfer between distantly spaced active sites. | Fatty acid biosynthesis, Polyketide synthases |
| Proximity in Clustered Assemblies | Metabolite transfer via bounded diffusion within a dense enzyme cluster. | Reduces transient time, regulates flux at branch points. | TCA cycle metabolon, Purine biosynthesis |
Protocol: Differentiating Substrate Channeling from Microenvironmental Effects
Observed rate enhancements in scaffolded enzyme systems are often attributed to substrate channeling. However, the scaffold itself can create a local microenvironment (e.g., altered pH, charge, crowding) that masquerades as channeling by independently modifying enzyme kinetics [15]. This protocol outlines a strategy to distinguish between these effects.
Key Research Reagents:
- Purified sequential enzymes (e.g., Glucose Oxidase and Horseradish Peroxidase).
- Scaffolding system (e.g., DNA nanostructures, protein scaffolds, synthetic polymers).
- Enzyme activity assay reagents (e.g., chromogenic/fluorogenic substrates).
- Control: Non-scaffolded enzyme mixture, enzymes attached to a non-interacting scaffold.
Procedure:
- Cascade Assembly: Assemble the two-enzyme cascade in three configurations:
- A. Freely Diffusing: Enzymes mixed in solution without a scaffold.
- B. Proximity-Scaffolded: Enzymes co-localized on a scaffold designed to bring them into close proximity (<10 nm).
- C. Microenvironment-Control: Enzymes immobilized on a scaffold that does not allow proximity but exposes them to the same scaffold material (e.g., separated beads on the same polymer).
- Kinetic Measurement: Initiate the reaction with the first substrate and monitor the formation of the final product over time. Ensure measurements capture the pre-steady-state (lag phase) and steady-state phases.
- Data Analysis:
- Compare the lag phase duration between conditions A, B, and C. A significantly shortened lag phase in condition B compared to both A and C provides strong evidence for substrate channeling [12] [15].
- Compare the steady-state rate ((V_{max})). If the rate in condition B is higher than in A, but similar to C, the enhancement is likely due to a general microenvironment effect rather than specific channeling [15].
- Cascade Assembly: Assemble the two-enzyme cascade in three configurations:
Data Interpretation: True substrate channeling is indicated by a specific reduction in transient time in the proximity-scaffolded condition. General rate enhancements across all scaffolded conditions suggest dominant microenvironmental effects, such as local pH changes or charge interactions provided by the scaffold material [12] [15].
Diagram 2: Contrasting free diffusion and substrate channeling.
Creation and Exploitation of New Binding Sites
Induced Proximity and Molecular Glues
PPIs can create novel, composite binding interfaces that do not exist on the individual proteins. This principle is harnessed by therapeutic strategies like molecular glues and targeted protein degradation, where a small molecule induces proximity between a target protein and an effector (e.g., an E3 ubiquitin ligase), leading to the target's ubiquitination and degradation [11].
Protocol: Detecting PPIs via the LinkLight Assay
The LinkLight platform is a functional, cell-based assay ideal for detecting transient PPIs, such as GPCR-β-arrestin recruitment, by converting them into a stable, luminescent signal [11].
Key Research Reagents:
- LinkLight Assay Kit: Contains vectors for Protein A-TEV protease and Protein B-permuted luciferase (pLuc) fusions.
- Cell line appropriate for the target proteins (e.g., HEK293).
- Transfection reagent.
- Ligand/compound library for screening.
- Luciferin substrate.
- Luminescence plate reader.
Procedure:
- Construct Design: Fuse the protein of interest (e.g., a GPCR) to TEV protease (Protein A). Fuse its binding partner (e.g., β-arrestin) to a permuted luciferase (pLuc) interrupted by a TEV cleavage site (Protein B).
- Cell Transfection: Co-transfect cells with the two fusion constructs.
- Ligand Stimulation & Incubation: Treat cells with ligands or test compounds for a predetermined time to induce the PPI.
- Signal Detection: Add a luciferin substrate to the cells. Measure luminescence using a plate reader. When Proteins A and B interact, the TEV protease cleaves the pLuc, allowing it to refold into an active enzyme that produces light. This cleavage event is irreversible, providing a "molecular memory" of the transient interaction [11].
Data Interpretation: An increase in luminescent signal indicates ligand-induced PPI. This assay is particularly powerful for profiling ligand bias (e.g., G-protein vs. β-arrestin signaling) and for screening molecular glues that induce novel PPIs [11].
The Scientist's Toolkit: Essential Research Reagents and Assays
Table 3: Key Research Reagent Solutions for PPI and Consequence Analysis
| Reagent / Assay Type | Core Function | Key Application in Analysis |
|---|---|---|
| LinkLight Functional Cell-Based Assay [11] | Detects transient PPIs via TEV protease-mediated luciferase complementation. | Ideal for measuring β-arrestin recruitment, pathway activation, and molecular glue discovery in live cells. |
| Turku Bioscience PPI Services [16] | Suite of assays using recombinant proteins, cell extracts, or intact cells. | Useful for high-throughput screening or hit expansion against specific PPIs with ultra-high sensitivity. |
| Steady-State Kinetics Assays [14] [13] | Measures (KM) and (k{cat}) to quantify catalytic efficiency. | Foundational for characterizing altered enzyme kinetics resulting from PPIs or conformational remodeling. |
| Computational Protein Design (Multistate) [13] | Predicts mutations to stabilize specific protein conformations. | Used to rationally remodel conformational landscapes to create enzymes with new selectivity or activity. |
| DNA/Protein Scaffolds [12] [15] | Provides nanoscale control over enzyme positioning. | Enables experimental testing of substrate channeling vs. microenvironmental effects in synthetic metabolons. |
Integrated Workflow for Analyzing PPI Consequences
The following diagram outlines a logical workflow for dissecting the biological consequences of a protein-protein interaction, from initial detection to functional validation.
Diagram 3: A logical workflow for analyzing PPI functional consequences.
The Link Between PPI Dysregulation and Disease Pathogenesis
Protein-protein interactions (PPIs) are fundamental to cellular life, governing the vast majority of biological processes including cell-to-cell interactions, metabolic control, and signal transduction [17]. These noncovalent contacts between residue side chains form the basis for protein folding, assembly, and the intricate networks that enable cellular communication [17]. The dysregulation of these critical interactions represents a central mechanism in the pathogenesis of numerous diseases, marking PPIs as attractive targets for therapeutic intervention [18] [2]. When PPIs are disrupted—whether through genetic mutation, altered expression, or external modulation—the consequences can be severe, leading to dysfunctional signaling pathways that drive conditions such as cancer, neurodegenerative disorders, and inflammatory diseases [18] [2].
The clinical relevance of PPI modulation is demonstrated by several FDA-approved therapies. Drugs such as venetoclax, sotorasib, and adagrasib specifically target dysregulated PPIs in cancer, while maraviroc and tocilizumab address PPIs in viral infections and inflammatory conditions, respectively [2]. These clinical successes underscore the importance of understanding PPI dysregulation and developing assays to detect and characterize these pathogenic interactions for drug discovery and development.
Mechanisms and Consequences of PPI Dysregulation
Fundamental Mechanisms of Dysregulation
PPI dysregulation occurs through multiple mechanistic pathways that disrupt normal cellular function. These include:
Disruption of Transient Signaling Complexes: Transient PPI interactions form the backbone of cellular signaling pathways. When these interactions are disrupted, either through excessive inhibition or stabilization, information flow through critical pathways like GPCR signaling is compromised [17] [18]. For example, biased agonism in GPCR pathways depends on selective protein partnerships, and dysregulation can lead to preferential signaling through pathogenic pathways [18].
Alteration of Stable Protein Complexes: Permanent PPI interactions form stable complexes that perform essential structural and enzymatic functions. Dysregulation of these complexes through mutations at interaction interfaces can lead to complete loss of function or dominant-negative effects that disrupt normal cellular architecture and function [17].
Hub Protein Dysfunction: Proteins with large numbers of interactions (hubs) including enzymes, transcription factors, and intrinsically disordered proteins are particularly vulnerable to dysregulation. When these hub proteins are affected, the consequences propagate through multiple cellular pathways simultaneously, leading to widespread cellular dysfunction [17] [2].
Energetic and Structural Basis
The structural basis of PPI dysregulation often centers on "hot spots"—specific residues within interaction interfaces whose substitution results in substantial decreases in binding free energy (ΔΔG ≥ 2 kcal/mol) [2]. These hot spots are characterized by their localized networked arrangement within tightly packed "hot" regions, which enables flexibility and capacity to bind multiple partners [2]. Disease-associated mutations frequently cluster in these critical regions, disrupting the hydrophobic effects and specific residue combinations that normally stabilize the interactions [2].
Table 1: Characteristics of PPI Dysregulation Mechanisms
| Dysregulation Mechanism | Structural Basis | Functional Consequence | Exemplary Diseases |
|---|---|---|---|
| Hot Spot Disruption | Mutations in energetically critical residues | Decreased binding affinity, loss of function | Cancer, rare genetic disorders |
| Allosteric Modulation | Binding at secondary sites altering interface geometry | Pathogenic activation or inhibition | Inflammatory diseases, metabolic disorders |
| Expression Imbalance | Altered stoichiometry of complex components | Non-productive complexes, dominant-negative effects | Neurodegenerative diseases |
| Post-translational Modification | Modified interface residues affecting electrostatics | Gained or lost interactions | Autoimmune diseases, cancer |
Advanced Detection Methods for PPI Dysregulation
Experimental Assay Platforms
The detection and characterization of dysregulated PPIs requires specialized assay platforms capable of capturing both stable and transient interactions:
LinkLight Functional Cell-Based Assay: This technology detects fleeting protein-protein interactions in living cells by converting transient binding events into stable, non-reversible luminescent signals [18]. The assay is based on tobacco etch virus (TEV) protease cleavage and luciferase complementation technology, where Protein A is fused to TEV protease and Protein B is fused to a permuted luciferase (pLuc) interrupted by TEV recognition/cleavage sequences [18]. When the proteins interact, TEV cleaves the recognition site, allowing luciferase refolding and generating a stable luminescent signal that persists even after protein dissociation [18].
Yeast Two-Hybrid (Y2H) Systems: As an in vivo method, Y2H screens a protein of interest against a random library of potential protein partners within the cellular environment, preserving post-translational modifications that may affect interactions [17].
Tandem Affinity Purification-Mass Spectrometry (TAP-MS): This in vitro method involves double tagging of the protein of interest at its chromosomal locus, followed by a two-step purification process and mass spectrometric analysis to identify protein interaction partners under native cellular conditions [17].
Protein-Fragment Complementation Assays (PCAs): These assays detect PPIs between proteins of any molecular weight expressed at endogenous levels by using split reporter proteins that only reassemble and become functional upon interaction of the target proteins [17].
Computational Prediction Methods
Computational approaches have become increasingly sophisticated in predicting PPI dysregulation:
Homology-Based Methods: These operate on the "guilt by association" principle, predicting interactions based on significant sequence similarity with known interactors. While accurate for well-characterized proteins, their applicability is limited when experimentally determined homologs are unavailable [2].
Template-Free Machine Learning Methods: Algorithms including Support Vector Machines (SVMs) and Random Forests (RFs) identify patterns in vast datasets of known interacting and non-interacting protein pairs, using features like amino acid sequences, protein structures, or interaction affinities to train predictive models [2].
Structure-Based Approaches: These methods predict protein-protein interactions based on structural similarity at primary, secondary, or tertiary levels, leveraging the growing repository of protein structural information [17] [2].
Table 2: PPI Detection Methods and Their Applications
| Method Category | Specific Techniques | Key Advantages | Limitations | Ideal for Detecting |
|---|---|---|---|---|
| In Vivo | Yeast Two-Hybrid (Y2H) | Cellular environment, PTMs preserved | False positives from spurious interactions | Novel interaction discovery |
| In Vitro | TAP-MS, Co-IP, Affinity Chromatography | Controlled conditions, identification of weak interactions | May miss context-dependent interactions | Stable complex identification |
| Functional Cell-Based | LinkLight, PCA | Captures transient interactions, physiological relevance | Requires specialized reagents | Signaling complex dynamics |
| In Silico | Sequence/Structure-based, Phylogenetic Profiles | High-throughput, low cost | Dependent on quality of input data | Prioritization for experimental validation |
Experimental Protocol: Detecting Dysregulated GPCR-β-Arrestin Interactions Using LinkLight
Background and Principle
GPCR signaling and regulation involves precisely orchestrated PPIs, with β-arrestin recruitment serving as a critical mechanism for receptor desensitization and internalization [18]. Dysregulation of GPCR-β-arrestin interactions contributes to numerous pathological conditions, including cardiovascular diseases, metabolic disorders, and inflammation [18]. This protocol describes the detection and quantification of these dysregulated interactions using the LinkLight assay platform, which converts transient recruitment events into stable luminescent signals through TEV protease-mediated cleavage and luciferase complementation [18].
Materials and Reagents
- Cell Line: HEK293 or specialized cell line expressing GPCR of interest
- Plasmids:
- pLinkLight-GPCR-TEV: Expression vector for GPCR-TEV fusion protein
- pLinkLight-β-arrestin-pLuc: Expression vector for β-arrestin-permuted luciferase fusion protein
- LinkLight Assay Kit: (Available from commercial providers like Reaction Biology) containing:
- Luciferin substrate
- Cell lysis buffer
- Assay dilution buffers
- Cell Culture Materials:
- Complete growth medium (DMEM with 10% FBS, 1% penicillin-streptomycin)
- Serum-free medium for transfection
- Trypsin-EDTA for cell detachment
- Transfection Reagent: Polyethylenimine (PEI) or commercial equivalent
- White Walled 96-Well or 384-Well Assay Plates: Luminescence-compatible
- Luminescence Plate Reader: Capable of detecting luciferase activity
Procedure
Day 1: Cell Seeding
- Harvest exponentially growing HEK293 cells by trypsinization.
- Count cells and adjust concentration to 150,000 cells/mL in complete growth medium.
- Seed 100 μL cell suspension (15,000 cells) per well in white-walled 96-well assay plates.
- Incubate plates overnight at 37°C, 5% CO₂ to achieve 70-80% confluency at time of transfection.
Day 2: Plasmid Transfection
- Dilute 50 ng total DNA per well (25 ng pLinkLight-GPCR-TEV + 25 ng pLinkLight-β-arrestin-pLuc) in 5 μL serum-free medium.
- Dilute 0.15 μL transfection reagent in 5 μL serum-free medium per well.
- Combine diluted DNA and transfection reagent, mix gently, and incubate 15-20 minutes at room temperature.
- Add 10 μL DNA-transfection reagent complex dropwise to each well.
- Gently swirl plate to ensure even distribution.
- Incubate transfected cells for 24 hours at 37°C, 5% CO₂.
Day 3: Ligand Stimulation and Signal Detection
- Prepare ligand dilutions in assay buffer at 2X final concentration.
- Remove culture medium from transfected cells and replace with 50 μL serum-free medium.
- Add 50 μL 2X ligand dilutions to appropriate wells, including vehicle controls.
- Incubate plates for predetermined optimal time (typically 2-6 hours) at 37°C, 5% CO₂.
- Equilibrate LinkLight luciferin substrate to room temperature.
- Add 25 μL luciferin substrate directly to each well without removing stimulation medium.
- Incubate plate for 10 minutes at room temperature.
- Measure luminescence using plate reader with integration time of 0.5-1 second per well.
Data Analysis and Interpretation
Raw Data Processing:
- Subtract background luminescence (vehicle-only control) from all test values.
- Calculate mean and standard deviation for replicate wells.
Dose-Response Analysis:
- Plot log(ligand concentration) versus normalized luminescence response.
- Fit data to four-parameter logistic equation to determine EC₅₀ values.
- Compare potency and efficacy values between wild-type and dysregulated PPIs.
Quality Control Parameters:
- Z'-factor > 0.5 indicates robust assay performance.
- Signal-to-background ratio > 3:1 demonstrates sufficient dynamic range.
Pathway Analysis Incorporating PPI Networks
The integration of PPI networks into pathway analysis represents a powerful strategy for identifying dysregulated biological processes in disease states. Methods such as the Pathway analysis method Using Protein-Protein Interaction network for case-control data (PUPPI) aggregate gene-gene interaction signals within pathways defined by PPI networks, increasing power to detect effects that might be missed when focusing solely on main effects [19]. This approach has successfully identified clinically relevant pathways, including the chaperones modulate interferon signaling pathway in Crohn's disease, which modulates interferon gamma and induces the JAK/STAT pathway implicated in disease pathogenesis [19].
Research Reagent Solutions for PPI Analysis
Table 3: Essential Research Reagents for PPI Dysregulation Studies
| Reagent Category | Specific Examples | Function & Application | Key Considerations |
|---|---|---|---|
| Cellular Assay Systems | LinkLight Assay Kit, Yeast Two-Hybrid Systems | Detect transient PPIs in physiological environments | Signal stability, cellular context preservation |
| Protein Purification Tools | TAP-Tag Systems, Affinity Chromatography Resins | Isolate protein complexes for interaction analysis | Maintain native protein conformations |
| Detection Reagents | Luminescent Substrates, Fluorescently-Labeled Antibodies | Quantify interaction strength and dynamics | Sensitivity, signal-to-noise ratio |
| Expression Vectors | TEV Fusion Constructs, Split-Reporter Plasmids | Express tagged proteins for interaction studies | Tag positioning effects on native interactions |
| Computational Resources | STRING Database, BioGRID, AlphaFold2 | Predict interactions and structural interfaces | Data quality, validation requirements |
The systematic analysis of PPI dysregulation provides critical insights into disease pathogenesis and reveals novel therapeutic opportunities. By employing integrated experimental and computational approaches—from cellular assays like LinkLight to pathway analysis tools like PUPPI—researchers can map dysregulated interaction networks with unprecedented resolution. As PPI-targeted therapies continue to advance, with several now achieving FDA approval, the methods and protocols outlined in this application note will support ongoing efforts to translate understanding of PPI dysregulation into effective treatments for cancer, inflammatory diseases, neurological disorders, and other conditions driven by aberrant protein interactions. The growing toolbox of PPI detection and modulation strategies positions this field as a cornerstone of 21st-century therapeutic development.
A Practical Toolkit: From Classic to Cutting-Edge PPI Assay Technologies
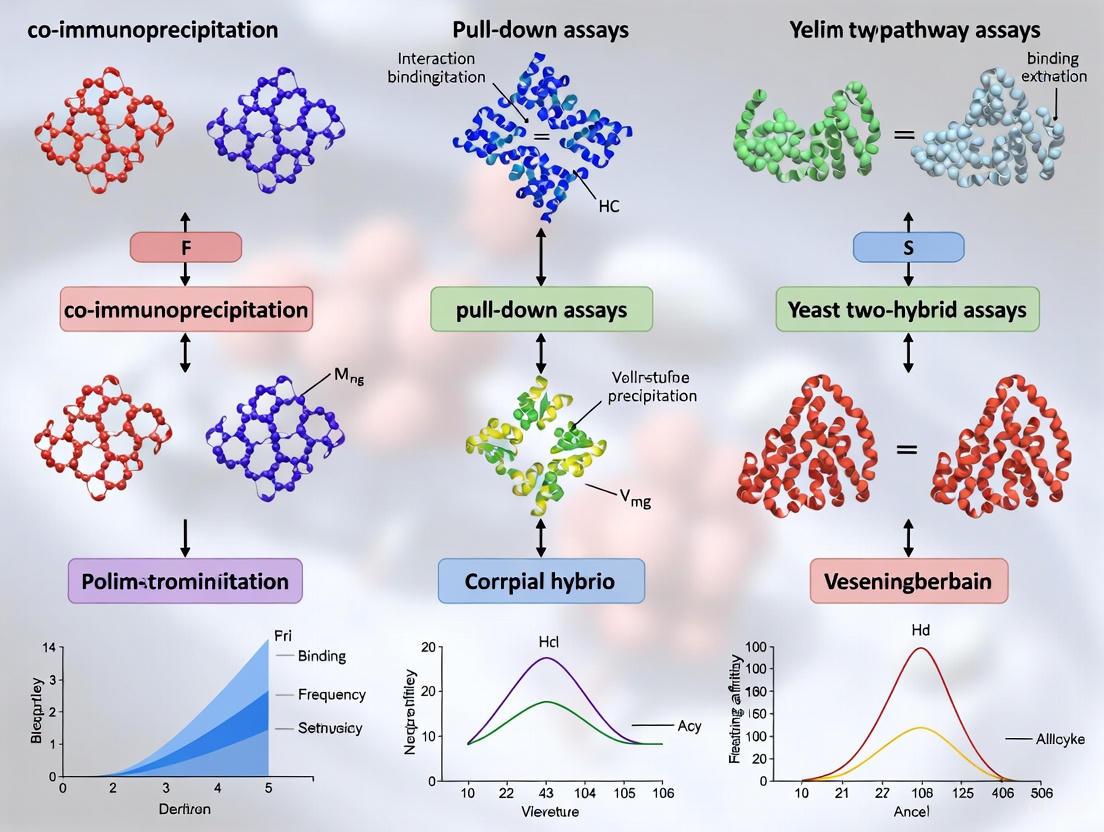
Protein-protein interactions (PPIs) are fundamental to cellular signaling and transduction, controlling a wide range of biological processes including signal transduction, metabolic control, and developmental regulation [17]. The majority of genes and proteins realize resulting phenotype functions as a set of interactions, with over 80% of proteins not operating alone but in complexes [17]. Elucidation of PPI networks contributes greatly to the analysis of signal transduction pathways and has become a major objective of systems biology [17] [20]. For researchers studying signaling pathways, the ability to reliably detect and characterize these interactions is paramount. Among the various techniques available, Co-immunoprecipitation (Co-IP) and pull-down assays have emerged as gold standard methods for PPI detection in physiologically relevant contexts, playing an increasingly important role in drug discovery and the development of PPI modulators [21] [2].
This application note provides detailed methodologies and comparative analysis of Co-IP and pull-down assays, framed within the context of signaling pathway analysis to support researchers in selecting and implementing these powerful techniques.
Core Principles and Comparative Analysis
Co-immunoprecipitation (Co-IP)
Co-IP is a classic in vivo method for studying protein interactions based on the specific interaction between antibodies and antigens under non-denaturing conditions [22]. When cells are lysed under these conditions, protein-protein interactions are preserved. A target "bait" protein is immunoprecipitated using a specific antibody immobilized on agarose or magnetic beads, and any "prey" proteins bound to the bait protein in vivo are co-precipitated [23] [22]. The isolated protein complexes can then be analyzed by western blotting or mass spectrometry to identify interaction partners [24].
The key advantage of Co-IP is its ability to isolate protein complexes from a natural cellular environment, preserving post-translational modifications that may be essential for interaction [17] [23]. This makes it particularly valuable for studying signaling pathways where such modifications regulate protein function and interaction dynamics.
Pull-Down Assays
Pull-down assays are a form of affinity purification similar to Co-IP but use tagged bait proteins instead of antibodies [22]. In this approach, a tagged bait protein is captured by a solid-phase affinity ligand that specifically binds to that tag [25]. Common tag systems include GST (glutathione S-transferase), polyhistidine (His-tag), and biotin, each with corresponding affinity resins (glutathione-sepharose for GST, nickel-nitrilotriacetic acid for His-tag, and streptavidin for biotin) [22]. The bait protein immobilized on the support can then be used to capture putative prey proteins from various protein samples [22].
While pull-down assays are powerful for determining direct interactions between known proteins and can detect proteins from in vitro transcription or translation systems, they may not always reflect physiological interactions since the proteins may not naturally encounter each other in the cell [22].
Comparative Analysis of PPI Detection Methods
Table 1: Comparison of Key PPI Detection Methodologies
| Method | Principle | Context | Key Applications | Advantages | Limitations |
|---|---|---|---|---|---|
| Co-IP [24] [22] | Antibody-mediated precipitation of protein complexes from cell lysates | In vivo (native cellular environment) | - Confirming hypothesized interactions- Studying protein complexes in native state- Analyzing post-translational modification-dependent interactions | - Studies interactions under physiological conditions- Preserves protein complexes in native form- High reliability for in vivo interactions | - May detect indirect interactions- May miss low-affinity/transient interactions- Requires specific antibody- Antibody might block interaction site |
| Pull-Down [25] [22] | Affinity purification using tagged bait proteins | In vitro (controlled conditions) | - Testing direct protein interactions- Screening putative prey proteins- Validating yeast two-hybrid results | - Determines direct interactions- No antibody required- Flexible experimental conditions | - May not reflect physiological conditions- Tag may interfere with protein function- Requires protein tagging |
| Yeast Two-Hybrid (Y2H) [17] | Reconstitution of transcription factor via protein interaction in yeast nuclei | In vivo (yeast system) | - High-throughput interaction screening- Mapping interaction networks | - High-throughput capability- Sensitive to transient interactions- Can screen complex libraries | - High false-positive rate- Limited to nuclear proteins- May miss interactions requiring PTMs |
| TAP-MS [17] | Tandem affinity purification with mass spectrometry | In vivo (native cellular environment) | - Comprehensive complex identification- Mapping protein interaction networks | - Identifies wide variety of complexes- Tests activeness of protein complexes- Low false-positive rate | - Time-consuming- Requires genetic modification- May miss transient complexes |
| Protein Microarrays [17] | High-throughput protein binding to immobilized probes | In vitro (high-throughput screening) | - Large-scale interaction screening- Antibody profiling- Biomarker discovery | - Simultaneous analysis of thousands of parameters- High-throughput capability | - May not reflect native protein conformations- Limited by protein immobilization |
Detailed Experimental Protocols
Co-IP Protocol for Signaling Pathway Analysis
Stage 1: Lysate Preparation [23]
Table 2: Lysis Buffer Selection Guide
| Protein Localization | Recommended Buffer | Composition | Application Notes |
|---|---|---|---|
| Membrane or Cytoplasmic (mild lysis) | NP-40 Lysis Buffer | 150 mM NaCl, 1% NP-40, 50 mM Tris-HCl pH=8.0, 0.15% (w/v) BSA, 10% (v/v) glycerol, protease/phosphatase inhibitors | Preserves weak protein interactions; suitable for most signaling complexes |
| Cytoplasmic or Nuclear (harsh lysis) | RIPA Lysis Buffer | 50 mM Tris-HCl pH=8.0, 150 mM NaCl, 1% NP-40, 0.5% sodium deoxycholate, 0.1% SDS, protease/phosphatase inhibitors | Disrupts nuclear membrane; use when studying transcription factors in signaling pathways |
- Cell Culture and Treatment: Culture appropriate cell lines and treat according to experimental design (e.g., growth factor stimulation for signaling pathway activation).
- Cell Lysis:
- Wash cells with ice-cold PBS
- Lyse cells in appropriate ice-cold lysis buffer (300 µL for 1-3×10⁷ cells)
- Incubate on ice for 10 minutes
- Sonicate 3 times briefly if needed to disrupt nuclear material
- Centrifuge at 8,000 × g for 10 minutes at 4°C
- Collect supernatant (lysate)
- Protein Quantification: Determine protein concentration using Bradford or BCA assay. Adjust concentrations to 1-2 mg/mL for consistent results.
Stage 2: Pre-clearing (Optional) [23]
- Incubate lysate with protein A/G beads alone or with control IgG for 30-60 minutes at 4°C
- Centrifuge to remove beads and non-specifically bound material
- Retain pre-cleared supernatant for immunoprecipitation
Stage 3: Immunoprecipitation [26] [24]
- Antibody Immobilization: Incubate specific antibody against bait protein with protein A/G-coupled agarose or magnetic beads for 1-2 hours at 4°C
- Complex Formation: Incubate antibody-bead complex with cell lysate (300 µg to 2 mg total protein) for 2-4 hours or overnight at 4°C with gentle rotation
- Washing: Pellet beads and wash 3-4 times with ice-cold lysis buffer to remove non-specifically bound proteins
- Elution: Elute bound proteins using Laemmli buffer (for WB) or low-pH elution buffer (for MS analysis)
Stage 4: Analysis [24]
- Western Blotting: Separate proteins by SDS-PAGE, transfer to membrane, and probe with antibodies against bait and potential prey proteins
- Input Control: Reserve 1-10% of original lysate as input control to confirm presence of target proteins
- Mass Spectrometry: For unknown interaction partners, analyze eluted proteins by LC-MS/MS
GST Pull-Down Assay Protocol
Stage 1: Bait Protein Preparation [22]
- GST Fusion Protein Expression: Express GST-tagged bait protein in appropriate expression system (E. coli, mammalian cells)
- Protein Purification: Purify GST fusion protein using glutathione-sepharose beads
- Immobilization: Incubate purified GST-bait protein with glutathione beads for 1-2 hours at 4°C
Stage 2: Prey Protein Preparation
- Source Selection: Prepare prey protein from:
- In vitro transcription/translation system
- Cell lysate expressing prey protein
- Purified prey protein
Stage 3: Binding Reaction [22]
- Incubate immobilized GST-bait protein with prey protein source in appropriate binding buffer for 2-4 hours at 4°C
- Include GST-only control to identify non-specific binding to GST tag
Stage 4: Washing and Elution
- Wash beads 3-4 times with binding buffer containing 150-300 mM NaCl to reduce non-specific binding
- Elute bound proteins with SDS-PAGE sample buffer or reduced glutathione elution buffer
Stage 5: Analysis
- Analyze eluted proteins by SDS-PAGE and western blotting with appropriate antibodies
- For quantitative analysis, use densitometry to compare band intensities [26]
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Research Reagents for Co-IP and Pull-Down Assays
| Reagent Category | Specific Examples | Function | Selection Considerations |
|---|---|---|---|
| Lysis Buffers [23] | NP-40 Buffer, RIPA Buffer | Solubilize proteins while preserving interactions | Choose based on protein localization and interaction stability; mild detergents preserve complexes |
| Protease Inhibitors [23] | Cocktail tablets, PMSF, AEBSF | Prevent protein degradation during processing | Essential for all steps; include phosphatase inhibitors for phosphoprotein studies |
| Bead Matrices [25] | Protein A/G Agarose, Magnetic Beads | Solid support for antibody or bait immobilization | Magnetic beads reduce mechanical stress on complexes; consider binding capacity |
| Tag Systems [22] | GST, His-tag, Biotin | Bait protein immobilization for pull-downs | GST offers high-affinity binding; His-tag works under denaturing conditions |
| Detection Antibodies [24] | Primary and secondary antibodies for WB | Target protein detection and visualization | Validate specificity for intended application; consider species compatibility |
| Elution Buffers [23] | Laemmli buffer, Low-pH glycine | Release bound proteins from beads | Choose based on downstream application (WB vs. functional assays) |
Applications in Signaling Pathway Research
Signaling Complex Analysis
Co-IP is particularly valuable for studying signaling complexes that form in response to extracellular stimuli. For example, in growth factor signaling pathways, receptor activation often leads to the formation of multi-protein complexes that include receptors, adaptor proteins, and effector enzymes [17]. Co-IP can capture these dynamic complexes from stimulated cells, allowing researchers to map the composition and regulation of signaling nodes.
The technique's ability to work with native proteins in a cellular context makes it ideal for studying how post-translational modifications (phosphorylation, ubiquitination) regulate complex formation and signaling output [23]. By comparing interactions under different stimulation conditions, researchers can build dynamic models of signaling pathway regulation.
Validation of Pathway Components
In pathway discovery research, high-throughput methods like yeast two-hybrid screens often generate large numbers of potential interactions that require validation in a more physiological context [17] [24]. Co-IP serves as an essential orthogonal validation method to confirm these putative interactions using endogenous proteins from relevant cell types.
For mapping linear signaling pathways, researchers can employ Co-IP to trace the flow of signal transduction from cell-surface receptors to nuclear transcription factors, confirming physical interactions between consecutive components in the pathway [20].
Drug Discovery Applications
The critical role of PPIs in cellular signaling has made them attractive targets for therapeutic intervention [2]. Co-IP assays play a crucial role in drug discovery by:
- Validating interactions targeted by PPI modulators
- Assessing the effects of drug candidates on pathogenic interactions
- Profiling ligand bias in GPCR signaling by comparing G protein vs. β-arrestin recruitment [18]
- Supporting mechanism-of-action studies for targeted protein degraders and molecular glues [2]
Advanced Co-IP derivations and complementary technologies like the LinkLight assay, which converts transient PPIs into stable luminescent signals, are increasingly being adopted in drug discovery pipelines for their sensitivity in detecting weak or transient interactions [21] [18].
Troubleshooting and Optimization
Common Challenges and Solutions
Low Signal or No Detection
- Cause: Insufficient antibody affinity or abundance of target protein
- Solution: Increase amount of starting material (up to 2 mg total protein); verify antibody specificity with positive control; try different antibody epitopes
High Background
- Cause: Non-specific binding to beads or antibody
- Solution: Include pre-clearing step; optimize wash stringency (increase salt concentration to 300-500 mM NaCl); use isotype control antibody to identify non-specific bands [23]
Inconsistent Results
- Cause: Variation in lysis efficiency or protein degradation
- Solution: Standardize lysis protocol; always use fresh protease inhibitors; aliquot and store lysates properly at -80°C
Critical Controls for Reliable Data
- Negative Controls: Isotype control antibody (same species as IP antibody) to identify non-specific binding [23] [24]
- Beads-Only Control: Beads without antibody to assess non-specific binding to bead matrix
- Input Control: 1-10% of original lysate to confirm presence of target proteins [24]
- Positive Control: Known interacting pair to validate experimental conditions
- Knockout/Knockdown Control: Cells lacking bait protein to confirm interaction specificity
Co-IP and pull-down assays remain indispensable tools for studying protein-protein interactions in signaling pathway research. While each method has distinct strengths and limitations, their complementary application provides powerful insights into the complex networks that regulate cellular signaling. Co-IP excels at capturing physiological interactions in their native context, making it ideal for validating interactions discovered through high-throughput methods and studying regulated complex formation in response to cellular stimuli. Pull-down assays offer precision for mapping direct interactions and characterizing binding domains.
The continued advancement of these techniques, including improved bead technologies, more specific antibodies, and integration with sensitive detection methods, ensures their ongoing relevance in both basic research and drug discovery. For researchers investigating signaling pathways, mastering these fundamental techniques provides a critical foundation for elucidating the complex protein interactions that underlie cellular communication and function.
Protein-protein interactions (PPIs) are fundamental to cellular signaling and transduction, governing processes such as immune response, growth, and differentiation [17] [2]. Mapping these interactions is crucial for understanding cellular function and complex phenotypes, and PPIs have become attractive targets for therapeutic drug development [27] [2]. In vivo techniques, particularly Yeast Two-Hybrid (Y2H) and Split-Ubiquitin systems, allow for the investigation of PPIs within a living cell, preserving the native structure, post-translational modifications, and subcellular context that are essential for studying signaling pathways [17] [27]. These genetic approaches provide a sensitive method to detect weak or transient interactions that might be lost in in vitro biochemical methods [27]. This document details the application and protocol for these two key in vivo systems within the broader context of signaling pathway analysis.
System Fundamentals and Comparative Analysis
Core Principles and Mechanisms
The Classical Yeast Two-Hybrid (Y2H) system is based on the modular nature of eukaryotic transcription factors [28]. The "bait" protein is fused to a DNA-binding domain (BD), and the "prey" protein is fused to a transcription activation domain (AD). If the bait and prey interact, the BD and AD are brought into proximity, reconstituting a functional transcription factor that drives the expression of reporter genes (e.g., HIS3, ADE2, LacZ), allowing yeast to grow on selective media or produce a colorimetric signal [28].
The Split-Ubiquitin System, particularly the Membrane Yeast Two-Hybrid (MYTH), is designed for studying membrane protein complexes [27]. The bait protein is fused to the C-terminal fragment of ubiquitin (Cub), which is itself fused to a transcription factor (e.g., LexA-VP16). The prey is fused to a mutated N-terminal fragment of ubiquitin (NubG). Interaction between bait and prey reconstitutes ubiquitin, which is recognized by cellular ubiquitin proteases (Ubp), leading to the cleavage and release of the transcription factor. This factor then enters the nucleus to activate reporter genes [29] [27].
The table below summarizes the key characteristics, strengths, and optimal applications for each system to guide researchers in selecting the appropriate methodology.
Table 1: Comparison of Yeast Two-Hybrid and Split-Ubiquitin Techniques
| Feature | Yeast Two-Hybrid (Y2H) | Split-Ubiquitin (e.g., MYTH/iMYTH) |
|---|---|---|
| Core Principle | Reconstitution of a transcription factor [28] | Reconstitution of ubiquitin and protease cleavage [27] |
| Primary Application | Soluble, nuclear, and cytoplasmic proteins [28] | Integral membrane proteins and their partners [27] |
| Cellular Environment | Nucleus | Native membrane environment |
| Key Advantage | Well-established; ideal for soluble protein libraries | Studies membrane proteins in their native context [27] |
| Common Reporter Genes | HIS3, ADE2, LacZ [28] |
HIS3, ADE2, LacZ [27] |
| Critical Consideration | Proteins must localize to the nucleus | Avoids overexpression artifacts with integrated systems (iMYTH) [27] |
Experimental Protocols and Workflows
Protocol: Classical Yeast Two-Hybrid Assay
This protocol is adapted for screening a protein of interest (bait) against a library of potential binding partners (prey) [28].
A. Main Instruments & Reagents
- Yeast Strains: AH109 or Y187 [28].
- Media: YPDA, synthetic dropout (SD) media lacking specific amino acids (e.g., -Leu/-Trp, -Leu/-Trp/-His) [28].
- Key Reagents: TE/LiAC buffer, PEG/LiAC, DMSO, X-β-Gal [28].
B. Step-by-Step Methodology
Yeast Transformation: Co-transform the purified bait (BD-X) and prey (AD-Y) plasmids into competent yeast cells using the LiAC method [28].
- Inoculate yeast into YPDA and incubate at 30°C with shaking until mid-log phase (A600 ~ 0.5).
- Pellet cells and resuspend in fresh TE/LiAC buffer.
- Combine plasmid DNA, carrier DNA, and competent cells in an EP tube.
- Add PEG/LiAC, mix by vortexing, and incubate at 30°C for 30 min.
- Add DMSO, heat-shock at 42°C for 15 min, then place on ice.
- Plate transformations on SD media lacking leucine and tryptophan (SD -Leu/-Trp) to select for cells containing both plasmids. Incubate at 30°C for 3-5 days until colonies appear [28].
Interaction Screening: Select for protein-protein interaction using more stringent selective media.
- Pick grown colonies and streak or spot onto SD media lacking leucine, tryptophan, and histidine (SD -Leu/-Trp/-His). The expression of the
HIS3reporter gene allows growth only if a successful PPI occurs [28]. - Incubate plates at 30°C for 3-7 days and monitor growth.
- Pick grown colonies and streak or spot onto SD media lacking leucine, tryptophan, and histidine (SD -Leu/-Trp/-His). The expression of the
Validation Assay (β-galactosidase Filter Assay): Confirm interactions through a second reporter gene,
LacZ.- Lift yeast colonies onto a sterile Whatman filter paper.
- Freeze the filter in liquid nitrogen for at least 13 seconds to permeabilize cells.
- Thaw the filter and place it on another filter pre-soaked with Z-buffer/X-β-Gal solution.
- Incubate at room temperature for 8-16 hours. Positive interaction is indicated by the development of blue colonies [28].
The workflow for this classical Y2H assay is summarized in the diagram below.
Protocol: Integrated Membrane Yeast Two-Hybrid (iMYTH)
iMYTH is used to study interactions involving integral membrane proteins by tagging the endogenous bait gene, avoiding overexpression artifacts [27].
A. Key Features
- Allows in vivo interaction tests for integral membrane proteins in their native environment [27].
- Bait and prey are expressed under native promoters from their genomic loci [27].
B. Step-by-Step Methodology
Strain Engineering: Genomically tag the endogenous gene of the membrane protein (bait) with the Cub-LexA-VP16 (CLV) construct. Generate a prey library where proteins are tagged with NubG at their amino or carboxyl terminus [27].
Selection for Interactors: Mate the CLV-tagged bait strain with the NubG-tagged prey library. Select for diploid yeast on appropriate media. The reconstitution of ubiquitin upon bait-prey interaction leads to cleavage of the transcription factor and activation of reporter genes like
HIS3orADE2[27].Interaction Confirmation: Identify positive interactors by growth on media lacking histidine or adenine. Specificity can be further tested by quantifying growth or using additional reporter assays [27].
The fundamental mechanism of the split-ubiquitin system used in iMYTH is illustrated below.
Advanced Applications and Modern Innovations
Next-Generation Interaction Screening (NGIS)
Traditional Y2H analysis, which relies on picking individual colonies and Sanger sequencing, is being superseded by Next-Generation Interaction Screening (Y2H-NGIS). This approach combines Y2H with deep sequencing to quantitatively analyze entire interactomes on a genome-wide scale [30]. Computational frameworks like Y2H-SCORES have been developed to tackle the associated informatics challenges. These tools rank candidate interactions based on metrics such as significant enrichment under selection, interaction specificity, and in-frame prey selection, leading to higher-confidence interactor lists and more reliable network models [30].
Selection for Interaction-Disrupting Mutations
The split-ubiquitin system can be ingeniously applied to select for mutations that specifically disrupt a given PPI. As demonstrated for the yeast proteins Bem1 and Cdc24, a randomized mutant library of the bait protein is selected under conditions where survival is contingent upon the loss of interaction [29]. When combined with next-generation sequencing, this method allows for comprehensive mapping of residue-specific contributions to a protein interface, providing critical insights for drug discovery by identifying "hot spots" [29] [2].
The Scientist's Toolkit: Essential Research Reagents
Successful execution of these techniques requires a suite of specialized reagents. The following table catalogs the key solutions for setting up Y2H and Split-Ubiquitin experiments.
Table 2: Key Research Reagent Solutions for Y2H and Split-Ubiquitin Experiments
| Reagent / Solution | Function / Application | Specific Examples / Notes |
|---|---|---|
| Yeast Strains | Host organism for the genetic assay | AH109, Y187 (for classical Y2H) [28] |
| BD and AD Vectors | Plasmid systems for expressing bait and prey fusions | pGBKT7 (BD), pGADT7 (AD) or similar [28] |
| Specialized MYTH Vectors | Plasmids for CLV and NubG fusions | pBT3-N, pPR3-N (commercial systems) |
| Selective Media | Selection of transformed yeast and interacting clones | SD -Leu/-Trp (double dropout), SD -Leu/-Trp/-His (triple dropout) [28] |
| Transformation Kit | High-efficiency introduction of DNA into yeast | LiAC/PEG method components [28] |
| β-galactosidase Substrate | Detection of LacZ reporter gene activity | X-β-Gal in Z-buffer [28] |
| Split-Ubiquitin Reporter | Detection of ubiquitin reconstitution | HIS3, ADE2, LacZ [27] |
Yeast Two-Hybrid and Split-Ubiquitin techniques are powerful, complementary in vivo systems for elucidating protein-protein interactions critical to signaling pathways. The classical Y2H remains a cornerstone for studying soluble proteins, while MYTH and its integrated variant, iMYTH, provide a unique window into the interactome of membrane proteins—a class of high therapeutic value. The ongoing innovation in these fields, such as next-generation sequencing integration and sophisticated computational analysis, continues to enhance the throughput, quantification, and reliability of PPI data. These advanced methods empower researchers in both academia and drug development to de-risk decisions and accelerate the discovery of novel therapeutic targets by providing high-quality, decision-ready data on complex cellular signaling networks.
Protein-protein interactions (PPIs) are fundamental regulators of cellular function, influencing critical biological processes including signal transduction, cell cycle regulation, and transcriptional control [1]. These interactions can be categorized as either stable or transient, with transient interactions being particularly challenging to capture due to their short-lived nature under physiological conditions [31] [32]. Traditional methods such as co-immunoprecipitation (Co-IP) and pull-down assays often fail to detect these fleeting encounters, creating a significant gap in our understanding of dynamic cellular processes [31] [32].
Crosslinking and label transfer strategies have emerged as powerful techniques to address this methodological limitation. By chemically "trapping" momentary interactions and transferring a detectable label between binding partners, these approaches provide researchers with a means to study weak and transient complexes that were previously inaccessible [31] [33]. This application note details the principles, methodologies, and practical implementation of these techniques within the context of signaling pathway analysis, providing researchers and drug development professionals with comprehensive protocols for investigating the dynamic interactome.
Core Principles of Label Transfer Technology
Label transfer incorporates crosslinking methodology to study protein-protein interactions by specifically labeling proteins that interact with a protein of interest [32]. This approach enables the discovery of new interactions, confirms putative interactions suggested by other methods, and investigates the interfaces of interacting proteins [32]. The fundamental innovation lies in its ability to capture molecular interactions at the exact moment they occur through photochemical crosslinking, then permanently mark the interacting partner via a transferable tag [31].
The label transfer method employs a label transfer reagent (LTR) containing three key functional elements: (1) a reactive group that covalently binds to a purified "bait" protein, (2) a photoactivatable group that crosslinks to interacting "prey" proteins upon UV exposure, and (3) a detectable label (biotin, fluorescent, or radioactive) that is transferred to the prey protein after cleavage of a spacer arm [32]. This strategic design allows researchers to permanently mark proteins engaged in transient interactions with their bait protein of interest, enabling subsequent detection, purification, and identification.
Recent advancements have led to the development of "tag and transfer" approaches that further refine this methodology. These innovative reagents incorporate a methanethiosulfonate (MTS) group for specific attachment to a reactive cysteine introduced into the bait protein, and a residue-unbiased diazirine-based photoactivatable crosslinking group to trap interacting partners [34]. The disulfide bond-containing linker enables reductive cleavage that transfers a thiol-containing tag onto the target protein, which can be alkylated and located by mass spectrometry sequencing [34].
Label Transfer Methodologies: Comparative Analysis
The evolution of label transfer reagents has progressed from early radioactive compounds to modern trifunctional reagents that more adequately segregate reactive sites from labels [32]. The table below summarizes the key characteristics of different label types used in transfer experiments:
Table 1: Comparison of Label Transfer Reagent Types
| Label Type | Detection Method | Sensitivity | Safety Considerations | Applications |
|---|---|---|---|---|
| Biotin | Streptavidin-HRP/AP, affinity purification | High | Minimal; ideal for most labs | Most applications; especially prey purification [32] |
| Radioactive (I-125) | Autoradiography | Very High | Significant; requires special handling | Historical studies; limited current use [32] |
| Fluorescent | Fluorescence imaging | Moderate | Moderate; light sensitivity | Cellular tracking; limited by stability [32] |
The trifunctional biotin-based reagent Sulfo-SBED represents one of the most widely used contemporary tools for label transfer applications [32]. Its structural and functional properties include:
- Amine-reactive NHS-ester group: For labeling a purified bait protein at the N-terminus and side chain of lysine residues
- UV light-activatable aryl azide group: For crosslinking nonspecifically to protein side chains and backbones of interacting proteins
- Cleavable disulfide bond (S-S): Can be reduced to release the crosslinker from the original bait protein
- Biotin group: Remains attached to target interacting protein after cleavage for affinity purification and detection [32]
The strategic advantage of biotin-based tags lies in their compatibility with both detection (through chromogenic or fluorescent methods) and purification (using streptavidin-coated beads), enabling researchers not only to identify interacting partners but also to isolate them for further characterization [32].
Experimental Protocol: Label Transfer Using Biotin-Based Reagents
Reagent Preparation
- Sulfo-SBED Solution: Prepare fresh Sulfo-SBED (Thermo Scientific) in anhydrous DMSO at a concentration of 1-5 mM. Protect from light and use immediately.
- Labeling Buffer: 20 mM HEPES, 150 mM NaCl, pH 7.5 (amine-free buffer recommended)
- Reduction Solution: 50 mM DTT or 50 mM TCEP in aqueous buffer
- Lysis/Binding Buffer: 50 mM Tris-HCl, 150 mM NaCl, 1% NP-40, pH 7.5 (with protease inhibitors)
Step-by-Step Procedure
Bait Protein Labeling:
- Incubate 50-100 µg of purified bait protein with 5-20 molar excess of Sulfo-SBED in labeling buffer for 30 minutes at room temperature, protected from light.
- Remove excess reagent by gel filtration or dialysis using amine-free buffer.
Protein Complex Formation:
- Incubate the labeled bait protein with prey protein or protein mixture (cell lysate, purified complex) for 30-60 minutes at 4°C or room temperature to allow complex formation.
UV-Induced Crosslinking:
- Transfer sample to a UV-transparent container and irradiate with long-wave UV light (365 nm) for 1-5 minutes using a suitable UV lamp. For higher efficiency, specialized LED platforms can achieve maximal crosslinking yields within 10 seconds [34].
- Note: Optimal UV exposure time should be determined empirically to maximize crosslinking while minimizing protein damage.
Label Transfer via Reduction:
- Add reduction solution to the crosslinked sample to a final concentration of 10-50 mM.
- Incubate for 30 minutes at 37°C to cleave the disulfide bond, transferring the biotin tag to the prey protein.
Detection and Analysis:
- Analyze by SDS-PAGE followed by Western blotting with streptavidin-HRP.
- For prey identification, perform streptavidin-based affinity purification followed by mass spectrometry analysis.
Diagram 1: Label transfer workflow using biotin-based reagents
Critical Optimization Parameters
Successful label transfer experiments depend on meticulous control of several parameters:
- Crosslinker-to-Protein Ratio: Typically 5:1 to 20:1 molar ratio; must be optimized to balance labeling efficiency with protein function preservation
- UV Exposure Time: Varies by lamp intensity; 1-5 minutes for standard UV lamps or as little as 10 seconds for high-intensity LED systems [34]
- Reduction Conditions: Concentration of reducing agent and incubation time must be sufficient for complete cleavage
- Control Experiments: Always include controls with bait protein alone and with non-specific proteins to assess background labeling
Advanced Applications: Quantitative Crosslinking/Mass Spectrometry (QCLMS)
Quantitative crosslinking/mass spectrometry (QCLMS) has emerged as a powerful method to probe protein structural dynamics in solution by quantitatively comparing crosslink yields between different conformational states [35] [33]. This approach uses isotope-labeled crosslinkers (e.g., BS³ and BS³-d4) to distinguish between different protein conformations or interaction states through mass differentials in mass spectrometry analysis [35].
The QCLMS workflow involves crosslinking parallel samples of different protein states with light (BS³) and heavy (BS³-d4) crosslinkers, mixing the samples in a 1:1 ratio, and analyzing by LC-MS/MS. Crosslinked peptides appear as doublets separated by 4 Da in mass spectra, with intensity ratios reflecting differences in crosslinking efficiency between conformational states [35]. Incorporation of replica analysis and label-swapping procedures is essential for robust quantification, addressing challenges of low reproducibility and signal intensity variations inherent in crosslinking experiments [35].
Table 2: Quantitative Crosslinking/Mass Spectrometry Applications
| Application | Experimental Design | Key Insights | References |
|---|---|---|---|
| Conformational Changes | Compare crosslink yields between protein states | Reveals subtle local and large-scale structural rearrangements | [35] |
| Complex Assembly | Monitor crosslinking patterns during assembly | Identifies binding interfaces and assembly pathways | [33] |
| Dynamic Interactions | Compare interaction strengths under different conditions | Quantifies affinity changes in transient complexes | [34] |
| Drug Effects | Assess crosslinking patterns with/without compounds | Maps ligand-induced conformational changes | [32] |
Tag-Transfer Crosslinking for Rapid Interactions
For particularly dynamic and transient interactions, such as chaperone-substrate complexes, traditional crosslinking approaches may still miss rapid interactions. The "tag and transfer" methodology addresses this challenge using reagents that incorporate a methanethiosulfonate (MTS) group for specific cysteine labeling and a diazirine-based photoactivatable group for rapid crosslinking [34]. These reagents enable maximal crosslinking yields within 10 seconds when used with high-intensity UV LED irradiation platforms, representing a 130-fold improvement compared to traditional mercury-xenon lamps [34].
The tag-transfer approach was successfully applied to map the dynamic interaction interface of the chaperone/substrate complex Skp/OmpA, where the binding interface involves many rapidly interconverting interactions [34]. In this system, traditional methods struggle to capture the transient interface, but the combination of specific cysteine labeling, rapid photoactivation, and tag transfer enabled precise mapping of interaction sites despite the dynamic nature of the complex.
Diagram 2: Tag-transfer crosslinking for rapid, transient interactions
Essential Research Reagent Solutions
Successful implementation of crosslinking and label transfer strategies requires access to specialized reagents and tools. The table below summarizes key research solutions for establishing these methodologies in the laboratory:
Table 3: Essential Research Reagents for Crosslinking and Label Transfer Studies
| Reagent/Tool | Supplier Examples | Application | Key Features |
|---|---|---|---|
| Sulfo-SBED | Thermo Scientific | Biotin-based label transfer | Trifunctional reagent with NHS-ester, aryl azide, and biotin [32] |
| BS³ / BS³-d4 | Thermo Scientific | Quantitative CLMS | Homobifunctional amine-reactive crosslinker with deuterated analogue [35] |
| MTS-Diazirine Reagents | Custom synthesis | Tag-transfer crosslinking | Cysteine-specific labeling with rapid photoactivation [34] |
| UV LED Platform | Custom assembly | Rapid crosslinking | High-intensity 365 nm irradiation for 10-second crosslinking [34] |
| Iodination Beads | Thermo Scientific | Radioactive labeling | Efficient iodine labeling while preventing oxidative damage [32] |
| Streptavidin Beads | Multiple suppliers | Prey purification | Affinity purification of biotinylated prey proteins [32] |
Troubleshooting and Technical Considerations
Despite its powerful applications, label transfer methodology presents several technical challenges that require careful consideration:
- Non-Specific Binding: Despite high specificity, label transfer can sometimes produce non-specific binding and misleading results. Appropriate controls are essential, including competition experiments with unlabeled bait protein [31].
- Complex Experimental Conditions: Success depends on meticulously controlled conditions, including light exposure intensity, incubation duration, and crosslinker choice. Deviations from optimal conditions can result in experimental failure [31].
- Low Abundance Prey Detection: For low abundance targets, the biotin-based system combined with streptavidin enrichment provides the sensitivity needed for detection [32].
- Reproducibility Challenges: Crosslinking is renowned for low reproducibility. Incorporating replicated analyses and label-swaps significantly improves quantification accuracy [35].
Recent advances in quantitative crosslinking/mass spectrometry have addressed many of these challenges through standardized workflows, improved data processing tools, and best practices that make these techniques accessible to researchers with limited initial expertise in crosslinking and quantitative proteomics [33]. The maturation of CLMS methodology and its fusion with quantitative proteomics now enables robust investigation of protein dynamics in solution at sufficient resolution to gain valuable biological insights [33].
Integration with Complementary Approaches
Crosslinking and label transfer strategies are most powerful when integrated with complementary structural and computational biology approaches. Recent advances in deep learning for protein-protein interaction prediction offer opportunities to combine experimental crosslinking data with computational models [1]. Graph neural networks (GNNs) based on graph structures and message passing can adeptly capture local patterns and global relationships in protein structures, providing predictive frameworks that complement experimental data [1].
Additionally, computational frameworks for analyzing higher-order interactions, such as protein triplets with cooperative or competitive relationships, can leverage experimental crosslinking data to validate and refine models of complex formation [36]. The integration of experimental crosslinking data with these computational approaches creates a powerful synergy for comprehensively mapping the dynamic protein interaction networks that govern cellular signaling pathways.
For drug discovery professionals, these integrated approaches provide unprecedented insights into the mechanisms of protein complex assembly and dynamics, enabling more targeted therapeutic interventions in pathological signaling pathways. The ability to capture transient interactions offers particular value for identifying allosteric binding sites and characterizing the mechanisms of action for small molecule inhibitors targeting protein-protein interactions.
Protein-protein interactions (PPIs) form the cornerstone of cellular signaling pathways, governing critical processes such as signal transduction, gene regulation, and metabolic homeostasis. Understanding these dynamic complexes requires analytical techniques capable of probing interactions in real-time without perturbing native biological states. This article details four key biophysical methods—Surface Plasmon Resonance (SPR), Förster Resonance Energy Transfer (FRET), Bio-Layer Interferometry (BLI), and Dynamic Light Scattering (DLS)—within the context of signaling pathway research. We provide comprehensive application notes, detailed experimental protocols, and practical implementation guidelines to support researchers in drug discovery and development.
Surface Plasmon Resonance (SPR)
Principle and Applications
SPR is a label-free optical technique that measures biomolecular interactions in real-time by detecting changes in the refractive index at a sensor surface [37]. When light excites surface plasmons in a thin metal layer (typically gold) under conditions of total internal reflection, the resonance angle is sensitive to mass changes at the surface, allowing direct monitoring of binding events [37] [38]. This enables determination of binding kinetics (association rate k_on, dissociation rate k_off), affinity (K_D), and concentration without requiring fluorescent or radioactive labels.
In signaling pathway research, SPR-based platforms like Biacore have been extensively applied to study interactions ranging from small ligands to whole cells [37]. Specific applications include receptor-ligand interactions, antibody-epitope mapping, kinase-substrate profiling, and transcription factor-DNA binding, providing critical insights into the kinetic parameters that govern signaling cascade dynamics and regulation [37] [38].
Experimental Protocol: Protein-Peptide Interaction Study
The following protocol adapts established SPR methodologies for studying signaling protein interactions, such as those between a purified protein receptor and its peptide ligand [39].
Materials and Reagents
- Instrument: Biacore X100 or comparable SPR system
- Sensor Chip: CM5 (carboxymethylated dextran matrix)
- Ligand: Purified signaling protein (>90% purity)
- Analyte: Synthetic peptide dissolved in DMSO
- Buffers:
- Immobilization buffer: 10 mM acetate buffer (pH 4.0-5.5)
- Running buffer: 10 mM HEPES (pH 7.5), 150 mM NaCl, 0.05% Tween-20
- Regeneration solution: 50 mM NaOH
- Coupling Reagents: Amine coupling kit (EDC, NHS, ethanolamine)
Procedure
A. pH Scouting for Ligand Immobilization
- Dilute the purified protein ligand to 5-200 μg/mL in 10 mM acetate buffers of varying pH (4.0, 4.5, 5.0, 5.5).
- Inject each sample over a separate flow cell for 1 minute at 10 μL/min.
- Identify the optimal pH that provides sufficient ligand concentration on the sensor surface (typically the highest pH yielding effective binding).
B. Ligand Immobilization via Amine Coupling
- Activate the carboxymethylated dextran surface with a 1:1 mixture of 0.4 M EDC and 0.1 M NHS for 7 minutes.
- Inject the protein ligand (diluted in optimal pH acetate buffer) over the activated surface for 10-15 minutes.
- Deactivate remaining active esters with 1 M ethanolamine-HCl (pH 8.5) for 7 minutes.
- One flow cell should remain unmodified to serve as a reference for background subtraction.
C. Analyte Binding Assay
- Prepare serial dilutions of the peptide analyte (0.39-12.5 μM) in running buffer containing 5% DMSO, precisely matching the DMSO concentration in all samples and running buffer.
- Inject analyte samples over both ligand and reference flow cells at 30 μL/min for 2-3 minutes association phase.
- Monitor dissociation in running buffer for 5-10 minutes.
- Regenerate the surface with a 30-second pulse of 50 mM NaOH between analyte cycles.
D. Data Analysis
- Subtract reference cell sensorgram from ligand cell sensorgram to account for bulk refractive index changes.
- Fit corrected sensorgrams to appropriate binding models (1:1 Langmuir, two-state, or bivalent) using Biacore Evaluation Software to extract
k_on,k_off, andK_Dvalues.
Figure 1: SPR Experimental Workflow. This diagram outlines the key steps in an SPR binding experiment, from surface preparation to data analysis.
Förster Resonance Energy Transfer (FRET)
Principle and Applications
FRET is a distance-dependent quantum mechanical phenomenon where energy transfers non-radiatively from an excited donor fluorophore to an acceptor chromophore through dipole-dipole coupling [40] [41]. FRET efficiency is inversely proportional to the sixth power of the distance between fluorophores, making it exceptionally sensitive to molecular proximity changes in the 1-10 nm range [41]. This characteristic enables researchers to monitor protein conformational changes, protein-protein interactions, and molecular clustering in real-time.
In signaling pathway analysis, FRET-based biosensors are particularly valuable for visualizing compartmentalized second messenger dynamics (cAMP, cGMP, Ca²⁺) and the spatiotemporal regulation of macromolecular complexes in live cells [40]. For instance, EPAC-based FRET sensors have revealed polarized cAMP accumulation at the leading edge of migrating fibroblasts, while PKG-based sensors have elucidated cGMP microdomains in cardiovascular signaling [40].
Experimental Protocol: Live-Cell Compartmentalized cAMP Signaling
Materials and Reagents
- FRET Biosensor: EPAC-based cAMP sensor (CFP-EPAC-YFP)
- Cell Line: Relevant signaling model (e.g., HEK293, primary fibroblasts)
- Imaging System: Inverted epifluorescence microscope with:
- CFP excitation (430-440 nm) and emission (465-495 nm) filters
- YFP excitation (500-520 nm) and emission (535-565 nm) filters
- Dual-emission imaging capability or filter wheel
- 40× or 60× oil-immersion objective
- Buffer: Physiological salt solution (e.g., Hanks' Balanced Salt Solution)
- Stimuli: Receptor agonists/antagonists relevant to pathway under study
Procedure
A. Cell Preparation and Transfection
- Plate cells onto poly-L-lysine-coated glass-bottom dishes 24-48 hours before imaging.
- Transfect with EPAC-FRET biosensor construct using appropriate method (lipofection, electroporation).
- Incubate for 24-48 hours to allow biosensor expression; serum-starve if required for pathway sensitivity.
B. FRET Imaging Acquisition
- Replace medium with imaging buffer and equilibrate cells at 37°C with 5% CO₂ for 30 minutes.
- Select fields with moderately expressing cells (avoid overexpressed artifacts).
- Acquire baseline images:
- CFP channel (donor): Ex 440 nm, Em 480 nm
- FRET channel (acceptor sensitized emission): Ex 440 nm, Em 535 nm
- YFP channel (acceptor direct excitation): Ex 515 nm, Em 535 nm
- Apply pathway stimulus (e.g., β-adrenergic agonist for cAMP elevation) and continue time-lapse acquisition every 30-60 seconds for 15-30 minutes.
C. Data Processing and Analysis
- Background subtract all images using cell-free regions.
- Calculate FRET ratio for each time point:
R = FRETchannel / CFPchannel - Normalize data as
ΔR/R₀ = (R - R₀) / R₀, where R₀ is baseline ratio. - Generate kinetic curves and compare across cellular compartments (membrane, cytosol, nuclear).
- Optional: Perform acceptor photobleaching controls to validate FRET efficiency.
Figure 2: FRET Experimental Workflow. This diagram illustrates the process for monitoring compartmentalized signaling in live cells using FRET biosensors.
Bio-Layer Interferometry (BLI)
Principle and Applications
BLI is a label-free optical technique that analyzes biomolecular interactions by measuring interference patterns of white light reflected from a biosensor tip [42] [43]. As molecules bind to the biosensor surface, the optical path length shifts, resulting in wavelength interference pattern changes measured in real-time [43]. This enables monitoring of binding kinetics and affinities without microfluidic systems, offering advantages in simplicity and versatility.
BLI has gained prominence in drug discovery for characterization of antibody-antigen interactions, receptor-ligand binding, and protein-nucleic acid complexes [42] [43]. Its "dip-and-read" format makes it particularly suitable for fragment-based screening, structure-activity relationship studies, and bioprocess monitoring during therapeutic antibody development [42]. A notable application includes analyzing carbohydrate-lectin binding specificity using streptavidin-coated tips with biotinylated glycans, providing kinetic parameters (K_D, k_on, k_off) for vaccine target validation [43].
Dynamic Light Scattering (DLS)
Principle and Applications
DLS (also known as photon correlation spectroscopy) measures Brownian motion of particles in solution by analyzing fluctuations in scattered laser light intensity [44]. The diffusion coefficient derived from these fluctuations enables calculation of hydrodynamic radius and size distribution through the Stokes-Einstein relationship. DLS provides information about protein hydrodynamic size, oligomeric state, aggregation propensity, and complex formation.
In signaling pathway research, DLS serves as a critical quality control tool for characterizing purified signaling proteins before functional studies [44]. Applications include monitoring protein complex assembly (e.g., ribonucleoprotein particles), detecting aggregates in macromolecular solutions, and analyzing ligand-induced size changes [44]. For students and researchers, DLS offers an accessible method for preliminary assessment of sample monodispersity and stability under various solution conditions.
Comparative Analysis of Biophysical Techniques
Table 1: Technical Specifications and Applications of Biophysical Methods
| Parameter | SPR | FRET | BLI | DLS |
|---|---|---|---|---|
| Measured Parameters | k_on, k_off, K_D, concentration |
Distance (1-10 nm), conformational changes, interaction proximity | k_on, k_off, K_D, concentration |
Hydrodynamic size, polydispersity, aggregation state |
| Throughput | Medium (multichannel systems available) | Low to medium (depends on imaging setup) | Medium to high (8-96 tips available) | High (rapid measurements) |
| Sample Consumption | Low (μg range) | Very low (single cells) | Low to medium | Medium (μg-mg range) |
| Label Requirement | Label-free | Requires dual fluorophore labeling | Label-free | Label-free |
| Key Applications in Signaling Research | Kinetic profiling of receptor-ligand interactions, epitope mapping, small molecule screening | Compartmentalized second messenger dynamics, conformational changes in live cells, protein complex assembly | Antibody characterization, fragment screening, protein-nucleic acid interactions | Protein complex size determination, aggregation monitoring, quality control |
| Information Depth | Kinetic and affinity data | Spatial and temporal resolution in living systems | Kinetic and affinity data | Hydrodynamic size and distribution |
Table 2: Advantages and Limitations in Signaling Pathway Studies
| Technique | Advantages | Limitations |
|---|---|---|
| SPR | Real-time kinetic data; label-free; sensitive to low molecular weight interactions; well-established data analysis methods [37] [38] | Requires immobilization; mass transport effects possible; limited throughput without advanced instrumentation |
| FRET | Single-cell resolution; subcellular compartmentalization; compatible with live-cell imaging; extremely distance-sensitive [40] [41] | Requires genetic engineering; photobleaching potential; spectral overlap challenges; quantitative interpretation complex |
| BLI | No microfluidics; minimal system maintenance; suitable for crude samples; higher throughput with tip-based format [42] [43] | Lower sensitivity than SPR in some systems; larger sample volume requirements; diffusion-limited kinetics |
| DLS | Rapid measurement; minimal sample preparation; non-destructive; measures polydispersity and aggregation [44] | Low resolution for heterogeneous mixtures; limited to size measurements; insensitive to small binding events |
Research Reagent Solutions
Table 3: Essential Materials for Biophysical Interaction Studies
| Reagent/Category | Specific Examples | Function in Experimental Design |
|---|---|---|
| Sensor Surfaces | CM5 chip (SPR), Streptavidin tips (BLI), HPA chip (membrane interactions) | Provides immobilization platform with specific coupling chemistries for different biomolecular classes [39] [38] |
| Labeling Systems | CFP/YFP FRET pairs, HaloTag, SNAP-tag | Enables site-specific fluorophore incorporation for FRET studies with minimal perturbation to protein function [40] |
| Immobilization Reagents | Amine coupling kit (EDC/NHS), biotinylation reagents, His-tag/NTA systems | Facilitates controlled attachment of biomolecules to sensor surfaces while preserving biological activity [39] |
| Reference Proteins | Stable signaling proteins (e.g., BSA, well-characterized antibodies) | Serves as controls for immobilization efficiency and background binding assessment |
| Buffer Components | HEPES, Tween-20, DMSO-compatible buffers | Maintains physiological pH and ionic strength while minimizing non-specific binding [39] |
The integrated application of SPR, FRET, BLI, and DLS provides a comprehensive biophysical toolkit for elucidating protein-protein interactions in signaling pathways. SPR excels in detailed kinetic analysis, FRET offers unparalleled spatiotemporal resolution in living cells, BLI provides robust interaction screening, and DLS ensures sample quality and complex integrity. The selection of appropriate technique(s) should be guided by specific research questions, sample availability, and required information depth. As signaling pathway complexity continues to emerge, these biophysical approaches will remain indispensable for mechanistic studies and therapeutic development in biomedical research.
Protein-protein interactions (PPIs) are fundamental to virtually all cellular processes, guiding signal transduction, regulating gene expression, and ensuring the coordinated functioning of biological pathways. The dysregulation of these interactions underpins numerous diseases, from cancer to neurodegeneration, making them attractive targets for therapeutic intervention [18]. For researchers investigating signaling pathways, the ability to accurately capture and quantify these interactions is paramount. Traditional methods for studying PPIs, including binding assays and proximity-based methods, often face limitations in capturing transient interactions or require complex instrumentation. Next-generation functional assays have emerged to address these challenges, offering enhanced sensitivity, physiological relevance, and compatibility with high-throughput screening. This article focuses on the LinkLight technology as a representative advanced platform and situates it within the broader landscape of innovative PPI analysis tools, providing detailed application notes and protocols for researchers and drug development professionals.
LinkLight Technology: Core Mechanism and Advantages
The LinkLight assay is a proprietary, cell-based technology that provides innovative tools for detecting protein-protein interactions with high specificity and sensitivity. This platform stands out for its ability to convert transient biological interactions into stable, measurable luminescent signals, making it particularly valuable for studying dynamic signaling events [45] [46].
Mechanism of Action
The LinkLight assay employs a sophisticated molecular design consisting of two key components:
- Protein A is fused to a Tobacco Etch Virus (TEV) protease.
- Protein B is fused to a permuted luciferase (pLuc) reporter.
The permuted luciferase has been engineered to be inactive in its native state through structural rearrangement: the N-terminal and C-terminal fragments of luciferase have been swapped and reconnected via a peptide linker containing a TEV protease cleavage sequence [45] [46]. When Proteins A and B interact, the TEV protease is brought into proximity with the cleavage site on the permuted luciferase. Proteolytic cleavage at this site allows the luciferase fragments to spontaneously refold into an active conformation, driven by fragment self-complementation affinity. The reconstituted active luciferase then generates a luminescent signal in the presence of its substrate, luciferin [46]. This cleavage event is irreversible, meaning the signal persists even after the proteins dissociate, creating a "molecular memory" of transient interactions [18].
Key Advantages Over Traditional Methods
LinkLight technology offers several distinct advantages that make it particularly suitable for signaling pathway research:
Specificity: Only tagged receptors generate signals, preventing interference from endogenous receptors or receptor family members [45]. This specificity is crucial for studying specific signaling cascades without background noise.
Sensitivity: Bioluminescent signals are more sensitive than fluorescence-based methods, enabling detection of weak or transient interactions [45].
Transcription-Independent Signaling: Unlike reporter gene assays, LinkLight does not involve transcription or translation signal cascades, thereby reducing false and off-target signals and providing immediate readouts [45].
Irreversible Signal Generation: The TEV cleavage event is irreversible, meaning the signal persists even after proteins dissociate, allowing detection of fleeting interactions [46] [18].
Physiological Relevance: The cell-based format preserves native cellular context and signaling machinery, providing biologically relevant data [46].
Robotic Adaptability: The homogenous luminescent readout is simple to operate and readily adaptable to high-throughput screening platforms [45].
Compared to other technologies like BRET, LinkLight avoids limitations related to spectrum separation, spatial distance and orientation requirements between donor and acceptor molecules, light intensity concerns, donor/acceptor ratio optimization, and inability to establish stable cell line assays [45].
Comparative Analysis of Next-Generation PPI Platforms
While LinkLight represents a significant advancement in functional PPI assays, other innovative platforms have emerged with complementary strengths. The table below provides a comparative analysis of current technologies.
Table 1: Comparative Analysis of Protein-Protein Interaction Assay Platforms
| Technology | Mechanism | Key Applications | Sensitivity | Throughput | Spatial Resolution |
|---|---|---|---|---|---|
| LinkLight | TEV protease cleavage of permuted luciferase | GPCR signaling, β-arrestin recruitment, transient PPIs | High (bioluminescence) | High (robotic adaptable) | Cellular level |
| Spatial Protein Proximity (Bio-Techne) | RNAscope-based in situ detection | Spatial visualization of protein interactions in intact tissues | High | Moderate | Subcellular |
| PLIP 2025 | Computational analysis of molecular interactions | Structural PPI analysis, drug interaction profiling | N/A | High (computational) | Atomic |
| PLM-interact | Protein language model AI prediction | Cross-species PPI prediction, mutation effect analysis | N/A | Very High (computational) | Sequence level |
Specialized Platforms for Specific Research Needs
Spatial Protein Proximity Detection: Bio-Techne's recently announced (2025) spatial proximity assay builds upon RNAscope technology to enable high-resolution visualization of protein interactions within intact tissues. This technology addresses the limitation of conventional methods that lose spatial fidelity, providing subcellular resolution for understanding how molecular signaling shapes disease processes in tissue context [47]. This is particularly valuable for research areas where context matters, such as immune checkpoint dynamics, bispecific antibody investigations, and synaptic protein interactions [47].
Computational Prediction Tools: PLM-interact represents a breakthrough in AI-driven PPI prediction. This method extends protein language models (PLMs) by jointly encoding protein pairs to learn their relationships, analogous to next-sentence prediction in natural language processing. The system achieves state-of-the-art performance in cross-species PPI prediction and can detect mutation effects on interactions [48]. Such computational approaches complement experimental methods by enabling large-scale interaction mapping and predictive modeling.
Protein-Ligand Interaction Profiler (PLIP): The 2025 update to PLIP now incorporates protein-protein interaction analysis alongside its traditional small-molecule focus. This tool analyzes molecular interactions in protein structures, detecting eight types of non-covalent interactions [49]. It is particularly valuable for understanding how therapeutic compounds like the cancer drug venetoclax mimic native protein interactions [49].
LinkLight Applications in Signaling Pathway Research
The versatility of LinkLight technology enables its application across diverse signaling pathway research areas. The platform's ability to capture transient interactions makes it particularly valuable for studying dynamic signaling events.
GPCR Signaling and β-Arrestin Recruitment
G-protein coupled receptors (GPCRs) represent approximately 35% of all FDA-approved drug targets, making them critically important in pharmaceutical research [18]. LinkLight provides a powerful approach for comprehensive GPCR characterization:
β-Arrestin Recruitment: LinkLight cell lines engineered with GPCRs fused to TEV protease and β-arrestin fused to permuted luciferase enable detection of β-arrestin recruitment upon receptor activation [46]. This is particularly valuable as β-arrestin recruitment is a broadly conserved pathway across most GPCR families, making it ideal for receptors with unknown coupling patterns [46].
Ligand Bias Profiling: By combining LinkLight β-arrestin data with secondary messenger assays (cAMP, Ca²⁺), researchers can identify ligands that preferentially activate specific signaling pathways (biased agonism) [46] [18]. This functional selectivity has important implications for drug development with improved therapeutic profiles.
14-3-3 Interactions: LinkLight can detect interactions with 14-3-3 scaffold proteins, which play important roles in signal stabilization downstream of GPCR activation [46].
Table 2: LinkLight Applications in Disease Research and Representative Targets
| Research Area | Signaling Pathways | Example Targets | Research Applications |
|---|---|---|---|
| Oncology & Immuno-Oncology | Chemokine signaling, tumor progression | CXCR4, CXCR7, adenosine receptors (A2A) | Investigate mechanisms of cancer spread, enhance anti-tumor immunity |
| Neuroscience | Neurotransmitter signaling | Dopamine, serotonin, glutamate receptors | CNS drug discovery, receptor signaling dynamics |
| Autoimmune Diseases | Immune regulation | Sphingosine-1-phosphate receptors | Develop therapies for multiple sclerosis, rheumatoid arthritis |
| Metabolic Disorders | Metabolic regulation | GLP-1, GIP, glucagon receptors | Therapeutic strategies for diabetes, obesity |
| Musculoskeletal Health | Bone and muscle regulation | Parathyroid hormone receptors | Identify targets for osteoporosis, muscular dystrophy |
Protocol: GPCR β-Arrestin Recruitment Assay Using LinkLight
Principle: This protocol detects β-arrestin recruitment to activated GPCRs using TEV protease-mediated luciferase activation in a live-cell format.
Materials:
- LinkLight cell line expressing GPCR-TEV and β-arrestin-pLuc fusions
- White-walled, clear-bottom 96- or 384-well microplates
- Ligand compounds for testing
- Luciferin substrate solution
- Cell culture medium (serum-free for stimulation)
- Luminescence plate reader
Procedure:
Cell Preparation:
- Thaw and culture LinkLight cells expressing your GPCR of interest according to provider specifications.
- Harvest cells at 80-90% confluence using gentle detachment methods.
- Resuspend cells in serum-free assay medium at a density of 0.5-1.0 × 10⁶ cells/mL.
Plate Seeding:
- Dispense 80-100 μL cell suspension per well in white-walled, clear-bottom microplates.
- Incubate plates at 37°C, 5% CO₂ for 16-24 hours to achieve 90-95% confluence.
Compound Treatment:
- Prepare serial dilutions of test ligands in serum-free assay medium.
- Remove plates from incubator and equilibrate to room temperature for 15 minutes.
- Add 10-20 μL compound solutions to cells, achieving desired final concentrations.
- Include controls: vehicle (negative control) and reference agonist (positive control).
- Incubate at room temperature for 60-90 minutes (or optimized timeframe).
Signal Detection:
- Add luciferin substrate to a final concentration of 100-200 μM.
- Incubate for 3-5 minutes to allow signal stabilization.
- Measure luminescence using a plate reader with integration time of 0.5-1 second/well.
Data Analysis:
- Calculate fold-change over vehicle control.
- Generate dose-response curves using nonlinear regression.
- Determine EC₅₀ values for agonist potency.
Technical Notes:
- Optimal assay conditions may vary by receptor; perform initial time-course and concentration-gradient experiments.
- For antagonist studies, pre-incubate with test compounds for 30 minutes before agonist addition.
- The persistent signal allows flexible read times from 30 minutes to several hours post-stimulation.
Research Reagent Solutions
Successful implementation of next-generation PPI assays requires specific reagents and tools. The following table outlines essential components for establishing LinkLight and related technologies in the research laboratory.
Table 3: Essential Research Reagents for Next-Generation PPI Assays
| Reagent/Cell Line | Function | Examples/Specifications |
|---|---|---|
| LinkLight GPCR Cell Lines | Engineered cells for specific PPI detection | 100+ validated GPCR/β-arrestin cell lines covering major receptor classes [46] |
| Permuted Luciferase Reporters | Signal generation upon cleavage | Firefly luciferase, Renilla luciferase, or β-lactamase permutations [45] |
| TEV Protease Fusion Vectors | Molecular component for interaction-dependent cleavage | Customizable expression vectors for protein-TEV fusions |
| Luciferin Substrate | Luciferase enzyme substrate | Commercial preparations optimized for sensitivity and stability [45] |
| Spatial Biology Detection Kits | Tissue-based PPI visualization | RNAscope-compatible protein proximity detection [47] |
| Computational Prediction Tools | In silico PPI analysis | PLIP web server, PLM-interact models [49] [48] |
Integration with Signaling Pathway Analysis Methods
LinkLight and complementary technologies generate the most value when integrated with broader signaling pathway analysis frameworks. Pathway analysis methodologies provide systems-level understanding by coupling high-throughput biological data with existing biological knowledge from databases, statistical testing, and computational algorithms [50].
Pathway Analysis Workflow Integration
The diagram below illustrates how LinkLight data can be integrated into a comprehensive pathway analysis workflow for signaling research.
Gene Set Variation Analysis (GSVA) and similar pathway analysis methods can incorporate LinkLight PPI data to identify enriched signaling pathways in different physiological or disease states [51]. For example, in major depressive disorder research, pathway analysis has revealed alterations in killing signaling pathways and immune infiltration patterns [51]. Similarly, ingenuity pathway analysis (IPA) of α-synuclein has identified neuroinflammation, Huntington's disease signaling, TREM1, and phagosome maturation as key canonical pathways in neurodegeneration [52].
Protocol: Integrating LinkLight Data with Pathway Analysis
Principle: This protocol describes a bioinformatics workflow for contextualizing LinkLight PPI data within broader signaling networks.
Materials:
- LinkLight experimental results (interaction scores or quantitative measurements)
- Pathway analysis software (IPA, clusterProfiler, GSEA)
- Reference databases (KEGG, Reactome, Gene Ontology)
- Statistical computing environment (R, Python)
Procedure:
Data Preparation:
- Compile significant PPIs identified through LinkLight screening.
- Convert protein identifiers to standardized format (e.g., UniProt IDs).
- Prepare background gene set appropriate for your experimental context.
Overrepresentation Analysis:
- Use Fisher's exact test or hypergeometric test to identify enriched pathways.
- Apply multiple testing correction (Benjamini-Hochberg FDR < 0.05).
- Visualize results using bar plots or dot plots of significant pathways.
Gene Set Enrichment Analysis (GSEA):
- Rank proteins based on LinkLight interaction metrics (e.g., fold-change).
- Perform pre-ranked GSEA against pathway databases.
- Identify pathways enriched at top or bottom of ranked list.
Network Visualization:
- Construct protein interaction networks using significant hits.
- Integrate with known pathway components from reference databases.
- Identify hub proteins and functional modules within networks.
Interpretation:
- Contextualize LinkLight findings within enriched pathways.
- Generate hypotheses about signaling mechanisms.
- Design follow-up experiments for pathway validation.
Technical Notes:
- For comprehensive analysis, integrate LinkLight data with complementary omics datasets.
- Use tools like PLIP [49] or PLM-interact [48] for structural insights into significant interactions.
- Consider tissue-specific or context-specific pathway databases when available.
Next-generation functional assays like LinkLight represent significant advancements in our ability to study protein-protein interactions within signaling pathways. By providing sensitive, specific, and physiologically relevant detection of even transient interactions, these technologies enable deeper understanding of signaling mechanisms in health and disease. The integration of experimental platforms like LinkLight with computational prediction tools and pathway analysis frameworks creates a powerful ecosystem for comprehensive signaling research. As these technologies continue to evolve, they will undoubtedly accelerate drug discovery and enhance our understanding of complex biological systems. Researchers are encouraged to select platforms based on their specific applications—LinkLight for functional cell-based studies of dynamic interactions, spatial methods for tissue context, and computational tools for large-scale prediction and modeling.
Protein-protein interactions (PPIs) are fundamental regulators of cellular functions, influencing a variety of biological processes including signal transduction, cell cycle regulation, and transcriptional regulation [1]. Understanding these interactions is crucial for elucidating the mechanisms of signaling pathways and for drug discovery. Traditional experimental methods for identifying PPIs, such as yeast two-hybrid screening and co-immunoprecipitation, are often time-consuming and resource-intensive [1]. The advent of deep learning, a cornerstone of artificial intelligence, has transformed computational PPI prediction by enabling automatic feature extraction from protein sequences and structures, offering unprecedented levels of accuracy and efficiency [1] [53]. Core deep learning architectures such as Graph Neural Networks (GNNs), Convolutional Neural Networks (CNNs), and Transformers have emerged as powerful tools for tackling various PPI tasks. These include interaction prediction, interaction site identification, and cross-species interaction prediction [1]. This article provides a detailed analysis of these architectures, their applications, and protocols tailored for signaling pathway research.
Core Deep Learning Architectures and Their Applications
Graph Neural Networks (GNNs)
GNNs are particularly suited for PPI prediction due to their ability to model graph-structured data, such as protein structures and interaction networks. By aggregating information from neighboring nodes, GNNs generate node representations that reveal complex interactions and spatial dependencies in proteins [1].
- Key Variants: Principal GNN architectures include Graph Convolutional Networks (GCNs), Graph Attention Networks (GATs), GraphSAGE, and Graph Autoencoders (GAEs) [1].
- Application in Signaling Pathways: GNNs can model the dynamic nature of signaling networks. For instance, a bioreaction–variation network, a GNN model, has been developed to infer hidden molecular and physiological relationships underlying interindividual variation in responses to physiological stimuli. This model uses a multi-head attention mechanism to capture both local topological features and directional dominance between connected nodes, making it possible to identify individualized signaling pathways from experimental data like gene expression changes [54].
- Specific Model - DCMF-PPI: This hybrid framework integrates a GAT with a protein language model (PortT5) to capture context-aware structural variations in protein interactions. It is specifically designed to address the dynamic nature of proteins and PPI structures during cellular processes [55].
Convolutional Neural Networks (CNNs)
CNNs are primarily used to extract hierarchical and spatial features from data, making them well-suited for processing protein sequences and structural images.
- Feature Extraction: CNNs can automatically learn relevant features from amino acid sequences, such as local motifs and patterns that are indicative of interaction interfaces [1] [53].
- Multi-Scale Analysis: In frameworks like DCMF-PPI, parallel CNNs are combined with wavelet transforms (in a module named MPSWA) to extract multi-scale features from diverse protein residue types and their dynamic coordinate changes, enhancing the representation of sequence and structural heterogeneity [55].
Transformers and Protein Language Models (PLMs)
Transformer models, with their self-attention mechanisms, excel at capturing long-range dependencies in sequential data. This capability is highly valuable for protein informatics, where sequence-structure-function relationships often hinge on distal interactions [56].
- Self-Attention Mechanism: The core innovation of the Transformer model, self-attention dynamically models pairwise relevance between all elements in a sequence (e.g., amino acids), enabling the model to capture intricate intra-sequence dependencies critical for understanding protein function [56].
- Protein Language Models (PLMs): Models like ESM-2 and ProtT5 are pre-trained on large corpora of protein sequences. They learn rich, contextualized representations of amino acids, capturing evolutionary, structural, and functional information [55] [57].
- Advanced Application - PLM-interact: This approach goes beyond using pre-trained PLMs as static feature extractors. It fine-tunes a PLM (ESM-2) jointly on protein pairs, analogous to the next-sentence prediction task in NLP. This allows the model to directly learn the relationships between interacting proteins, leading to state-of-the-art performance in cross-species and virus-host PPI prediction, as well as in predicting the effects of mutations on interactions [57].
Table 1: Summary of Core Deep Learning Models in PPI Prediction
| Architecture | Core Function | Key Applications in PPI | Exemplary Models |
|---|---|---|---|
| Graph Neural Network (GNN) | Models graph-structured data and node relationships. | PPI network analysis, dynamic interaction prediction, residue-level interaction mapping. | DCMF-PPI [55], Bioreaction-Variation Network [54] |
| Convolutional Neural Network (CNN) | Extracts local and hierarchical spatial features. | Sequence motif detection, protein interface prediction, multi-scale feature fusion. | DeepPPI [55], DCMF-PPI (MPSWA module) [55] |
| Transformer / PLM | Captures long-range dependencies in sequences. | Protein representation learning, cross-species PPI prediction, mutation effect analysis. | PLM-interact [57], ESM-2 [57], ProtT5 [55] |
Quantitative Performance Comparison
Benchmarking studies on cross-species PPI prediction demonstrate the evolving performance of deep learning models. The following table summarizes the Area Under the Precision-Recall Curve (AUPR) for several state-of-the-art methods when trained on human data and tested on other species.
Table 2: Cross-Species PPI Prediction Performance (AUPR)
| Model | Mouse | Fly | Worm | Yeast | E. coli |
|---|---|---|---|---|---|
| PLM-interact | 0.945 | 0.852 | 0.881 | 0.706 | 0.722 |
| TUnA | 0.925 | 0.772 | 0.821 | 0.641 | 0.655 |
| TT3D | 0.785 | 0.642 | 0.681 | 0.553 | 0.605 |
| D-SCRIPT | 0.659 | 0.499 | 0.523 | 0.387 | 0.415 |
| PIPR | 0.645 | 0.456 | 0.481 | 0.351 | 0.378 |
| DeepPPI | 0.632 | 0.441 | 0.467 | 0.343 | 0.369 |
Data adapted from benchmarking in [57]. AUPR values are estimated from graphical data for illustrative purposes. PLM-interact shows consistent improvements, particularly on evolutionarily distant species.
Application Notes and Protocols
Protocol 1: Cross-Species PPI Prediction using a Fine-Tuned PLM
This protocol describes the use of PLM-interact for predicting interactions in a new species using a model trained on human data, which is valuable for studying conserved signaling pathways.
- Objective: To predict binary protein-protein interactions for a target species (e.g., mouse, fly) using a deep learning model trained on human PPI data.
- Input Data Preparation:
- Protein Pairs: Compile a list of candidate protein pairs for the target species.
- Sequence Format: For each protein, obtain its amino acid sequence in FASTA format. Ensure sequences are from a standardized database like UniProt for consistency.
- Model Inference:
- Load Model: Initialize the PLM-interact model with pre-trained weights (e.g., the version based on ESM-2 with 650M parameters) [57].
- Sequence Processing: The model jointly tokenizes and processes the two protein sequences in a pair.
- Prediction: For each protein pair, the model outputs a probability score between 0 and 1, representing the likelihood of interaction.
- Output and Analysis:
- Interaction Score: A probability score threshold (e.g., 0.5) can be used to classify pairs as interacting or non-interacting. The score can also be treated as a confidence measure.
- Validation: Where possible, validate high-confidence novel predictions with experimental data or through literature mining.
Protocol 2: Predicting Mutation Effects on Signaling PPIs
This protocol uses a fine-tuned PLM to assess how point mutations might disrupt or enhance specific PPIs critical to a signaling pathway.
- Objective: To quantify the effect of a single-point mutation on the strength of a known protein-protein interaction.
- Input Data:
- Wild-type Protein Pair: The amino acid sequences of the two interacting proteins (A and B).
- Mutant Protein: The sequence of protein A containing a specific point mutation (e.g., A_mutant).
- Experimental Procedure:
- Wild-type Prediction: Run PLM-interact with the wild-type pair (A, B) to obtain a baseline interaction probability, P_wt.
- Mutant Prediction: Run PLM-interact with the mutant pair (Amutant, B) to obtain the mutant interaction probability, Pmut.
- Calculate Effect: The mutation effect (ΔP) is calculated as: ΔP = Pmut - Pwt. A negative ΔP suggests a disruptive mutation, while a positive ΔP suggests a enhancing mutation [57].
- Output and Analysis:
- Effect Size: Report ΔP as a quantitative measure of the mutation's impact.
- Pathway Implications: Interpret the result in the context of the signaling pathway. For example, a disruptive mutation in a key receptor-ligand interaction could indicate a potential mechanism for pathway dysregulation.
Protocol 3: Modeling Dynamic PPIs with a GNN Framework
This protocol outlines the use of dynamic GNN-based models like DCMF-PPI to account for protein structural variations in PPI networks.
- Objective: To predict PPIs while incorporating the dynamic structural states of proteins.
- Input Data Preparation:
- Protein Sequences: Obtain amino acid sequences for all proteins of interest.
- Structural Dynamics: Generate dynamic information for proteins with known structures. This can be achieved using tools like Normal Mode Analysis (NMA) and the Elastic Network Model (ENM) to simulate protein motion and produce temporal adjacency matrices representing different active states [55].
- Feature Extraction: Use a protein language model (e.g., PortT5) to generate residue-level embeddings for each protein [55].
- Model Inference with DCMF-PPI:
- The framework processes data through two parallel branches:
- PortT5-GAT Branch: Integrates residue-level features with dynamic temporal dependencies using a Graph Attention Network (GAT).
- MPSWA Branch: Uses CNNs and wavelet transforms to extract multi-scale features from dynamic residue coordinates.
- An adaptive gating mechanism fuses the features from both branches.
- A Variational Graph Autoencoder (VGAE) learns probabilistic latent representations to model the dynamic evolution of the PPI network [55].
- The framework processes data through two parallel branches:
- Output and Analysis:
- The model outputs a probabilistic prediction for each protein pair interaction.
- The integrated dynamic features allow the model to capture context-dependent interactions that may be transient or condition-specific, providing a more realistic view of PPIs in cellular signaling.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Databases and Tools for Deep Learning-Based PPI Prediction
| Resource Name | Type | Function in PPI Research |
|---|---|---|
| STRING | Database | Repository of known and predicted PPIs, useful for training and validation [1]. |
| BioGRID | Database | Database of protein and genetic interactions from high-throughput studies [1]. |
| IntAct | Database | Protein interaction database and analysis toolset, also provides mutation data [1] [57]. |
| PDB | Database | Source for 3D protein structures, essential for structure-based methods and dynamics simulation [1]. |
| ESM-2 | Protein Language Model | Pre-trained transformer model for generating state-of-the-art protein sequence representations [57]. |
| ProtT5 | Protein Language Model | Transformer-based PLM used to generate residue-level embeddings for protein sequences [55]. |
| AlphaFold2/3 | Structure Prediction | Provides highly accurate protein structure predictions, which can be used as input for structure-based PPI models [1] [57]. |
| PyTorch Geometric | Library | A library for deep learning on graphs, commonly used to implement GNN models for PPI [54]. |
Workflow and Pathway Visualizations
PPI Prediction with a Transformer (PLM-interact)
Dynamic PPI Prediction with a GNN Framework
Integrating PPI Prediction in Signaling Pathway Analysis
Solving Experimental Challenges: A Troubleshooting Guide for Reliable PPI Data
Combating False Positives and Negatives in Co-IP and Pull-Down Assays
In the analysis of cellular signaling pathways, protein-protein interactions (PPIs) serve as fundamental regulatory mechanisms governing cell fate, proliferation, and response to extracellular stimuli. Co-immunoprecipitation (Co-IP) and pull-down assays represent cornerstone techniques for validating and characterizing these interactions, yet their utility is frequently compromised by false positives (spurious interactions) and false negatives (undetected true interactions). These artifacts can significantly distort our understanding of signaling networks and lead to erroneous conclusions in both basic research and drug discovery pipelines. Estimates from proteomic studies suggest that false-positive rates in high-throughput interaction screens can reach 25-45%, while false-negative rates may be as high as 75-90% [58]. For researchers investigating signaling pathways, these inaccuracies present a substantial barrier to generating reliable, physiologically relevant models of cellular communication. This application note provides a detailed framework of strategic and technical considerations to mitigate these challenges, enhancing the reliability of Co-IP and pull-down data within the broader context of signaling pathway analysis.
Accurate interpretation of Co-IP and pull-down results requires a thorough understanding of potential error sources. False positives and negatives arise from distinct methodological pitfalls, which are summarized in Table 1 below.
Table 1: Common Sources of False Positives and Negatives in Co-IP and Pull-Down Assays
| Error Type | Specific Source | Impact on Data |
|---|---|---|
| False Positives | Non-specific antibody binding [59] | Detection of interactions not occurring in vivo |
| Protein "stickiness" (promiscuous interactions) [58] | Non-physiological associations with abundant proteins | |
| Incomplete washing of beads [59] [60] | Co-precipitation of non-specifically bound proteins | |
| Overexpression of bait protein [61] | Ectopic, non-physiological interactions due to aberrant stoichiometry | |
| Antibody masking (target comigrates with antibody chains) [59] | Obscured detection of true interactions | |
| False Negatives | Transient or weak interaction affinity [62] [60] | Failure to capture biologically relevant but labile complexes |
| Non-physiological lysis conditions [59] [60] | Disruption of native protein complexes | |
| Inefficient pull-down due to poor antibody affinity [59] | Incomplete isolation of the bait and its partners | |
| Epitope masking in native complex [59] | Antibody cannot access its binding site on the properly folded bait | |
| Interaction stability during wash steps [62] | Dissociation of genuine complexes under stringent conditions |
The following diagram illustrates how these error sources are introduced at various stages of a typical Co-IP workflow, providing a logical framework for implementing corrective measures.
Diagram: Error Introduction Points in Co-IP Workflow
Optimized Protocols for Robust PPI Detection
Co-Immunoprecipitation Under Native Conditions
This protocol is designed to preserve transient interactions in signaling complexes, which are crucial for accurate pathway mapping.
Sample Preparation and Lysis
- Lysis Buffer Formulation: Use a non-denaturing lysis buffer such as PBS or Tris-HCl (pH 7.4) containing a mild non-ionic detergent (e.g., 0.1-1% Triton X-100 or NP-40) [60]. The optimal detergent concentration and type should be empirically determined for your specific bait protein, particularly for membrane-associated signaling proteins.
- Inhibitor Cocktails: Supplement the buffer with fresh protease and phosphatase inhibitors to preserve post-translational modifications that often regulate signaling interactions [59]. Avoid repeated freeze-thaw cycles of lysates, as this can disrupt labile complexes.
Pre-Clearing and Immunoprecipitation
- Pre-Clearing: Incubate the cell lysate with the bead matrix (e.g., Protein A/G) alone for 30-60 minutes at 4°C. This step reduces non-specific binding [59].
- Antibody-Bead Complex Formation: For most applications, incubate the antibody with the bead matrix for 1 hour at 4°C before adding the pre-cleared lysate. This can improve capture efficiency compared to adding free antibody directly to the lysate.
- Incubation: Incubate the lysate with the antibody-bead complex for 2-4 hours to overnight at 4°C with gentle agitation. Longer incubation times may increase yield but also risk non-specific associations.
Washing and Elution
- Wash Buffer Stringency: Perform 3-4 washes with 10-20 bead volumes of lysis buffer. For interactions with known high affinity, a single wash with a buffer containing 150-500 mM NaCl can reduce non-specific binding without disrupting the complex [59] [60].
- Elution Methods: For downstream Western blot analysis, elute proteins by boiling beads in 1X SDS-PAGE sample buffer for 5-10 minutes. For functional assays, gentle elution methods such as competitive elution with a peptide corresponding to the antibody's epitope can be used to preserve complex integrity [60].
GST Pull-Down Assay for Direct Interaction Mapping
GST pull-downs are ideal for confirming direct binary interactions identified from Co-IP screens or for mapping specific interaction domains within a signaling protein.
Protein Preparation
- Expression and Purification: Express the GST-tagged bait protein in an appropriate system (e.g., E. coli). Purify using glutathione-sepharose beads under native conditions. Ensure the purified bait is free of major contaminants or degraded fragments.
- Prey Protein Source: The "prey" protein can be in vitro transcribed/translated (IVTT) with a detectable tag (e.g., 35S-methionine labeling, HA-tag), or it can be sourced from a relevant cell lysate.
Binding Reaction
- Blocking: Pre-block the glutathione beads with 1-5% BSA in binding buffer for 30 minutes to reduce non-specific binding.
- Incubation: Immobilize the purified GST-bait protein on the beads. Then, incubate with the prey protein in a binding buffer (e.g., 50 mM Tris pH 7.5, 150 mM NaCl, 0.1% NP-40, 1 mM DTT) for 1-2 hours at 4°C.
- Critical Controls: Always include a control with GST-alone protein incubated with the prey to identify interactions that are mediated by the GST tag or the beads themselves.
Washing and Analysis
- Wash beads 3-4 times with 1 mL of binding buffer. The stringency can be adjusted by increasing the salt concentration (up to 300 mM NaCl) or adding 0.1% SDS to the final wash for challenging prey proteins.
- Elute by boiling in SDS-PAGE sample buffer and analyze by Western blot or autoradiography.
The Scientist's Toolkit: Essential Reagents and Controls
The reliability of interaction data is heavily dependent on the quality of reagents and the inclusion of proper controls. Table 2 below details key components of a robust Co-IP or pull-down experiment.
Table 2: Research Reagent Solutions and Experimental Controls for Reliable PPI Studies
| Reagent / Control | Function & Importance | Technical Considerations |
|---|---|---|
| High-Specificity Antibodies [59] | Recognizes bait protein with minimal cross-reactivity; foundational for Co-IP. | Select antibodies validated for IP/Co-IP. Monoclonal antibodies often offer higher specificity. |
| Protein A/G Beads [59] [60] | Binds antibody Fc region to immobilize the bait-prey complex. | Choose based on host species of antibody (e.g., Protein A for rabbit IgG). |
| Magnetic Beads [59] [60] | Facilitate rapid separation with minimal mechanical loss. | Ideal for high-throughput applications or when working with low-abundance proteins. |
| Protease/Phosphatase Inhibitors [59] | Preserves protein integrity and signaling-relevant PTMs during lysis. | Use fresh cocktails; specific inhibitors may be needed for certain pathways (e.g., kinases). |
| Negative Control IgG [59] | Distracts non-specific binders; baseline for signal interpretation. | Use species- and isotype-matched antibodies from pre-immune serum or non-specific targets. |
| Bait Knockout/Knockdown Lysate [61] | Gold-standard control for antibody specificity. | Confirms that "prey" detection depends on the presence of the bait protein. |
| Reverse Co-IP [60] | Validates complex formation; tests reciprocity. | Immunoprecipitate the "prey" protein and blot for the "bait" protein. |
| Competition Assay [60] | Confirms interaction specificity. | Pre-incubate antibody with excess free antigen (epitope peptide); should abolish pull-down. |
Advanced Strategies for Challenging Interactions
Stabilizing Transient Interactions
Weak or transient interactions, common in dynamic signaling pathways, often require stabilization for detection.
- Chemical Crosslinking: Incorporate crosslinkers like dithiobis(succinimidyl propionate) (DSP) or BS³ into the protocol before lysis to covalently stabilize interactions [60]. Crosslinking conditions (concentration, duration) must be optimized to balance stabilization with the introduction of artifacts.
- Crosslinking Enhanced Co-IP: This variant uses reversible crosslinkers to stabilize weak complexes, allowing for more stringent wash conditions that would otherwise dissociate the interaction [60].
Integration with Orthogonal Methods
No single assay can unequivocally confirm a PPI. Corroborating Co-IP/pull-down data with orthogonal techniques is essential for building confidence.
- Proximity Ligation Assay (PLA): Allows for in situ visualization of PPIs with high specificity and sensitivity, confirming the interaction in a cellular context [61].
- Bioinformatic Corroboration: Leverage the growing power of deep learning-based PPI prediction tools (e.g., RoseTTAFold2-Lite) to assess the plausibility of a detected interaction within known biological networks [63].
- Combined Assay Platforms: Utilize novel combined methods like LuTHy, which integrates a proximity-based readout (BRET) with a co-purification readout (LUMIER) in a single system, providing two independent lines of evidence for an interaction [62].
The meticulous application of the optimized protocols, stringent controls, and advanced strategies outlined in this document will significantly enhance the accuracy and biological relevance of protein-protein interaction data derived from Co-IP and pull-down assays. By systematically combating false positives and negatives, researchers can construct more reliable models of signaling pathways, thereby accelerating the discovery of novel therapeutic targets and advancing our fundamental understanding of cellular communication networks.
Optimizing Crosslinking for Capturing Weak or Transient Interactions
Weak or transient protein-protein interactions (PPIs) are fundamental regulators of cellular signaling pathways, yet their dynamic nature and low affinity present significant challenges for detection and analysis using conventional methods [64]. Crosslinking mass spectrometry (XL-MS) has evolved as a powerful technique to study these elusive interactions in their native cellular environment, providing both interaction partners and structural information [65]. Recent methodological breakthroughs in crosslinking chemistry, enrichment strategies, and mass spectrometric instrumentation have dramatically improved the sensitivity and throughput required to capture these biologically significant but technically challenging interactions. This Application Note provides detailed protocols and optimized workflows specifically designed to enhance the identification of weak or transient PPIs, enabling researchers in signaling pathway analysis and drug development to obtain more comprehensive interactome maps.
Key Advancements in Crosslinking Technologies
MS-Cleavable, Enrichable Crosslinkers
The development of specialized crosslinking reagents has been pivotal for improving the detection of low-abundance crosslinked peptides. Disuccinimidyl bis-sulfoxide (DSBSO), an MS-cleavable, enrichable linker, demonstrates surprising membrane permeability despite charged nitrogen atoms within its net neutral azide residue, making it particularly suitable for in vivo studies [65]. This membrane permeability enables the capture of transient interactions in their native cellular environment before stabilization by crosslinking. The azide tag allows for efficient enrichment via click chemistry, significantly reducing sample complexity and enhancing detection of low-abundance species.
Other advanced crosslinkers like PhoX (DSPP) and DSSO provide additional options with different specificities and fragmentation characteristics [66]. These MS-cleavable crosslinkers generate characteristic signature ions upon collisional activation, facilitating confident identification of crosslinked peptides within complex mixtures.
Orthogonal Enrichment Strategies
Effective enrichment of crosslinked peptides is crucial for reducing sample complexity and enhancing sensitivity for detecting weak or transient interactions. A streamlined workflow combining affinity enrichment with size exclusion chromatography (SEC) has demonstrated remarkable improvements in identification rates [65].
Table 1: Performance Comparison of Enrichment Strategies
| Enrichment Method | Unique Crosslinks Identified | Background Linear Peptides | Key Advantages |
|---|---|---|---|
| Affinity Enrichment Only | ~1,000-2,000 | High | Specific capture of crosslinked peptides |
| SEC Only | 1,500-2,500 | Moderate | Separation by size |
| Combined Affinity + SEC | >3,000 | Low | Maximum sensitivity, minimal background |
| SEC Early Fractions | ~90% of total crosslinks | Very Low | CSM/Monolink ratio >6 |
The transition from Sepharose-based to magnetic bead technology for affinity enrichment has significantly improved washing efficiency and recovery, with Cytiva beads demonstrating 314% improvement in crosslink identification compared to non-enriched samples in benchmark tests using crosslinked Cas9 [65]. Optimizing the bead volume to protein ratio to 100 μL/mg represents a critical parameter for maximizing recovery while minimizing non-specific binding.
Advanced Mass Spectrometry Instrumentation
Recent advancements in mass spectrometry instrumentation have dramatically enhanced crosslink identification rates. Comparative studies between Orbitrap Astral and Orbitrap Eclipse instruments demonstrate significant performance differences for XL-MS applications [66].
Table 2: Instrument Performance for Crosslink Identification
| Parameter | Orbitrap Astral | Orbitrap Eclipse |
|---|---|---|
| Unique Residue Pairs | 40% more than Eclipse | Baseline |
| MS1 Sensitivity | Superior for low-abundance precursors | Moderate |
| Optimal Fragmentation | Single HCD | Minimal dependence on fragmentation strategy |
| FAIMS Benefit | 30% increase in identifications | Standard improvement |
| Low Sample Amounts | Excellent performance | Reduced performance |
The Astral's combination of high MS1 sensitivity and rapid scan speed enables detection of approximately 48% additional crosslinks that are unique to FAIMS-enabled acquisitions, with particularly pronounced benefits at mid-range injection amounts (250 ng) [66]. This enhanced sensitivity is crucial for detecting low-abundance crosslinks derived from weak or transient interactions.
Optimized Protocol for Capturing Weak/Transient Interactions
In Vivo Crosslinking with DSBSO
Materials Required:
- DSBSO crosslinker (freshly prepared)
- K562 cells (or relevant cell line for signaling pathway)
- Quenching solution (Tris-HCl, ammonium bicarbonate)
- Lysis buffer (including protease inhibitors)
Procedure:
- Cell Preparation: Grow K562 cells to 70-80% confluence in appropriate medium. Harvest cells, count, and aliquot approximately 10⁷ cells per condition.
- Crosslinking: Prepare 2 mM DSBSO in pre-warmed culture medium. Replace existing medium with DSBSO-containing medium and incubate at 37°C for 30-45 minutes. Critical: Optimize incubation time for specific transient interactions—shorter times may capture more transient interactions while longer times increase crosslinking efficiency.
- Quenching: Remove crosslinking solution and add fresh medium containing 50 mM Tris-HCl (pH 7.5) or 20 mM ammonium bicarbonate to quench unreacted crosslinker. Incubate for 10 minutes at room temperature.
- Cell Lysis: Wash cells twice with ice-cold PBS. Lyse cells using appropriate lysis buffer with sonication or mechanical disruption. Note: Maintain native conditions without denaturants to preserve weak interactions.
- Clear Lysate: Centrifuge at 16,000 × g for 15 minutes at 4°C to remove insoluble material. Transfer supernatant to fresh tube and determine protein concentration.
Sample Preparation and Affinity Enrichment
Materials Required:
- DBCO-functionalized magnetic beads (Cytiva recommended)
- FASP digestion filters
- Trypsin/Lys-C mix
- Cu-free click reaction buffers
- Size exclusion columns (Superdex Increase 200)
Procedure:
- Filter-Aided Sample Preparation: Transfer 0.25-1 mg protein lysate to 30 kDa MWCO filters. Process according to standard FASP protocol with Trypsin/Lys-C digestion (1:50 enzyme:protein, 16 hours at 37°C).
- Peptide Recovery: Collect digested peptides by centrifugation. Acidify with 1% trifluoroacetic acid (TFA). Optional: Desalt using C18 columns, though recent protocols indicate this step can be omitted with only 13% reduction in crosslink identifications but 38% reduction in background peptides. [65]
- Click Chemistry Enrichment:
- Aliquot 25 μL DBCO-magnetic bead slurry per 0.25 mg protein digest.
- Wash beads twice with PBS with 0.5 M NaCl (reduces nonspecific binding).
- Resuspend peptides in PBS with 0.5 M NaCl and incubate with beads for 2 hours at room temperature with gentle rotation.
- Collect beads on magnet and wash sequentially with: PBS, 1 M NaCl, 80% ethanol, and 100% acetonitrile.
- On-bead Digestion (if necessary): For incomplete digestion, add fresh Trypsin/Lys-C in 50 mM ammonium bicarbonate and digest for 4 hours at 37°C.
- Elution: Elute crosslinked peptides from beads using 30% acetonitrile in 0.1% TFA.
Orthogonal Size Exclusion Chromatography
- SEC Setup: Equilibrate Superdex Increase 200 column with 30% acetonitrile in 0.1% TFA at 0.1 mL/min.
- Fractionation: Inject entire affinity-enriched sample and collect 0.5-minute fractions for 30 minutes.
- Fraction Selection: Based on retention time, pool early-eluting fractions (typically minutes 8-14, containing larger crosslinked peptides). Note: Analysis of a single SEC fraction can yield ~90% of all crosslinks when measurement time is limited. [65]
- Concentration: Concentrate pooled fractions using vacuum centrifugation to approximately 5 μL.
LC-MS/MS Analysis with FAIMS
Materials Required:
- Orbitrap Astral mass spectrometer (or equivalent)
- Aurora Ultimate column (25 cm, 1.6 μm)
- Mobile phases A (0.1% formic acid) and B (80% acetonitrile, 0.1% formic acid)
Procedure:
- Chromatography: Inject concentrated SEC fractions onto Aurora Ultimate column. Apply 90-minute gradient: 5-28% B over 75 minutes, 28-45% B over 15 minutes, 45-95% B over 1 minute, hold at 95% B for 5 minutes.
- FAIMS Optimization: Use compensation voltages of -48V, -60V, and -75V for optimal crosslink identification. [66]
- MS Method:
- MS1: Resolution 120,000; AGC target 500; Injection time 6 ms; Mass range 375-1500 m/z
- MS2: Data-dependent acquisition; Isolation window 1.4 m/z; HCD fragmentation at 30%; Charge states 3-8 included
- Data Acquisition: Acquire data using Orbitrap Astral with aforementioned parameters. Critical: Use single HCD fragmentation rather than stepped HCD for improved performance on Astral platform.
Workflow Visualization
Diagram 1: The optimized workflow for capturing weak or transient protein interactions, featuring in vivo crosslinking followed by orthogonal enrichment strategies prior to advanced mass spectrometric analysis.
Research Reagent Solutions
Table 3: Essential Research Reagents for Crosslinking Studies
| Reagent/Category | Specific Product | Function in Workflow |
|---|---|---|
| MS-Cleavable Crosslinker | DSBSO (Disuccinimidyl bis-sulfoxide) | Stabilizes weak/transient interactions in live cells; contains azide handle for enrichment |
| Magnetic Beads | Cytiva NHS-Activated Magnetic Beads | High-density DBCO functionalization for efficient click chemistry enrichment |
| Chromatography Column | IonOpticks Aurora Ultimate (25 cm, 1.6 μm) | Superior peak sharpness and separation efficiency for complex peptide mixtures |
| Size Exclusion Resin | Superdex Increase 200 | Orthogonal enrichment separating crosslinked peptides from monolinks/linear peptides |
| Mass Spectrometer | Orbitrap Astral with FAIMS | Enhanced sensitivity and scan speed for low-abundance crosslink detection |
| Protease | Trypsin/Lys-C Mix | Efficient digestion while maintaining crosslink stability |
Data Analysis and Validation
Following data acquisition, process raw files using specialized crosslinking search software (such as XlinkX, MaxLynx, or Kojak). Search parameters should include:
- MS1 and MS2 mass tolerances appropriate for instrument capabilities
- Specific cleavage rules for DSBSO (sulfoxide cleavage)
- Fixed modifications: carbamidomethylation (C)
- Variable modifications: oxidation (M), acetylation (protein N-term)
- Crosslinker-specific settings: DSBSO (138.0098 Da dead-end, 156.0203 Da looplink, 318.0041 Da crosslink)
Validate identified crosslinks using:
- False discovery rate estimation at ≤1% using target-decoy approach
- Manual verification of signature ions for a subset of crosslinks
- Correlation with known structural data or orthogonal interaction methods
Application to Signaling Pathway Research
This optimized protocol enables researchers to capture previously undetectable transient interactions in signaling pathways, such as:
- Kinase-substrate interactions with rapid turnover
- Receptor complex assembly/disassembly dynamics
- Nuclear transport interactions
- Transcription factor complex formation
The combination of in vivo crosslinking with DSBSO, orthogonal enrichment, and advanced mass spectrometry provides unprecedented sensitivity for mapping the complete architecture of signaling pathways, identifying novel interaction partners, and revealing mechanistic insights into cellular regulation.
The Yeast Two-Hybrid (Y2H) system remains one of the most powerful and widely employed techniques for detecting protein-protein interactions (PPIs) in vivo, contributing significantly to interaction databases and functional genomics projects [67] [1]. However, its effectiveness is frequently compromised by two persistent technical challenges: self-activation and bait protein toxicity. Self-activation occurs when bait proteins independently activate reporter gene transcription without interacting with prey proteins, complicating screening procedures and generating false positives [68]. Toxicity manifests when bait protein expression inhibits yeast growth, preventing the establishment of viable screening strains [69]. These issues are particularly prevalent in large-scale interaction mapping projects, where up to 5% of baits can exhibit self-activation properties—several orders of magnitude higher than the frequency of genuine interactors [68]. Within signaling pathway research, where accurate PPI mapping is crucial, addressing these artifacts is essential for generating reliable data. This application note provides detailed protocols and strategic solutions to identify, troubleshoot, and overcome these challenges, enabling more robust Y2H experiments in signaling pathway analysis.
Understanding the Pitfalls: Causes and Consequences
Self-Activation: Mechanisms and Identification
Self-activation in Y2H systems arises from multiple mechanisms. Certain bait proteins inherently possess transcriptional activation domains, a characteristic common among transcription factors and signaling molecules [68]. Other baits may acquire artificial transactivation capability when fused to the DNA-Binding Domain (DBD), often due to exposed acidic patches or intrinsic disorder that nonspecifically recruits the transcriptional machinery [67] [68]. Spurious self-activators can also originate from cloning artifacts, such as out-of-frame fusions in random libraries or PCR-induced mutations in directed cloning strategies [68].
The consequences of unchecked self-activation are severe for interaction studies. It generates false positives that overwhelm true interaction signals, compromises screening efficiency by increasing background noise, and ultimately leads to inaccurate protein interaction maps that misrepresent signaling networks [67] [68]. Systematic comparisons of Y2H variants have demonstrated that different vector systems and fusion orientations detect substantially different PPI subsets, highlighting the methodological sensitivity to such technical artifacts [67].
Table 1: Common Causes and Characteristics of Self-Activating Baits
| Category | Molecular Basis | Frequency in Libraries | Reporter Gene Response |
|---|---|---|---|
| Natural Transcriptional Activators | Native activation domain function | Variable (depends on protein set) | Strong, concentration-independent |
| Artificial Activators | Non-specific recruitment of transcription machinery | ~1% of random E. coli sequences [68] | Weak to moderate, may be concentration-dependent |
| Cloning Artifacts | Out-of-frame fusions, mutation-induced | Up to 5% in large-scale screens [68] | Variable |
| Signaling Proteins | Post-translational modifications or co-factor mimicry | Common in kinase/phosphatase studies | Context-dependent |
Bait Toxicity: Origins and Impact
Bait toxicity presents a complementary challenge in Y2H systems, particularly when studying signaling proteins. Toxicity mechanisms include inhibition of essential yeast processes through improper signaling cascade activation, proteostatic burden from misfolded or aggregation-prone proteins, and pore formation or membrane disruption, especially with toxin-domain containing proteins like the PFT protein in wheat [69] [67]. For signaling pathway researchers, this is particularly problematic when studying pro-apoptotic proteins, kinases with broad specificity, or proteins involved in stress responses.
The functional impact includes aberrant yeast colony morphology, reduced transformation efficiency, and complete failure to establish stable bait strains [69]. In the case of the Fhb1 PFT protein, expression of the full-length protein or its ETX/MTX2 toxin domain severely inhibited yeast growth, while the agglutinin domains alone were well-tolerated [69]. Such toxicity prevents screening altogether rather than generating false positives, making it a more absolute but often addressable barrier.
Strategic Solutions and Experimental Design
Genetic Selection Against Self-Activators
A powerful genetic strategy for eliminating self-activators employs negative selection before library screening. This approach utilizes the URA3 reporter gene under the control of GAL upstream activating sequences. When expressed, URA3 converts 5-fluoroorotic acid (5-FOA) into a toxic product, enabling counterselection against self-activating baits [68].
The implementation workflow begins with transforming the bait plasmid into the Y2H yeast strain and selecting transformants on synthetic dropout medium lacking tryptophan (SD/-Trp). These transformants are then replica-plated onto SD/-Trp plates containing 5-FOA. Colonies expressing self-activators fail to grow due to URA3 expression, while non-activators grow normally. The 5-FOA-resistant colonies are recovered and can proceed to mating with library strains [68]. This pre-clearing step efficiently removes the majority of self-activators, significantly improving the signal-to-noise ratio in subsequent screens.
Molecular Engineering to Circumvent Toxicity
For toxic baits, several molecular engineering strategies have proven effective. Domain mapping and truncation identifies non-toxic protein regions while retaining interaction capacity, as demonstrated with the PFT protein where the agglutinin domains were non-toxic while the ETX/MTX2 domain inhibited growth [69]. Terminal fusion switching provides an alternative approach; fusing the bait to the Activation Domain (AD) rather than the DBD can sometimes alleviate toxicity, as successfully implemented for the transcription factor FOXA3 [70].
Inducible promoter systems that decouple bait expression during strain establishment from screening phases offer another solution, though they require specialized vector systems. Additionally, lower-copy number CEN/ARS vectors (versus 2µ plasmids) reduce bait expression levels, potentially mitigating toxicity while maintaining sufficient levels for interaction detection [67].
Diagram: Strategic approaches for mitigating bait toxicity in Y2H systems. Multiple molecular engineering strategies can convert a toxic bait into a viable screening strain.
Vector and System Selection
The choice of Y2H vector system significantly impacts the success of detecting signaling protein interactions. Systematic comparisons reveal that no single vector combination detects all interactions, with N-terminal versus C-terminal fusions exhibiting markedly different interaction profiles [67]. For example, in Varicella Zoster Virus screens, N-terminal baits with N-terminal preys (NN) produced the highest number of interactions, while NC screens yielded the lowest [67].
Table 2: Y2H Vector Systems and Their Applications for Problematic Baits
| Vector | Promoter | Fusion Orientation | Selection | Advantages for Challenging Baits |
|---|---|---|---|---|
| pGBKT7g | t-ADH1 | N-terminal DBD | Trp1, Kanamycin | Strong expression, 2µ origin |
| pGADT7g | fl-ADH1 | N-terminal AD | Leu2, Ampicillin | Compatible with fusion switching |
| pGBKCg | t-ADH1 | C-terminal DBD | Trp, Kanamycin | Alternative topology for screen |
| pGADCg | fl-ADH1 | C-terminal AD | Leu, Ampicillin | Reduces terminal accessibility issues |
| pDEST32 | fl-ADH1 | N-terminal DBD | Leu2, Gentamicin | CEN origin, lower copy number |
| pDEST22 | fl-ADH1 | N-terminal AD | Trp1, Ampicillin | Gateway compatibility |
Combining results from multiple vector systems significantly increases interaction detection rates. While individual assays detected variable portions of a gold-standard interaction set, combining three or four separate Y2H assays detected up to 78-83% of true positive interactions [67]. This systematic approach to vector selection is particularly valuable for comprehensive mapping of signaling complexes.
Detailed Experimental Protocols
Protocol 1: Bait Validation and Self-Activation Testing
Purpose: To identify and eliminate self-activating baits prior to library screening.
Materials:
- Y2HGold or similar reporter strain (multiple GAL4-responsive reporters)
- Bait plasmid (pGBKT7-based or equivalent)
- SD/-Trp, SD/-Trp/-His, SD/-Trp/-His/-Ade plates
- X-α-Gal solution (for colorimetric assay)
- 3-AT (3-amino-1,2,4-triazole) for background suppression
- 5-FOA plates for counterselection
Procedure:
- Transform bait plasmid into Y2HGold yeast strain, select on SD/-Trp (3-5 days, 30°C).
- Patch 10-20 colonies on SD/-Trp/-His and SD/-Trp/-His/-Ade plates.
- Include positive (known activator) and negative (empty vector) controls.
- Assess growth after 3-5 days: no growth indicates no self-activation.
- For baits showing weak self-activation, titrate with 1-50 mM 3-AT to suppress background.
- For colorimetric assessment, include X-α-Gal in plates; blue color indicates activation.
- For confirmed self-activators, implement 5-FOA counterselection:
- Grow bait transformants in SD/-Trp liquid medium to OD600 ~1.0
- Plate ~10^6 cells on SD/-Trp + 5-FOA plates
- Incubate 3-5 days at 30°C
- Restreak resistant colonies for further analysis
Troubleshooting:
- If most baits show self-activation, consider C-terminal fusions or domain truncations.
- For variable self-activation between colonies, sequence bait plasmid to check for mutations.
- If 5-FOA selection is too stringent, use lower concentrations (0.1-0.5 mg/mL).
Protocol 2: Toxicity Assessment and Mitigation
Purpose: To identify toxic baits and implement corrective strategies.
Materials:
- Y2HGold or AH109 yeast strains
- Bait plasmids (full-length and truncated versions)
- SD/-Trp plates and liquid medium
- Cloning reagents for domain truncation
- Alternative vectors (C-terminal fusions, inducible promoters)
Procedure:
- Transform full-length bait plasmid, select on SD/-Trp (3-5 days, 30°C).
- Compare transformation efficiency with empty vector control:
- >10-fold reduction indicates significant toxicity
- Assess colony size: microscopic colonies suggest growth inhibition.
- For toxic baits, generate domain-truncated constructs:
- Identify functional domains using Pfam, SMART databases
- Clone individual domains into bait vector
- Test truncated constructs for toxicity as above
- Alternative fusion orientation:
- Clone bait into AD vector (pGADT7) instead of DBD vector
- Test for toxicity and self-activation
- If toxicity persists, consider:
- Lower-copy CEN/ARS vectors (e.g., pDEST32)
- Weaker promoters (minimal ADH1)
- Inducible expression systems (GAL1 promoter)
Validation:
- Confirm truncated baits retain interaction capability with known partners
- Test expression levels by Western blotting if antibodies available
- Ensure non-toxic baits don't show aberrant localization in yeast
Diagram: Comprehensive workflow for bait validation in Y2H systems. The decision tree guides researchers through toxicity and self-activation testing toward appropriate mitigation strategies.
Protocol 3: Library Screening with Pre-Cleared Baits
Purpose: To screen Y2H library with validated, non-self-activating, non-toxic baits.
Materials:
- Pre-validated bait strain
- Mate & Plate library or custom prey library
- SD/-Trp/-Leu (DDO) and SD/-Trp/-Leu/-His/-Ade (QDO) plates
- X-α-Gal for blue-white selection
- 3-AT for background suppression (concentration determined in Protocol 1)
Procedure:
- Grow bait strain in SD/-Trp to OD600 = 0.8 (~50 mL culture).
- Mix with library strain (prey) in 2:1 ratio (bait:prey).
- Incubate in YPDA medium overnight at 30°C with gentle shaking (40-50 rpm).
- Plate mating mixture on QDO plates + X-α-Gal + optimal 3-AT concentration.
- Incubate at 30°C for 3-7 days, monitoring for colony formation and color development.
- Pick colonies appearing after 3-5 days (avoid very late-appearing colonies).
- Restreak positive colonies on fresh QDO plates to confirm phenotype.
- Isolate prey plasmids and sequence for identification.
- Confirm interactions by co-transforming purified prey with original bait.
Quality Control:
- Include positive interaction control pairs in each screening batch
- Calculate screening coverage (number of clones screened vs. library complexity)
- Verify prey plasmid identity by restriction digest or PCR
- Retest interactions through fresh co-transformation
The Scientist's Toolkit: Essential Reagents and Solutions
Table 3: Key Research Reagent Solutions for Y2H Troubleshooting
| Reagent/Resource | Specific Examples | Function/Application | Considerations for Signaling Proteins |
|---|---|---|---|
| Y2H Vectors | pGBKT7g, pGADT7g, pDEST22/32 | DBD and AD fusion platforms | Gateway compatibility for high-throughput; C-terminal fusion vectors for topology issues |
| Yeast Strains | Y2HGold, AH109, Y187 | Reporter strains with multiple auxotrophic markers | Varying stringency with different reporter combinations |
| Selection Agents | 3-AT, 5-FOA | Suppress background, counterselect self-activators | 3-AT concentration must be optimized for each bait |
| Domain Analysis Tools | SMART, Pfam, PredictProtein | Identify domains for truncation strategies | Conserved signaling domains often fold independently |
| Library Construction Kits | Make Your Own "Mate & Plate" Library | Build custom tissue/time-specific libraries | Essential for signaling studies where interactions are context-dependent |
| Negative Selection Plates | SD + 5-FOA + appropriate dropouts | Eliminate self-activating baits from pools | Critical preprocessing step for large-scale signaling studies |
| Interaction Databases | BioGRID, STRING, IntAct | Validate found interactions against known data | Signaling interactions often conserved but context-dependent |
Addressing self-activation and toxicity challenges requires a systematic approach combining strategic vector selection, genetic counter-selection, and molecular engineering of problematic baits. The protocols outlined here provide a comprehensive framework for researchers studying signaling pathways to overcome these technical hurdles. By implementing bait validation workflows, utilizing appropriate negative selection strategies, and applying molecular solutions like domain truncation and fusion switching, investigators can significantly enhance the reliability and coverage of their Y2H screens. As Y2H methodology continues to evolve—integrating with next-generation sequencing in techniques like DoMY-Seq [71] and benefiting from computational predictions [72] [1]—these fundamental approaches to addressing classical pitfalls remain essential for generating high-quality protein interaction data in signaling pathway research.
The reliability of conclusions drawn from protein-protein interaction (PPI) assays is foundational to research in signaling pathway analysis and drug development. Inherent complexity and variability of biological systems mean that without careful experimental design, results can be misleading or irreproducible [73]. The strategic inclusion of critical controls mitigates these risks by accounting for experimental noise, bias, and artifacts. This document provides a structured framework for designing robust PPI experiments, ensuring that observed phenomena are biologically meaningful rather than mere consequences of methodological flaws. By integrating principles of Design of Experiments (DoE) with specific protocols for interaction assays, we empower researchers to generate high-quality, interpretable data that can confidently inform subsequent research and development stages [74].
Core Principles of Experimental Design for PPI Assays
A well-designed experiment is not the result of post-hoc statistical analysis but is built upon a foundation of core principles established during the planning phase. These principles are crucial for managing the complexity and high-dimensionality of data in modern -omics research, including PPI studies [73].
Replication and Power Analysis: Replication involves independent repeat runs of each experimental condition and is essential for estimating experimental error and improving precision [74]. A key challenge is distinguishing between technical replicates (multiple measurements from the same biological sample) and biological replicates (independent biological samples), with the latter being critical for drawing generalizable conclusions [73]. Conducting a power analysis before an experiment helps optimize sample size, ensuring a high probability of detecting true effects while avoiding resource waste on underpowered studies [73].
Randomization: This is the process of randomly assigning samples to treatment groups and randomizing the order of experimental runs. It helps prevent systematic bias from unknown or unmeasured confounding variables, such as instrument drift or subtle environmental changes over time [74].
Blocking: Blocking is a technique used to reduce variability from known nuisance factors. Researchers group experimental units into homogeneous "blocks" and then randomize treatments within each block. For example, if an experiment must be conducted over several days, "day" can be treated as a block to account for day-to-day variation, thereby isolating the true treatment effect more clearly [74].
Factorial Experimentation: Traditional "one-factor-at-a-time" (OFAT) approaches are inefficient and incapable of detecting interactions between factors. Factorial designs, where multiple factors are varied simultaneously and orthogonally, allow for efficient assessment of both individual factor effects and their interactions [74]. This is particularly valuable in complex PPI assay development, where factors like pH, temperature, and salt concentration may interact.
Table 1: Core Principles of Experimental Design for PPI Assays
| Principle | Definition | Role in PPI Assays | Common Pitfalls |
|---|---|---|---|
| Replication | Independent repetition of experimental conditions. | Distinguishes consistent interactions from random noise; provides estimate of variance. | Pseudoreplication (treating technical replicates as biological replicates). |
| Randomization | Random assignment of samples to treatment order. | Minimizes bias from unmeasured confounders (e.g., cell passage number, reagent lot). | Non-random sequence of experiments introducing temporal bias. |
| Blocking | Grouping similar experimental units to control for a known nuisance variable. | Accounts for batch effects in reagents, different operators, or multiple sequencing runs. | Failure to identify a major source of variation (e.g., different cell incubators). |
| Factorial Design | Varying multiple factors simultaneously to study main effects and interactions. | Efficiently optimizes multiple assay parameters (e.g., buffer conditions, temperature). | Using OFAT approaches, which miss interactions and are resource-inefficient. |
Essential Controls for PPI Experiments
Controls are the benchmark against which experimental results are validated. Their omission renders the biological interpretation of data ambiguous.
Negative Controls
Negative controls are designed to fail to produce the expected outcome, helping to identify background signal or non-specific binding.
- Empty Vector/Bait-Less Control: In yeast two-hybrid (Y2H) or co-immunoprecipitation (Co-IP), this involves expressing the prey protein with an empty bait vector or a non-interacting bait protein. It confirms that the signal is specific to the interaction between the proteins of interest.
- Knockout/Knockdown Control: Using cells where one of the interacting partners has been genetically deleted or silenced (e.g., via CRISPR/Cas9 or siRNA). This provides a baseline for non-specific antibody binding in Co-IP or background fluorescence.
- Isotype Control: In Co-IP, using a non-specific antibody of the same isotype as the immunoprecipitating antibody. This identifies proteins that bind non-specifically to the antibody bead matrix.
- Solvent/Vehicle Control: When testing the effect of a small molecule inhibitor on a PPI, the compound's solvent must be tested alone to rule out solvent-induced effects.
Positive Controls
Positive controls are designed to successfully produce the expected outcome, verifying that the experimental system is functioning correctly.
- Known Interacting Pair: Including a well-characterized protein pair with a strong, validated interaction in every run of the assay (e.g., FRET or Bioluminescence Resonance Energy Transfer (BRET) standards). This ensures reagents and equipment are working properly.
- Constitutively Active Component: In functional assays, a protein known to always induce a signal can serve as a control for the reporting system's integrity.
Experimental and Normalization Controls
These controls account for variability in sample preparation and measurement.
- Loading Controls: In Western blot analysis following Co-IP, proteins like GAPDH, Actin, or Tubulin are probed to confirm equal protein loading across lanes.
- Expression Controls: In overexpression systems, verifying that the bait and prey proteins are expressed at comparable levels is crucial to avoid misinterpreting low signal due to lack of expression as a lack of interaction.
Table 2: Critical Controls for PPI Assay Validation
| Control Category | Specific Example | Protocol Application | Interpretation of Result |
|---|---|---|---|
| Negative Control | Empty vector in Y2H. | Co-transform prey plasmid with empty bait plasmid. | Growth on selective media indicates autoactivation; experiment is invalid. |
| Negative Control | Isotype control in Co-IP. | Use non-specific IgG for immunoprecipitation. | Bands in MS/Western blot indicate non-specific binding. |
| Positive Control | Known interacting pair in FRET. | Express calibrated FRET standard pair. | Low FRET efficiency suggests instrument or reagent failure. |
| Normalization Control | Total protein load in Co-IP. | Probe for a housekeeping protein in the whole-cell lysate. | Uneven bands indicate unequal loading, requiring normalization. |
Detailed Protocol: Co-Immunoprecipitation (Co-IP) with Controlled Design
This protocol integrates DoE principles and critical controls for a robust Co-IP experiment to validate a putative PPI.
Objective: To confirm a physical interaction between Protein X (bait) and Protein Y (prey) in a mammalian cell line and assess the interaction's dependence on a specific signaling pathway.
Pre-Experimental Planning
- Define Objective and Hypothesis: The objective is confirmation and initial characterization. The hypothesis is that Protein X interacts with Protein Y, and this interaction is strengthened upon activation of the MAPK/ERK pathway.
- Determine Response Variables: Primary response is the intensity of the prey protein band (Protein Y) on a Western blot, normalized to the bait protein band (Protein X) in the IP eluate. Secondary response is the ratio of interaction strength (+EGF / -EGF).
- Determine Factors and Levels: A simplified 2-factor design is used:
- Factor A: EGF Treatment (2 levels: 0 ng/mL, 100 ng/mL to activate MAPK/ERK pathway).
- Factor B: Cell Line (2 levels: Wild-Type, Protein X Knockout).
- Select Experimental Design: A full factorial design with blocking is selected. The entire experiment is replicated three times (n=3 biological replicates) on different days to ensure generalizability. The run order for processing cell lysates is randomized to prevent bias.
Reagents and Materials
Table 3: Research Reagent Solutions for Co-IP Protocol
| Item | Function | Example/Catalog # |
|---|---|---|
| Mammalian Expression Vectors | For expressing tagged bait (Protein X-Flag) and untagged prey (Protein Y). | pcDNA3.1(+) |
| Cell Line | Model system for the interaction; HEK293T are highly transfectable. | HEK293T (ATCC CRL-3216) |
| Protein X Knockout Line | Critical negative control cell line. | Generated via CRISPR/Cas9 |
| Transfection Reagent | For introducing plasmid DNA into mammalian cells. | Polyethylenimine (PEI) or commercial lipofectamine. |
| Anti-Flag Affinity Gel | For immunoprecipitation of the bait protein. | Sigma A2220 |
| Normal Mouse IgG | Isotype control antibody for negative control IP. | Santa Cruz Biotechnology sc-2025 |
| EGF | Signaling pathway activator for factorial design. | PeproTech AF-100-15 |
| Lysis Buffer | To extract proteins while preserving interactions. | RIPA buffer + protease/phosphatase inhibitors. |
| Primary Antibodies | For detection of bait (Anti-Flag) and prey (Anti-Protein Y) via Western blot. | Custom or commercial specific antibodies. |
Step-by-Step Workflow
- Cell Seeding and Transfection: Seed HEK293T (Wild-Type and Protein X KO) cells in multiple T75 flasks. The following day, transfert with plasmids encoding: (a) Protein X-Flag + Protein Y, (b) Empty Vector + Protein Y.
- Treatment and Stimulation: 24 hours post-transfection, serum-starve cells for 4-6 hours. Treat with the appropriate level of Factor A (0 or 100 ng/mL EGF) for 15 minutes.
- Cell Lysis: Place cells on ice, wash with cold PBS, and lyse in IP-compatible lysis buffer. Centrifuge to clear lysates.
- Protein Quantification: Normalize all lysates to the same protein concentration.
- Immunoprecipitation: For each lysate, set up two IP reactions:
- Test IP: Incubate with Anti-Flag Affinity Gel.
- Control IP: Incubate with Normal Mouse IgG coupled to beads.
- Incubate at 4°C for 2-4 hours with gentle rotation.
- Washing and Elution: Wash beads 3-5 times with lysis buffer. Elute proteins with 2X Laemmli sample buffer.
- Analysis: Analyze eluates and corresponding whole-cell lysates by SDS-PAGE and Western blotting. Probe membranes sequentially for the prey (Protein Y) and the bait (Protein X-Flag).
Data Analysis and Interpretation
- Analyze Western blot data by quantifying band intensities.
- The interaction is considered specific if the prey (Protein Y) is detected in the test IP (Anti-Flag) from wild-type cells but is absent in the control IP (IgG) and in the test IP from the Protein X Knockout cells.
- The effect of EGF is assessed by comparing the normalized prey/bait ratio between the -EGF and +EGF conditions in the wild-type cells. Statistical significance is determined using a two-way ANOVA, which can account for the effects of both EGF and cell line, as well as their interaction.
Visualizing Experimental Workflows and Signaling Pathways
The following diagrams, generated using Graphviz DOT language, illustrate the core experimental logic and a relevant signaling pathway context. The color palette and contrast are designed per specified guidelines to ensure clarity and accessibility.
Experimental Workflow for Controlled Co-IP
MAPK/ERK Pathway Influencing PPIs
Managing Expression Levels and Environmental Conditions for In Vivo Assays
Within the broader research on protein-protein interaction (PPI) assays for signaling pathway analysis, the fidelity of in vivo data is paramount. A significant challenge in this field is the inherent gap between simplified in vitro models and the complex physiological reality of a living organism [75]. Environmental conditions and precise management of gene expression are not merely background variables; they are active determinants of whether observed molecular interactions accurately reflect true biological function or are artifacts of the experimental system. This application note provides detailed protocols grounded in the principle of Toxicogenomics (TGx), leveraging advanced computational corrections to bridge the in vitro to in vivo (IVIVE) gap, thereby ensuring that data derived from protein-protein interaction assays within signaling pathways is both reliable and translatable [75].
Key Concepts and Environmental Parameters
In vivo assays are influenced by a multitude of internal environmental factors that are absent in cell culture. These include complex pharmacokinetic and pharmacodynamic (PK/PD) profiles, systemic immune responses, and the multifaceted interactions between different cell types [75]. In the context of signaling pathways, these factors can modulate PPIs by altering protein expression levels, post-translational modifications, and subcellular localization.
Critical Environmental Modulators of Genetic Risk and protein function have been identified through frameworks that characterize transcriptional responses to diverse environmental perturbations [76]. Furthermore, the competitive and cooperative dynamics within protein triplets—a higher-order interaction motif—can be influenced by cellular conditions, which in turn affect signaling output [36]. systematically controlling for these variables is essential for reproducible and meaningful in vivo results.
Table 1: Key Environmental and Experimental Parameters for In Vivo Assays
| Parameter Category | Specific Factors | Impact on PPI & Signaling Assays | Considerations for Data Interpretation |
|---|---|---|---|
| Genetic & Expression Control | dsRNA length & concentration [77]; Target gene selection (e.g., ebony, laccase 2) [77] | Determines efficacy and specificity of gene knockdown in RNAi assays, directly modulating PPI components. | Optimize dsRNA dose and length to minimize off-target effects and competition [77]. |
| Systemic Physiological Factors | Cell types; Culture conditions; Time course of exposure; Measured endpoints [75] | Inner-environmental reactions significantly affect genetic variation and gene abundance, confounding PPI data. | Use strategies like Non-negative Matrix Factorization (NMF) to factor out inner-environmental signals [75]. |
| Assay Validation & Design | Randomization; Statistical power & sample size; Reproducibility across runs [78] | Ensures biologically meaningful effects on signaling pathways are statistically significant and not random. | Follow pre-study, in-study, and cross-study validation procedures as per Assay Guidance Manual [78]. |
| Data Presentation | Use of frequency tables, histograms, and frequency polygons [79] [80] | Clear graphical presentation allows for correct interpretation of quantitative data distributions and trends. | For comparative data (e.g., treatment vs. control), use frequency polygons for clearer visualization [79]. |
Detailed Experimental Protocols
Protocol: A Two-Step In Vivo RNAi Assay to Validate Components of Environmental RNAi
This protocol, adapted from research on the Western Corn Rootworm, provides a robust system for validating genes involved in environmental RNAi, a technique critical for manipulating expression levels of PPI components in vivo [77].
I. Principle A two-step feeding assay is used to determine if a candidate gene is involved in the systemic RNAi pathway. Larvae are first fed dsRNA targeting a candidate gene, then fed dsRNA targeting a visual marker gene. Successful knockdown of the marker gene indicates the candidate gene is not essential for RNAi, while a lack of knockdown implicates the candidate in the pathway.
II. Reagents and Materials
- dsRNA synthesized for: a) Candidate gene(s) (e.g., Dcr-2, Ago-2, sid-1-like genes), b) Marker genes (e.g., ebony or laccase 2 for cuticle pigmentation).
- Artificial diet for insect feeding.
- Equipment for dsRNA synthesis and purification.
- Imaging system or visual scoring apparatus for phenotype assessment.
III. Procedure
- dsRNA Preparation: Synthesize and purify dsRNA for both candidate and marker genes. The study optimized dsRNA length to >= 60 bp for effective systemic RNAi and minimized competitive effects [77].
- Primary Feeding (Candidate Gene Knockdown):
- Prepare diet infused with candidate gene dsRNA. A dose of 100 ng/cm² of diet is recommended as a starting point [77].
- Feed larvae for a period of 2-3 days.
- Secondary Feeding (Marker Gene Knockdown):
- Transfer larvae to a fresh diet infused with marker gene dsRNA (ebony or laccase 2).
- Feed for an additional 2-3 days.
- Phenotypic Analysis:
- Monitor and score larvae for the expected phenotypic change (e.g., cuticle pigmentation alteration).
- A clear phenotypic change indicates an active RNAi response, meaning the initial candidate gene knockdown did not impair the pathway.
- A lack of phenotypic change suggests the candidate gene is essential for environmental RNAi.
IV. Data Analysis Compare the incidence and penetrance of the marker gene phenotype between negative control groups (e.g., fed with non-specific dsRNA) and experimental groups. Statistical validation of the assay performance is critical, ensuring the Minimum Significant Difference (MSD) is established during pre-study validation [78].
Protocol: Computational Simulation of In Vivo Data from In Vitro Corrections
This protocol uses a computational strategy to refine in vitro PPI data, enhancing its correlation with in vivo conditions [75].
I. Principle Post-modified Non-negative Matrix Factorization (NMF) is used to deconvolute gene expression profiles from in vivo assays. The algorithm factorizes the data to estimate the expression profiles and contents of major factors, including drug effects and inner-environmental reactions. The isolated environmental factor can then be used to correct in vitro data to better simulate an in vivo profile.
II. Data Input Requirements
- Gene expression data from in vivo assays (e.g., TGx data from liver studies).
- Corresponding gene expression data from in vitro assays (e.g., cultured HepG2 cells).
III. Procedure
- Data Pre-processing: Normalize and prepare both in vivo and in vitro gene expression matrices.
- Factor Decomposition: Apply post-modified NMF to the in vivo data to factorize the matrix into two non-negative matrices, representing metagenes and metasamples. This identifies latent factors, including those related to the inner environment.
- Factor Isolation: Identify and retain the factor(s) corresponding to the inner-environmental response, segregating them from the direct drug-effect signals.
- In Vitro Simulation: Utilize the identified environmental factor to correct the in vitro data, generating a simulated in vivo profile.
IV. Data Analysis The similarity between the real in vivo data and the NMF-simulated data should be quantitatively compared to the similarity between real in vivo and raw in vitro data. The published method achieved similarities of 0.72-0.75 for simulated data versus 0.56-0.70 for direct comparison, demonstrating a significant improvement [75].
Workflow Visualization
The following diagram illustrates the logical workflow for managing expression levels and bridging the in vitro-in vivo gap, as detailed in the protocols above.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for In Vivo Expression and Interaction Analysis
| Research Reagent / Assay | Primary Function | Application in PPI & Signaling Pathway Research |
|---|---|---|
| Cell Viability Assays (ATP-based) [81] | Measures ATP levels as a marker of metabolically active, viable cells. | Critical for normalizing PPI data (e.g., co-IP efficiency) to cell number and health after genetic or environmental perturbation. |
| Cytotoxicity Assays (LDH-based) [81] | Measures lactate dehydrogenase (LDH) release from cells with compromised membranes. | Used to distinguish specific pathway modulation from general cytotoxic effects in response to a treatment. |
| Double-Stranded RNA (dsRNA) [77] | Triggers sequence-specific gene silencing via the RNA interference (RNAi) pathway. | Foundational for knocking down expression of specific proteins in a complex to study their role in PPIs and pathway flux in vivo. |
| Tetrazolium Reduction Assays (e.g., MTS, MTT) [81] | Measures metabolic activity of cells via enzymatic conversion of a substrate to a colored formazan product. | A less sensitive, but cost-effective method for assessing cell proliferation and viability in larger-scale, lower-throughput in vitro screens. |
| Real-Time Viability Assays [81] | Uses engineered luciferase and prosubstrate for kinetic, non-lytic monitoring of cell viability. | Enables longitudinal studies of signaling pathway activity and PPI dynamics in the same cell population over time, reducing well-to-well variability. |
Building Confidence: Validation Strategies and Comparative Method Analysis
Protein-protein interactions (PPIs) are fundamental to virtually every cellular process, forming the backbone of signaling pathways that control cell growth, differentiation, and death. The identification of true protein interactors is therefore crucial to understanding molecular functions in both physiological and pathological contexts [82]. However, PPIs are dynamic, context-dependent, and vary in strength and stability, making them challenging to capture comprehensively with any single experimental approach. Significant research efforts have been wasted on repeatedly identifying the same abundant peptides or those from "sticky proteins" that bind nonspecifically to many baits in interaction analyses [82]. This application note demonstrates why a multi-method approach is indispensable for establishing high-confidence PPI networks, providing detailed protocols and analytical frameworks for researchers engaged in signaling pathway analysis and drug development.
Comparative Analysis of PPI Assay Methods
Different PPI assay techniques offer complementary strengths and address specific limitations. The selection of methods should be guided by the biological question, required throughput, and desired confirmation level.
Table 1: Comparison of Key Protein-Protein Interaction Assay Methods
| Method Type | Principal Technique | Key Strengths | Inherent Limitations | Optimal Use Case |
|---|---|---|---|---|
| Affinity Purification-MS | Tandem Affinity Purification (TAP) with SFB-tag [82] | High specificity due to two-step purification; mild elution conditions; small tags minimize protein folding disruption | May lose weakly interacting or transient proteins; requires recombinant tagging | Identification of stable complexes under near-physiological conditions |
| Proximity-Based Labeling | BioID [82] | Non-toxic labeling; captures weak/transient interactions in living cells; extensively validated across studies | Poor temporal resolution; limited application in vivo due to low catalytic activity | Mapping interaction proximities in living cells |
| Enzyme Complementation | Split-Luciferase Assay [83] | High temporal resolution; suitable for high-throughput compound screening; monitors interaction dynamics | Requires protein fusion constructs; may not reflect endogenous complexes | High-throughput screening of compound libraries; real-time interaction dynamics |
| Computational Integration | Pathway-Centric PTM Analysis [84] | Provides holistic context of cellular pathways; enables integration of multiple data types | Dependent on quality of prior knowledge databases; computational expertise required | Interpreting PTM data in signaling pathways; multi-omics integration |
Detailed Experimental Protocols
SFB-Tag Based Tandem Affinity Purification Mass Spectrometry (TAP/MS)
Principle and Workflow
The SFB (S-protein, FLAG, Streptavidin-Binding Peptide) tandem tag system enables two-step purification under both native and denaturing conditions, significantly reducing nonspecific binding compared to single-step affinity purification [82]. The FLAG-tag facilitates detection via western blotting, while the SBP-tag enables high-yield purification with streptavidin beads and gentle biotin elution.
Required Materials and Reagents
- Plasmid Construction: Gateway cloning system with attB1/attB2 homologous sequences in primers [82]
- Cell Lines: HEK293T (high transfection efficiency) or lentiviral transduction for difficult-to-transfect cells (e.g., MCF10A, JURKAT)
- Lysis Buffer: 50 mM Tris-HCl (pH 7.5), 150 mM NaCl, 1 mM EDTA, 0.5% NP-40, plus fresh protease and phosphatase inhibitors
- Purification Matrices: Streptavidin-conjugated beads (first step), S-protein agarose (second step)
- Elution Buffer: 2 mg/mL biotin in lysis buffer
- Mass Spectrometry: Trypsin digestion, LC-MS/MS analysis
Step-by-Step Protocol
Plasmid Preparation (Timing: 1 week)
- Amplify gene of interest from cDNA using Phusion DNA polymerase with attB1/attB2 homologous sequences in primers [82]
- Perform BP recombination reaction to create entry clone
- Perform LR recombination reaction to generate SFB-tagged destination vector
- Validate construct by sequencing
Stable Cell Line Generation (Timing: 2-3 weeks)
- Transfect HEK293T cells with SFB-tagged plasmid using preferred method (e.g., calcium phosphate, lipofectamine)
- Select with appropriate antibiotics for 10-14 days
- Confirm bait protein expression and correct subcellular localization by western blotting and immunofluorescence
- For difficult-to-transfect cells, use lentiviral delivery system
Tandem Affinity Purification (Timing: 2 days)
- Day 1: Lyse 5 × 10^7 cells in 1 mL lysis buffer for 30 minutes at 4°C with rotation
- Centrifuge at 13,000 × g for 15 minutes at 4°C and collect supernatant
- Incubate supernatant with 50 μL streptavidin-conjugated beads for 2 hours at 4°C
- Wash beads 3 times with 1 mL lysis buffer
- Incubate beads with 200 μL elution buffer for 30 minutes at 4°C
- Collect eluate and incubate with 30 μL S-protein agarose for 2 hours at 4°C
- Wash S-protein agarose 3 times with 1 mL lysis buffer
- Day 2: Elute bound proteins with 2× SDS sample buffer for western blot or MS-compatible buffer for mass spectrometry
Mass Spectrometry and Data Analysis
- Digest proteins with trypsin (1:50 enzyme-to-protein ratio) overnight at 37°C
- Desalt peptides using C18 stage tips
- Analyze by LC-MS/MS using 2-hour gradient
- Process raw data using MaxQuant or similar software
- Validate interactions through at least two biological replicates and comparison with control purifications
Split-Luciferase Complementation Assay for PPI Dynamics
Principle and Workflow
Split-luciferase complementation assays monitor PPIs by expressing bait and prey proteins fused to complementary fragments of a luciferase enzyme. Upon interaction, the fragments reconstitute, generating a measurable luminescent signal [83]. This method is particularly valuable for high-throughput screening and monitoring interaction dynamics.
Required Materials and Reagents
- Split-Luciferase Vectors: N-terminal and C-terminal luciferase fragments
- Cell Lysis Buffer: Compatible with luciferase activity (avoid strong detergents)
- Luciferase Substrate: D-luciferin or commercial stable substrates
- Detection Platform: Luminescence-compatible microplate reader
- Controls: Known interacting and non-interacting protein pairs
Step-by-Step Protocol
Sensor Design and Validation
- Clone genes of interest into N-terminal and C-terminal luciferase fragment vectors
- Validate fusion protein expression by western blotting
- Test complementation with known interacting partners as positive control
Assay Optimization by 2D Titration
- Co-transfect varying amounts of bait and prey constructs (e.g., 0.1-2.0 μg each)
- Measure luminescence signal 24-48 hours post-transfection
- Identify optimal DNA ratios that maximize signal-to-background ratio
- Determine linear range of the assay for quantitative comparisons
High-Throughput Screening Applications
- Seed cells in 96- or 384-well plates
- Transfer optimized DNA amounts using automated systems
- Add compound libraries 24 hours post-transfection
- Incubate with compounds for desired timeframe (typically 4-24 hours)
- Measure luminescence using automated plate readers
- Calculate Z'-factor to confirm assay robustness (>0.5 is acceptable)
Time-Course Competition Assays
- Express split-luciferase tagged proteins
- Add competing proteins or compounds at different time points
- Monitor luciferase activity continuously or at fixed intervals
- Calculate interaction kinetics and inhibition parameters
Research Reagent Solutions
Table 2: Essential Research Reagents for Multi-Method PPI Analysis
| Reagent Category | Specific Product/System | Function in PPI Analysis | Key Considerations |
|---|---|---|---|
| Affinity Tags | SFB-tag (S-protein, 2×FLAG, SBP) [82] | Tandem purification with high specificity and yield | Small size (84 aa) minimizes protein folding disruption; enables gentle biotin elution |
| Proximity Labeling Enzymes | BioID [82] | Captures proximal interactions in living cells | 310 aa size may affect fusion protein localization; requires biotin supplementation |
| Split-Reporter Systems | Split-Luciferase [83] | Monitors dynamic PPIs in high-throughput format | Signal proportional to interaction strength; suitable for kinetic studies |
| Cell Line Systems | HEK293T [82] [83] | High transfection efficiency for transient and stable expression | Well-characterized background; suitable for most signaling studies |
| Pathway Analysis Databases | PhosphoSitePlus [84] | Provides PTM context for interaction data | Focus on phosphorylation but includes other modifications; human-mouse-rat focus |
| Computational Tools | Pathway Enrichment Algorithms [84] | Identifies signaling pathways from PPI networks | Correct for multiple testing; consider pathway topology in analysis |
Data Integration and Interpretation Framework
Pathway-Centric Analysis of PPI Data
Protein function is dynamically modulated by post-translational modifications (PTMs) that should be studied not in isolation, but in the holistic context of cellular pathways [84]. High-throughput PTM platforms can quantify over 17,000 phosphosites per sample, generating complex datasets that require sophisticated computational integration [84].
Multi-Layer Validation Strategy
- Primary Validation: Confirm interactions identified by one method with an orthogonal technique (e.g., validate TAP-MS hits with split-luciferase)
- Functional Validation: Use genetic approaches (knockdown, knockout, or dominant-negative mutants) to test functional significance
- Pathway Contextualization: Map validated interactions onto known signaling pathways using enrichment tools and prior knowledge networks
- Physiological Relevance: Assess interactions under different physiological conditions (e.g., stress, differentiation, drug treatment)
Addressing Technical Challenges
- Nonspecific Binders: Use tandem purification and comparative analysis with control baits to eliminate sticky proteins [82]
- Weak/Transient Interactions: Employ crosslinking or proximity labeling to capture elusive interactions
- False Positives in High-Throughput Screens: Implement robust statistical cutoffs and require consistency across biological replicates
- Localization Artifacts: Verify correct subcellular localization of tagged proteins to ensure physiological relevance
The complex nature of protein-protein interactions in signaling pathways demands a multi-method approach that leverages complementary techniques. Tandem affinity purification provides high-confidence identification of stable complexes, split-luciferase assays enable dynamic monitoring and high-throughput screening, while proximity labeling captures weak or transient interactions in living cells. Computational integration of these datasets within pathway contexts further enhances biological insights. By implementing the detailed protocols and analytical frameworks presented here, researchers can build comprehensive, high-confidence PPI networks that accelerate signaling pathway analysis and drug development.
Protein-protein interactions (PPIs) are fundamental to most biological processes, controlling cellular mechanisms from gene expression and cell growth to motility and apoptosis [85]. The vast majority of proteins do not function in isolation but rather interact with others—either in stable complexes or through transient associations—to achieve proper biological activity [85]. Understanding these interactions within signaling pathways is therefore critical for elucidating cellular function, disease mechanisms, and drug discovery targets.
Characterizing PPIs presents significant methodological challenges. Interactions can be stable or transient, strong or weak, and occur within diverse cellular compartments [85]. Furthermore, traditional population-averaged readouts, such as western blots, often mask the complex spatial, temporal, and heterogeneous dynamics of signaling pathways that are only apparent at the single-cell level [86]. This application note establishes a comparative framework for evaluating PPI assay methodologies based on three critical parameters: sensitivity (the ability to detect true positive interactions), specificity (the ability to exclude false positives), and throughput (experimental scalability). This framework provides researchers with a structured approach for selecting optimal assays for their specific signaling pathway analysis needs.
Key Methodologies for PPI Analysis in Signaling Pathways
A diverse array of techniques is available for studying PPIs, each with distinct strengths and limitations. The choice of method depends on the nature of the interaction (stable vs. transient), the required physiological context, and the research objectives. The most common methods can be categorized into in vitro affinity purification techniques and in cellulo energy transfer and complementation assays.
Affinity Purification-Based Methods
Co-immunoprecipitation (Co-IP) is a widely used technique for identifying stable or strong protein interactions in a near-native cellular context. In this method, an antibody immobilized on a support binds a target "bait" protein, which then co-precipitates its binding partner "prey" from a cell lysate [85]. The interacting proteins are typically detected by SDS-PAGE and western blot analysis. While co-IP is valuable for validating suspected interactions, associated proteins identified through this method require further verification to confirm their functional relationship to the target antigen [85].
Pull-down assays operate on a similar principle but use a tagged "bait" protein (e.g., GST-, polyHis-, or streptavidin-tagged) immobilized on appropriate beads to purify interacting proteins from a lysate [85]. This approach is particularly useful for studying strong interactions when no suitable antibody is available for co-IP.
Crosslinking is often employed to stabilize transient or weak interactions that might otherwise disassemble during cell lysis and purification steps. Covalent crosslinking of interacting proteins "freezes" the complex, allowing subsequent analysis by co-IP, pull-down assays, electrophoresis, or mass spectrometry while maintaining the original interaction state [85].
Cell-Based Energy Transfer and Complementation Assays
Cell-based assays maintain PPIs within their native environment, preserving subcellular localization, multi-protein complexes, and post-translational modifications. This ensures that identified compounds are cell-permeable and can reach their intracellular targets [87].
Förster Resonance Energy Transfer (FRET) and Bioluminescence Resonance Energy Transfer (BRET) are energy transfer assays where donor and acceptor proteins are fused to interaction partners. PPI complex formation brings the proteins close enough for energy transfer to occur [87]. FRET uses fluorescence energy from a donor protein, while BRET relies on luciferase-induced chemiluminescence as the donor energy source [87].
Bimolecular Protein Complementation Assays utilize reporter proteins split into two fragments that are fused to potential interaction partners. When the proteins interact, the reporter is reconstituted, restoring fluorescence (e.g., in Bimolecular Fluorescent Complementation, BiFC) or enzymatic activity (e.g., in Bimolecular Luminescence Complementation, BiLC) [87]. An important distinction is that FRET, BRET, and BiLC are generally reversible, making them suitable for studying dynamic processes, while BiFC and other enzymatic complementation assays are typically irreversible [87].
Table 1: Comparison of Major PPI Assay Methodologies
| Method | Interaction Type | Context | Sensitivity | Specificity | Throughput | Key Applications in Signaling Pathways |
|---|---|---|---|---|---|---|
| Co-immunoprecipitation (Co-IP) [85] | Stable, Strong | In vitro (Lysate) | Moderate | High | Low-Moderate | Validation of suspected interactions; analysis of protein complexes. |
| Pull-down Assays [85] | Stable, Strong | In vitro (Lysate) | Moderate | High | Low-Moderate | Mapping interactions with purified bait proteins; antibody-free approach. |
| Crosslinking [85] | Transient, Weak | In vitro (Lysate) | High (for transient) | Moderate | Low | Capturing fleeting interactions in signaling cascades. |
| FRET/BRET [87] | Reversible, Dynamic | In cellulo (Live Cells) | High | High | Moderate-High | Real-time kinetics of signaling events; ligand-induced interactions. |
| Fluorescent Protein Complementation (e.g., BiFC) [87] | Stable, Irreversible | In cellulo (Live Cells) | High | Moderate | Moderate-High | Detecting weak interactions; spatial localization of complexes. |
| Enzyme Complementation (e.g., BiLC) [87] | Stable, Irreversible | In cellulo (Live Cells or Lysate) | Very High | Moderate | High | High-throughput screening for drug discovery. |
Advanced Single-Cell and High-Throughput Approaches
Technological advancements now enable researchers to move beyond population-averaged readouts to study signaling pathways at single-cell resolution. These approaches reveal critical insights into cellular heterogeneity and dynamic signaling patterns that are obscured in bulk analyses [86].
Single-Cell Analysis
Fluorescent biosensors are genetically encoded tools that couple fluorescent proteins to activity-sensing domains, allowing real-time monitoring of signaling activities in living cells with high spatio-temporal resolution [86]. For example, kinase activity reporters (e.g., AKAR for PKA, EKAREV for ERK) use FRET backbones to detect phosphorylation events, crucial processes in signaling pathways regulating cell growth, immune response, and apoptosis [86]. Newer designs, such as kinase translocation reporters (KTRs), measure phosphorylation through changes in the subcellular localization of a fluorescent protein, offering an alternative to intensity-based measurements and facilitating multiplexing [86].
Measurement techniques for these biosensors include:
- Fluorescence microscopy for live-cell imaging and capturing signaling dynamics.
- Flow cytometry for quantifying signaling states across thousands of cells at a single time point, with modern systems capable of simultaneously measuring >10 parameters [86].
High-Throughput Signaling Pathway Analysis
Protein array technology provides a platform for high-throughput, multiplexed analysis of signaling pathways. This approach allows simultaneous detection of multiple pathway indicators, dramatically improving detection efficiency [88]. Key applications in signaling pathway analysis include:
- Phosphorylation Status Detection: Using phospho-specific antibodies to evaluate activation states of pathways like MAPK, PI3K/Akt, and JAK/STAT [88].
- Kinase Activity Assay: Utilizing kinase-substrate arrays with peptide substrates to directly measure kinase enzymatic activity [88].
- Pathway Activation/Inhibition Profiling: Quantifying how drugs or genetic perturbations affect entire pathways by monitoring key nodes and downstream effectors [88].
- Cross-Talk Analysis: Studying complex interactions between different signaling pathways (e.g., MAPK, PI3K/Akt, NF-κB) using multi-pathway arrays to identify alternative survival routes or combination therapy targets [88].
Table 2: Performance Metrics of Advanced PPI and Signaling Analysis Platforms
| Platform/Technology | Sensitivity | Specificity | Throughput | Multiplexing Capacity | Primary Readout |
|---|---|---|---|---|---|
| Flow Cytometry [86] | High | High | High (10,000s of cells) | High (≥10 parameters) | Fluorescence intensity per cell |
| Fluorescence Microscopy [86] | High | High | Low-Moderate | Moderate (3-4 colors typically) | Spatio-temporal fluorescence dynamics |
| Protein Array Chips [88] | High (e.g., near-infrared detection) | High | Very High | Very High (Multi-indicator joint testing) | Fluorescence from immobilized probes |
| FRET/BRET (Microplate Reader) [87] | Moderate-High | High | High | Low | Energy transfer ratio |
| Protein Complementation (Luciferase) [87] | Very High | Moderate | High | Low | Luminescence intensity |
Experimental Protocols
Protocol 1: Co-Immunoprecipitation for Stable Complex Analysis
Objective: To isolate and identify proteins in a stable complex with a target protein of interest from a cell lysate.
Materials:
- Lysis Buffer: (e.g., RIPA buffer) supplemented with protease and phosphatase inhibitors.
- Antibody: Specific antibody for the target "bait" protein.
- Beaded Support: Protein A/G magnetic or agarose beads.
- Cell Lysate: Prepared from relevant cell line or tissue.
- Wash Buffer: Mild detergent-based buffer.
- Elution Buffer: Low pH buffer or SDS-PAGE sample buffer.
- Detection Method: SDS-PAGE and western blot reagents.
Procedure:
- Prepare Cell Lysate: Lyse cells in ice-cold lysis buffer. Clarify by centrifugation at 10,000-14,000 × g for 10 minutes at 4°C.
- Pre-clear Lysate: Incubate lysate with beads alone for 30-60 minutes to reduce non-specific binding. Pellet beads and collect supernatant.
- Form Antibody-Bead Complex: Incubate the bait-specific antibody with beaded support for 30-60 minutes. Wash to remove unbound antibody.
- Immunoprecipitation: Incubate the antibody-bound beads with the pre-cleared lysate for 1-2 hours at 4°C with gentle agitation.
- Wash Complexes: Pellet beads and wash 3-5 times with wash buffer to remove non-specifically bound proteins.
- Elute Bound Proteins: Add elution buffer and heat to 95-100°C for 5-10 minutes to dissociate the immune complexes.
- Analyze: Separate eluted proteins by SDS-PAGE and perform western blot analysis to detect the bait and co-precipitated "prey" proteins.
Protocol 2: FRET-Based Kinase Activity Assay in Live Cells
Objective: To measure the dynamic activity of a specific kinase (e.g., ERK) in live cells in response to stimuli using a FRET biosensor.
Materials:
- FRET Biosensor DNA: Plasmid encoding kinase activity reporter (e.g., EKAREV for ERK).
- Cell Line: Appropriate mammalian cell line (e.g., HEK293, HeLa).
- Transfection Reagent: For introducing DNA into cells.
- Imaging Equipment: Fluorescence microscope capable of ratio-metric imaging and appropriate filter sets for FRET pairs (e.g., CFP/YFP).
- Stimuli/Inhibitors: Pathway-specific activators or inhibitors.
Procedure:
- Cell Preparation and Transfection: Plate cells on imaging-appropriate dishes (e.g., glass-bottom). Transfect with the FRET biosensor DNA.
- Microscope Setup: Configure microscope for time-lapse imaging, with settings for CFP (donor) and YFP (acceptor) channels. Use minimal excitation light to minimize photobleaching and phototoxicity.
- Acquire Baseline: Image cells for 10-30 minutes to establish baseline FRET ratios. The FRET ratio is typically calculated as (YFP emission with CFP excitation)/(CFP emission with CFP excitation).
- Apply Stimulus: Add pathway stimulant (e.g., EGF for ERK activation) or inhibitor directly to cells during continuous imaging.
- Monitor Kinase Activity: Continue time-lapse imaging to capture changes in the FRET ratio, which inversely correlates with kinase activity.
- Data Analysis: Calculate FRET ratios over time for individual cells. Analyze single-cell trajectories to reveal heterogeneous responses and dynamic patterns within the population.
Visualization of a Generalized Signaling Pathway and Experimental Workflow
The following diagrams, generated using Graphviz DOT language, illustrate a simplified growth factor signaling pathway and a generalized workflow for selecting and implementing PPI assays.
Growth Factor Signaling Pathway
PPI Assay Selection and Workflow
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for PPI and Signaling Analysis
| Reagent/Material | Function/Application | Example Uses |
|---|---|---|
| Phospho-Specific Antibodies [88] | Detect phosphorylation status of specific proteins to evaluate pathway activation. | Western blot, Co-IP, protein arrays for MAPK, Akt, STAT pathways. |
| Protein A/G Magnetic Beads [85] | Solid support for antibody immobilization during immunoprecipitation. | Co-IP for isolating protein complexes from lysates. |
| FRET Biosensor Plasmids [86] [87] | Genetically encoded reporters for dynamic kinase activity in live cells. | EKAREV (ERK), AKAR (PKA) for single-cell signaling dynamics. |
| Crosslinking Reagents [85] | Stabilize transient protein interactions for subsequent analysis. | Homobifunctional, amine-reactive crosslinkers to capture fleeting PPIs. |
| Protease & Phosphatase Inhibitors [85] | Preserve protein integrity and post-translational modifications during lysis. | Added to lysis buffers for Co-IP, pull-downs, and sample prep for arrays. |
| Kinase-Substrate Peptide Arrays [88] | Profile kinase enzymatic activity and screen for kinase inhibitors. | High-throughput kinome profiling in disease or drug response. |
| Lectin Arrays [88] | Detect protein glycosylation patterns, a key modification controlling signaling. | Characterize glycoprotein modifications on receptors or adhesion molecules. |
| Near-Infrared Fluorescent Dyes [88] | Provide stable, high-sensitivity signals for multiplexed detection. | Protein chip detection platforms for high-throughput signaling analysis. |
Selecting the appropriate methodology for analyzing protein-protein interactions in signaling pathways requires careful consideration of the trade-offs between sensitivity, specificity, and throughput. Traditional techniques like co-IP offer high specificity for validation studies, while modern cell-based assays (FRET, BRET, complementation) provide the sensitivity and dynamic range needed for functional analysis in physiologically relevant contexts. For comprehensive signaling pathway profiling, particularly in drug discovery, high-throughput platforms like protein arrays are unmatched in their multiplexing capacity and efficiency.
A robust strategy often involves a combination of methods: using high-throughput screens to identify potential interactions or modulators, followed by lower-throughput, high-specificity validation and detailed mechanistic studies in live cells. Furthermore, leveraging single-cell technologies is crucial for unraveling the complex heterogeneity and temporal dynamics inherent in cellular signaling networks, moving beyond the limitations of population-averaged data. By applying this comparative framework, researchers can make informed decisions to optimally design experiments that accurately characterize PPIs and advance our understanding of signaling pathway biology.
Protein-protein interactions (PPIs) are fundamental regulators of cellular function, forming the backbone of signal transduction networks that control cell fate, proliferation, and response to external stimuli. For researchers investigating signaling pathways, systematic PPI analysis provides an essential framework for understanding the functional organization of the proteome and identifying novel components of regulatory circuits [89]. The experimental characterization of PPIs through methods such as yeast two-hybrid screening, co-immunoprecipitation, and mass spectrometry has generated vast interaction datasets [1]. These data are compiled, curated, and expanded in public databases that have become indispensable resources for the research community. Among these, STRING, BioGRID, and the Database of Interacting Proteins (DIP) provide complementary approaches to PPI data compilation, validation, and analysis, enabling researchers to build robust interaction networks for hypothesis generation and experimental validation in signaling pathway research.
Database Characteristics and Comparative Analysis
STRING (Search Tool for the Retrieval of Interacting Genes/Proteins) compiles, scores, and integrates protein-protein association information drawn from experimental assays, computational predictions, and prior knowledge. Its goal is to create comprehensive and objective global networks that encompass both physical and functional interactions [90]. The database incorporates multiple evidence channels including gene neighborhood, gene fusions, co-expression, experimental data, annotated pathways, and text mining [91]. Each interaction receives a confidence score between 0-1, with 0.5 indicating approximately 50% likelihood of being a false positive [91]. The latest version introduces regulatory networks with directionality of regulation and offers downloadable network embeddings for machine learning applications [90].
BioGRID (Biological General Repository for Interaction Datasets) is an open-access repository of protein, genetic, and chemical interactions curated from high-throughput datasets and individual focused studies [92]. The database maintains themed curation projects focusing on specific biological processes with disease relevance, including synthetic protein interactions, autism spectrum disorder, Alzheimer's Disease, COVID-19 Coronavirus, and signaling pathways such as the ubiquitin-proteasome system [92] [93]. As of late 2025, BioGRID contains over 2.25 million non-redundant interactions from more than 87,000 publications [92].
DIPS (Database of Interacting Protein Structures) and its enhanced version DIPS-Plus focus on structurally resolved protein complexes, providing atomic-level and residue-level features for machine learning of protein interfaces [94]. The database contains 42,112 complexes with multiple residue-level features including surface proximities, half-sphere amino acid compositions, and profile hidden Markov model-based sequence features [94]. This structural focus makes it particularly valuable for understanding the physical basis of interactions in signaling complexes and for predicting the functional consequences of genetic variations.
Quantitative Comparison of Database Content
Table 1: Comparative Analysis of Major PPI Databases
| Feature | STRING | BioGRID | DIPS-Plus |
|---|---|---|---|
| Primary Focus | Functional & physical associations | Curated physical & genetic interactions | Structurally resolved complexes |
| Interaction Count | ~210,914 (E. coli example) | ~2,251,953 non-redundant interactions | 42,112 complexes |
| Evidence Types | Experiments, text mining, predictions, pathways | Manual curation from literature | X-ray, NMR, EM structures |
| Coverage Scope | 14,000+ organisms | Multiple species focus | Structural complexes |
| Confidence Scoring | Combined score (0-1) with evidence channels | No scoring system | Interface residue annotations |
| Special Features | Regulatory directions, pathway enrichment | CRISPR screens, themed projects | Residue-level features for ML |
| Data Availability | CC BY 4.0, APIs, full downloads | Monthly updates, web services | CC BY 4.0 |
| Update Frequency | Periodic major releases | Monthly curation updates | Expanded versions |
Table 2: STRING Evidence Channel Distribution for E. coli K12 MG1655 (Score ≥0.400)
| Evidence Channel | Normal | Transferred |
|---|---|---|
| Gene Neighborhood | 7,851 | 11,177 |
| Gene Fusion | 514 | - |
| Gene Cooccurrence | 35,497 | - |
| Gene Coexpression | 12,376 | 3,154 |
| Experiments/Biochemistry | 5,301 | 4,113 |
| Annotated Pathways | 6,726 | 1,727 |
| Textmining | 27,445 | 7,119 |
| Total | 210,914 |
Database-Specific Protocols for Signaling Pathway Analysis
STRING Protocol for Pathway-Centric Network Construction
Objective: To identify functionally enriched interaction networks within a specific signaling pathway using STRING's integrated association scores.
Procedure:
- Input Preparation: Compile a list of core proteins from your signaling pathway of interest using UniProt identifiers or gene symbols.
- Database Query: Access STRING (https://string-db.org/) and input your protein list using the "Multiple Proteins" search option.
- Organism Selection: Specify the relevant model organism to limit interactions to appropriate orthologs.
- Confidence Thresholding: Set the minimum interaction score to 0.7 (high confidence) to reduce false positives.
- Evidence Filtering: Use the "Data Settings" tab to select evidence channels relevant to your research question:
- For physical interactions: Select "Experiments" and "Databases"
- For functional associations: Select "Textmining" and "Co-expression"
- Network Analysis: Execute the search and utilize STRING's functional enrichment tools to identify overrepresented GO terms and pathways.
- Validation: Examine individual interaction evidence by clicking on network edges to review source publications and experimental methods.
- Export: Download the interaction network in PSI-MI TAB 2.5 format for further analysis in Cytoscape.
Application Note: This protocol is particularly effective for placing novel signaling components within established pathways and identifying potential crosstalk mechanisms between parallel signaling cascades [91] [90].
BioGRID Protocol for Experimental Validation of Physical Interactions
Objective: To retrieve empirically validated physical interactions for hypothesis testing and experimental design.
Procedure:
- Target Identification: Identify key signaling proteins of interest using standard gene nomenclature.
- Advanced Search: Access BioGRID (http://thebiogrid.org/) and utilize the "Advanced Search" feature with the following parameters:
- Organism: [Your species of interest]
- Evidence Type: "Physical Interaction"
- Method: Specify appropriate experimental systems (e.g., Two-hybrid, Co-immunoprecipitation, Affinity Capture)
- Themed Project Screening: Check the "Themed Curation Projects" for relevant signaling pathways (e.g., Ubiquitin-Proteasome System, Kinome).
- Data Filtering: Apply filters to focus on high-throughput studies or manually curated small-scale studies based on research needs.
- Interaction Validation: Cross-reference identified interactions with experimental details in source publications.
- CRISPR Integration: For functional validation, consult the BioGRID ORCS database to identify relevant CRISPR screening data.
- Export Format: Download interactions in MITAB format for compatibility with most network visualization tools.
Application Note: This protocol provides a foundation for designing co-immunoprecipitation experiments to confirm suspected interactions in signaling cascades, with BioGRID offering specific methodological details from source publications [92] [89].
DIPS-Plus Protocol for Structural Analysis of Signaling Complexes
Objective: To identify interface residues and structural features of signaling complexes for mutagenesis studies and drug discovery.
Procedure:
- Complex Identification: Access DIPS-Plus dataset through its GitHub repository or publication supplements.
- Structure Filtering: Filter complexes by experimental method (X-ray, NMR, EM) and resolution quality where applicable.
- Feature Extraction: Utilize provided scripts to extract residue-level features including:
- Surface accessibility
- Half-sphere amino acid compositions
- Secondary structure elements
- HMM-based evolutionary features
- Interface Prediction: Apply built-in machine learning models or train custom classifiers using the provided benchmark protocols.
- Visualization: Generate structure diagrams highlighting predicted interface regions using molecular visualization software.
- Mutation Analysis: Corrogate interface predictions with known pathological mutations from ClinVar or COSMIC.
Application Note: This structural approach is particularly valuable for understanding the molecular basis of dominant-negative mutations in signaling proteins and for rational design of interface-disrupting peptides [94].
Integrated Workflow for Signaling Pathway Validation
The following diagram illustrates a comprehensive workflow for validating signaling pathway components using complementary information from STRING, BioGRID, and DIPS-Plus:
Integrated Database Validation Workflow
Table 3: Essential Research Reagents for PPI Validation in Signaling Pathways
| Reagent/Resource | Function in PPI Validation | Application Examples |
|---|---|---|
| Phospho-specific Antibodies | Detection of signaling pathway activation states | Western blot, immunofluorescence for MAPK, PI3K/Akt pathways [88] |
| Co-immunoprecipitation Kits | Empirical validation of physical interactions | Validation of STRING-predicted interactions [95] |
| PathScan ELISA Kits | Multiplexed signaling node analysis | Simultaneous detection of multiple phosphorylated signaling proteins [95] |
| CRISPR/Cas9 Systems | Functional validation of PPIs | Gene editing to test essentiality of BioGRID-curated interactions [92] |
| Near-Infrared Protein Arrays | High-throughput PPI screening | Signaling pathway activation/inhibition profiling [88] |
| Yeast Two-Hybrid Systems | Binary interaction detection | Validation of putative interactions from all databases [89] |
| Structural Visualization Tools | Analysis of interaction interfaces | Visualization of DIPS-Plus structural data [94] |
Case Studies in Signaling Pathway Research
Wnt Signaling Pathway Elucidation
A seminal study demonstrating the integration of database resources focused on the Wnt signaling pathway [89]. Researchers began with a core set of known Wnt pathway components and expanded this network using STRING's functional associations. Through BioGRID curation, they identified novel Axin-1 interactions with ANP32A and CRMP1, which were subsequently validated experimentally as modulators of Wnt signaling [89]. This systematic approach connected previously uncharacterized gene products to established disease-relevant pathways, showcasing how PPI databases can drive discovery in signaling biology.
Virus-Host Interaction Signaling
Recent advances in protein language models like PLM-interact have demonstrated exceptional performance in predicting virus-host PPIs, achieving state-of-the-art results in cross-species benchmarks [48]. This approach is particularly valuable for understanding how viral proteins hijack host signaling networks. By training on human PPI data and testing on divergent species, researchers validated the model's ability to identify interaction interfaces relevant to infectious disease mechanisms [48].
Signaling Network Connectivity Analysis
Research on lysine acetylation revealed that this post-translational modification preferentially targets large macro-molecular complexes with broad regulatory scope [91]. Using STRING, the authors demonstrated that the acetylome has significantly higher network connectivity than random expectations (roughly six interactions per node versus less than three expected by chance) [91]. This systems-level analysis provided insights into how acetylation regulates signaling pathway crosstalk and coordinated cellular functions.
Emerging Technologies and Future Directions
The field of PPI analysis is rapidly evolving with several technological advances shaping future approaches to signaling pathway research. Protein language models (PLMs) like ESM-2 and their extensions (e.g., PLM-interact) now enable more accurate prediction of PPIs from sequence alone, with architectures that jointly encode protein pairs to learn their relationships [48]. Deep learning approaches, particularly graph neural networks (GNNs), are being applied to structural data from resources like DIPS-Plus to predict interaction interfaces with increasing accuracy [1]. For experimental validation, BioGRID's themed curation projects now include synthetic protein interactions, tracking de novo designed proteins that interact with specific targets [93]. These AI-designed proteins and binders represent novel research tools and potential therapeutic agents for modulating signaling pathways.
The integration of these advanced computational approaches with comprehensive database resources is creating new paradigms for signaling pathway analysis. Researchers can now move from static interaction maps to dynamic models that incorporate structural information, evolutionary constraints, and contextual cellular data. As these resources continue to expand and integrate multi-omics data, they will provide increasingly powerful platforms for understanding the complex signaling networks that underlie both normal physiology and disease states.
Protein-protein interactions (PPIs) are fundamental regulators of cellular function, acting as the molecular basis for signal transduction, cell cycle regulation, and transcriptional control [1]. In cancer, these intricate interaction networks become dysregulated, driving disease pathogenesis. Mapping PPIs within signaling pathways therefore provides crucial insights into oncogenic mechanisms and reveals potential therapeutic targets [96] [97]. This application note details a structured framework for integrating computational and experimental PPI analysis to map signaling pathways in cancer, using a case study on the natural compound naringenin in breast cancer. We summarize key methodologies, provide validated experimental protocols, and outline essential bioinformatics tools to support researchers in conducting comprehensive PPI-based pathway analysis.
Key PPI Assays and Methodologies
Diverse biochemical, genetic, and cell biological methods have been developed to map interactomes, each with distinct strengths and limitations. Selecting the appropriate method requires careful consideration of research goals, the nature of the PPIs being studied, and available resources [96]. The table below summarizes major PPI mapping technologies used in signaling pathway analysis.
Table 1: Key Protein-Protein Interaction Assays for Signaling Pathway Analysis
| Assay Name | Principle | Key Advantages | Key Limitations | Best Suited For |
|---|---|---|---|---|
| Yeast Two-Hybrid (Y2H) | Reconstitution of transcription factor via DNA-Binding and Activation Domain fusion proteins [96]. | Simple, established, low-cost; scalable for large-scale screening; in vivo environment [96]. | High false-positive rate; requires nuclear localization; may lack PTMs from native organism [96]. | Discovery of novel binary interactions. |
| Membrane Yeast Two-Hybrid (MYTH) | Split-ubiquitin system reconstitution, releasing a transcription factor [96]. | Designed for membrane proteins; in vivo context. | Can be technically challenging. | Studying interactions involving full-length membrane proteins. |
| Affinity Purification Mass Spectrometry (AP-MS) | Purification of protein complexes via tagged bait, followed by MS identification [96]. | Identifies co-complex associations; detects interactions under near-physiological conditions. | Cannot distinguish direct from indirect interactions; false positives from contaminants. | Mapping protein complexes and interactomes. |
| BioID-MS | Proximity-based biotinylation using a promiscuous biotin ligase fused to bait protein [96]. | Captures transient, weak interactions; labels proximal proteins in living cells. | Identifies proximity, not necessarily direct binding. | Identifying proximal proteins and weak/transient interactions. |
No single method can capture the full complexity of the interactome. Combining complementary assays maximizes the coverage of true positive interactions while maintaining high specificity [98]. For instance, integrating binary interaction data from Y2H with co-complex data from AP-MS provides a more comprehensive network view.
Integrated Workflow for PPI Mapping in Cancer Pathways
The following diagram illustrates the integrated computational and experimental workflow for mapping cancer-related signaling pathways using PPI data, as demonstrated in the naringenin case study [97].
Case Study: Mapping Naringenin's Action in Breast Cancer
A recent study on the flavanone naringenin (NAR) provides a prototypical example of this integrated approach to elucidate a natural compound's anti-cancer mechanism in breast cancer [97].
Computational Target Identification and Network Analysis
- Target Screening: Researchers identified 62 overlapping protein targets between naringenin and breast cancer using SwissTargetPrediction, STITCH, GeneCards, OMIM, and CTD databases [97].
- PPI Network Construction: These 62 common targets were used to construct a protein-protein interaction network from the STRING database, which was visualized and analyzed using Cytoscape software [97].
- Topological Analysis: Using the CytoNCA plugin in Cytoscape, researchers performed topological analysis calculating degree centrality, betweenness centrality, closeness centrality, and eigenvector centrality to identify the most influential nodes (proteins) within the network [97].
- Pathway Enrichment: Gene Ontology (GO) and KEGG pathway enrichment analysis revealed that naringenin's potential targets were significantly involved in the PI3K-Akt signaling pathway and the MAPK signaling pathway, both critically implicated in breast cancer pathogenesis [97].
Table 2: Core Targets Identified in the Naringenin Breast Cancer Case Study
| Target Gene | Protein Name | Degree Centrality | Key Role in Cancer | Binding Affinity with Naringenin (kcal/mol) |
|---|---|---|---|---|
| SRC | Proto-oncogene tyrosine-protein kinase SRC | High | Regulates cell proliferation, survival, motility, and invasion [97]. | -9.1 [97] |
| PIK3CA | Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha | High | Central node in PI3K-Akt pathway; promotes cell growth and survival [97]. | -8.5 [97] |
| BCL2 | B-cell lymphoma 2 | Medium | Anti-apoptotic protein; inhibits programmed cell death [97]. | -7.8 [97] |
| ESR1 | Estrogen receptor | Medium | Hormone receptor driving a major subtype of breast cancer [97]. | -8.2 [97] |
Experimental Validation
- Molecular Docking: Confirmed strong binding affinities between naringenin and the core targets SRC, PIK3CA, BCL2, and ESR1 [97].
- Molecular Dynamics (MD) Simulations: MD simulations further validated these interactions, confirming stable binding conformations and favorable binding free energies over time, with SRC emerging as a particularly stable complex [97].
- In Vitro Functional Assays: Using MCF-7 human breast cancer cells, the study demonstrated that naringenin inhibits proliferation, induces apoptosis, reduces cell migration, and increases reactive oxygen species (ROS) generation, thereby confirming the computationally predicted anti-cancer phenotypes [97].
Detailed Experimental Protocols
Protocol: Constructing a Pathway-Specific PPI Network
This protocol describes the construction and analysis of a PPI network from a list of candidate genes, using the naringenin study as a template [97].
Research Reagent Solutions & Materials:
- Computer with internet access
- Gene list: A set of genes/proteins of interest (e.g., from omics studies).
- STRING database: A public resource for known and predicted PPIs.
- Cytoscape software: An open-source platform for network visualization and analysis.
- Cytoscape Plugins: CytoNCA (for topological analysis).
Procedure:
- Input Gene List: Compile a non-redundant list of gene symbols for your proteins of interest.
- Query the STRING Database:
- Navigate to the STRING website (https://string-db.org/).
- Select "Multiple Proteins" and enter your gene list.
- Set the organism to Homo sapiens.
- Set the minimum required interaction score to "High confidence (0.700)" to increase the quality of interactions [97].
- Download the resulting interaction data in a format compatible with Cytoscape (e.g., .tsv or .xgmml).
- Import and Visualize in Cytoscape:
- Open Cytoscape and import the network file.
- Apply a force-directed layout algorithm (e.g., "Prefuse Force Directed Layout") to visualize clusters and highly connected nodes.
- Perform Topological Analysis:
- Install the CytoNCA plugin via the Cytoscape App Manager.
- Run CytoNCA to calculate key network centrality measures for each node: Degree, Betweenness, Closeness, and Eigenvector.
- Filter nodes to identify core targets by selecting those with values above the network's average for all four centralities [97].
Protocol: Yeast Two-Hybrid Screening for Binary Interactions
The Y2H system is a powerful genetic method for detecting binary PPIs [96].
Research Reagent Solutions & Materials:
- Y2H Strains: Saccharomyces cerevisiae strains with engineered reporter systems (e.g., AH109, Y2HGold).
- Bait and Prey Vectors: Plasmids for expressing proteins as fusions with DNA-Binding Domain (BD) and Activation Domain (AD).
- Selective Media: Synthetic Dropout (SD) media lacking specific nutrients (e.g., -Leu/-Trp for selection of plasmids, -Leu/-Trp/-His/-Ade for interaction selection).
- cDNA Library: A library of cDNAs cloned into the prey vector for screening.
Procedure:
- Clone Bait Gene: Clone the gene of interest (bait) into the BD vector. Verify the construct by sequencing.
- Transform Bait into Yeast: Introduce the bait plasmid into the appropriate yeast strain and select on SD/-Trp (or appropriate selective medium). Test for autoactivation of reporter genes.
- Mate with Prey Library:
- The bait strain is mated with a yeast strain pre-transformed with the prey cDNA library.
- Diploid yeast cells are selected on SD/-Leu/-Trp medium.
- Screen for Interactions:
- Plate the mated yeast cells on high-stringency selective medium (SD/-Leu/-Trp/-His/-Ade). Only yeast cells where the bait and prey proteins interact will activate the reporter genes and grow.
- Colonies that grow are picked, and the prey plasmid is isolated and sequenced to identify the interacting protein partner.
- Validation: Confirm positive interactions using secondary assays, such as β-galactosidase tests, to minimize false positives.
Visualization and Analysis Tools
Effective visualization is critical for interpreting complex PPI networks. Several web-based and standalone tools are available.
Table 3: Key Resources for PPI Data Visualization and Analysis
| Resource Name | Type | Key Features | Use Case |
|---|---|---|---|
| Cytoscape [99] [100] | Standalone Software | Highly customizable network visualization; vast app ecosystem for analysis; handles large datasets. | In-depth, customizable network analysis and figure generation. |
| STRING [99] [97] | Web Database / Viewer | Integrated interaction data from multiple sources; user-friendly; direct pathway and functional enrichment. | Initial network construction and functional annotation. |
| IntAct [99] | Web Database / Viewer | Open-source database; provides molecular interaction data; integrated visualization with Cytoscape Web. | Accessing curated, experimental PPI data. |
| SFARI Gene PIN [101] | Specialized Web Resource | Features a "Ring Browser" for visualizing curated ASD-related protein interactions; manually curated data. | Exploring specific, high-quality curated networks in neurobiology. |
The following diagram illustrates the data flow and key steps in the visualization and analysis of a PPI network using these tools.
The integrated workflow presented here, combining computational PPI network analysis with targeted experimental validation, provides a powerful framework for deconstructing complex signaling pathways in cancer. The naringenin case study demonstrates how this approach can generate testable hypotheses, identify key regulatory nodes (like SRC and PIK3CA), and ultimately elucidate the mechanism of action of therapeutic compounds. As deep learning models continue to advance, their integration with these established methods promises to further enhance the accuracy and scope of PPI prediction and pathway mapping, accelerating oncology drug discovery [1].
{#compilation-topic}
Benchmarking Computational Predictions Against Experimental Gold Standards
This application note provides a detailed protocol for the validation of computational protein-protein interaction (PPI) predictions against experimental gold standards. Within signaling pathway research, accurate PPI data are critical for understanding cellular processes, including signal transduction, stress responses, and metabolic control [17]. The framework outlined herein covers the selection of experimental reference datasets, the execution of major computational prediction methods, and the implementation of a rigorous quantitative benchmarking workflow. By integrating guidelines from comprehensive benchmarking studies [102] and leveraging recent advancements in machine learning (ML) for PPIs [5], this document aims to equip researchers with a standardized approach for assessing the reliability and applicability of computational tools in drug discovery and systems biology.
Protein-protein interactions form the backbone of cellular signaling networks. The majority of genes and proteins realize resulting phenotype functions as a set of interactions [17]. Computational methods for predicting PPIs have emerged as powerful, high-throughput complements to traditional experimental approaches, which are often resource-intensive and less scalable [5] [17]. The central challenge, however, lies in ensuring these computational predictions are accurate and biologically relevant.
Rigorous benchmarking is the cornerstone of validating computational methods. It involves the systematic comparison of method performance using well-characterized reference datasets to determine strengths and weaknesses and to provide actionable recommendations [102]. In fast-moving fields, the establishment of sustained, community-driven benchmarking frameworks, akin to the Critical Assessment of Structure Prediction (CASP) challenge, is instrumental for tracking progress [103]. This is particularly true for PPI predictions in signaling pathways, where errors can propagate through network models and lead to incorrect biological conclusions. This protocol provides a comprehensive, practical guide for conducting such benchmarking exercises, framed within the context of signaling pathway analysis.
Background Concepts
The Role of PPIs in Signaling Pathways
In the context of signaling pathways, PPIs are often transient and controlled by specific conditions, such as post-translational modifications, which can alter interaction affinities and specificities [85] [5]. Proteins involved in the same cellular processes are repeatedly found to interact with each other [17]. The result of two or more proteins interacting can:
- Modify the kinetic properties of enzymes.
- Create a new binding site for small effector molecules.
- Change the specificity of a protein for its substrate.
- Serve a regulatory role in an upstream or downstream event [85] [17].
Categories of PPI Detection Methods
PPI detection methods are broadly classified into three categories, each with a distinct role in generating data for benchmarking:
- In Vivo Methods: Techniques like Yeast Two-Hybrid (Y2H) are performed in a living organism and are ideal for identifying potential interactions within a cellular context [17].
- In Vitro Methods: Techniques such as Co-immunoprecipitation (Co-IP) and affinity purification are conducted in a controlled environment outside a living organism. These are excellent for confirming direct physical interactions and isolating stable complexes [85] [17].
- In Silico Methods: Computational techniques that use sequence, structure, genomic context, or other biological data to predict interactions. These include sequence-based approaches, structure-based approaches, and machine learning models [5] [17].
Experimental Gold Standards and Reference Data
The performance of any computational benchmark is largely determined by the quality of the reference data used for validation [102].
Key Experimental Techniques for Ground Truth
The following table summarizes core experimental methods used to generate high-confidence PPI data for benchmarking.
Table 1: Key Experimental Methods for PPI Validation
| Method | Category | Key Principle | Application in Benchmarking |
|---|---|---|---|
| Yeast Two-Hybrid (Y2H) [17] | In Vivo | Detects interactions by reconstituting a functional transcription factor. | Excellent for large-scale interaction discovery and mapping networks. |
| Co-immunoprecipitation (Co-IP) [85] [17] | In Vitro | Uses an antibody against a "bait" protein to co-precipitate its binding partners ("prey") from a cell lysate. | Confirms interactions in a near-native cellular context; ideal for validating complex formations. |
| Pull-Down Assays [85] | In Vitro | Uses an immobilized "bait" protein (e.g., GST-tagged) to purify binding partners from a lysate. | Useful for studying strong/stable interactions when no antibody is available for Co-IP. |
| Tandem Affinity Purification (TAP) [17] | In Vitro | Involves double-tagging a protein of interest for a two-step purification process, followed by MS analysis. | Identifies components of multi-protein complexes under intrinsic cellular conditions. |
| Crosslinking [85] | In Vitro | Uses covalent crosslinkers to stabilize transient protein interactions before analysis. | Captures fleeting interactions that might be lost during other purification methods. |
Curating and Managing Reference Datasets
A critical step is the compilation and curation of PPIs from public databases and literature to create a unified benchmark set.
- Data Sources: Key resources include databases like BIOGRID and STRING for known interactions, and rice-specific resources like RicePPINet for species-focused studies [5]. Structural data from AlphaFold predictions can also provide valuable features for benchmarking structure-based methods [5].
- Data Curation: A rigorous, automated procedure is essential. This includes:
- Standardization: Converting chemical structures to a standard format (e.g., SMILES) and neutralizing salts [104].
- Removal of Inorganics: Identifying and removing inorganic and organometallic compounds [104].
- Handling Duplicates: Averaging experimental values for duplicates with low standard deviation and removing those with ambiguous or highly variable values [104].
- Outlier Removal: Using statistical methods like Z-score calculation to exclude intra- and inter-dataset outliers [104].
Computational PPI Prediction Methods
Computational methods can be grouped based on the input data they use. The selection of methods for benchmarking should be comprehensive and unbiased [102].
Table 2: Categories of Computational PPI Prediction Methods
| Method Category | Key Principle | Example Features | Applicability |
|---|---|---|---|
| Sequence-Based [5] [17] | Predicts interactions based on information encoded in the protein sequence. | Amino acid composition, dipeptide frequency, physiochemical properties, evolutionary coupling. | Broadly applicable, especially when 3D structures are unknown. |
| Structure-Based [5] | Leverages protein 3D structures to identify potential binding interfaces. | Surface topology, complementary shape, residue-residue contacts, docking scores. | Highly accurate for proteins with known or reliably predicted structures (e.g., via AlphaFold). |
| Genomic Context-Based [17] | Infers functional linkage based on genomic patterns. | Gene fusion, gene neighborhood, phylogenetic profiles. | Useful for predicting interactions in conserved pathways. |
| Machine Learning (ML) [5] | Uses algorithms to learn complex patterns from labeled training data (positive and negative PPIs). | Combinations of sequence, structure, and genomic features. Random Forest (RF) and Support Vector Machine (SVM) are widely used. | Powerful for integrating diverse data types and making large-scale predictions; performance depends on feature selection and training data quality. |
The Benchmarking Protocol
This section details a step-by-step protocol for executing a PPI prediction benchmark.
Defining Scope and Selecting Methods
- Purpose: Clearly define the benchmark's goal (e.g., comparing new ML models against established tools or general benchmarking of all available methods) [102].
- Method Selection: For a neutral benchmark, include all available methods that meet predefined criteria (e.g., software availability, usability). Justify the exclusion of any widely used methods. When introducing a new method, compare it against a representative subset of state-of-the-art and baseline methods [102].
Dataset Preparation and Curation
- Acquire Data: Collect PPIs from curated sources listed in Section 3.2.
- Define Negative Samples: A critical and challenging step. Common strategies include:
- Randomly pairing proteins from different subcellular compartments.
- Selecting proteins with distinct localizations to make physical interaction unlikely [5].
- Apply Curation Pipeline: Implement the data curation steps outlined in Section 3.2 using tools like RDKit for structure standardization [104].
- Split Datasets: Partition the curated data into training and hold-out test sets. Use robust validation schemes like Leave-One-Protein-Out (LOPO) to assess a model's ability to predict interactions for novel proteins [5].
Quantitative Performance Assessment
- Run Predictions: Execute all selected computational methods on the hold-out test set.
- Calculate Metrics: Compute standard performance metrics by comparing predictions to the experimental ground truth.
Table 3: Key Quantitative Metrics for PPI Prediction Benchmarking
| Metric | Formula/Principle | Interpretation |
|---|---|---|
| Accuracy | (TP + TN) / (TP + TN + FP + FN) | Overall correctness of the model. |
| Precision | TP / (TP + FP) | Proportion of predicted interactions that are correct. |
| Recall (Sensitivity) | TP / (TP + FN) | Proportion of true interactions that were correctly predicted. |
| F1-Score | 2 * (Precision * Recall) / (Precision + Recall) | Harmonic mean of precision and recall. |
| Area Under the Curve (AUC) | Area under the Receiver Operating Characteristic (ROC) curve. | Overall performance across all classification thresholds. |
| Balanced Accuracy [104] | (Sensitivity + Specificity) / 2 | Useful for imbalanced datasets. |
TP: True Positive; TN: True Negative; FP: False Positive; FN: False Negative
Evaluation and Interpretation
- Rank Methods: Rank methods based on the primary evaluation metrics (e.g., F1-score, AUC) [102].
- Analyze Trade-offs: Examine secondary measures such as computational runtime, scalability, and user-friendliness. Highlight different strengths and trade-offs among the top-performing methods [102].
- Assess Applicability Domain: Evaluate whether the model's predictions are reliable for the specific chemical space of interest, as performance can vary across different types of proteins and chemicals [104].
Workflow Visualization
The following diagram illustrates the complete benchmarking workflow, from data preparation to final evaluation.
Benchmarking Workflow for PPI Predictions
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Research Reagents and Materials
| Item | Function in PPI Analysis |
|---|---|
| Specific Antibodies [85] | Essential for Co-IP to immunoprecipitate the "bait" protein and its interacting partners. |
| Tagged Fusion Proteins (GST-, PolyHis-) [85] | Used as "bait" in pull-down assays; the tag allows for immobilization on appropriate beads. |
| Protein A/G Magnetic Beads [85] | Provide a solid support for antibody immobilization during Co-IP, simplifying washing and elution. |
| Crosslinking Reagents [85] | Homobifunctional, amine-reactive crosslinkers (e.g., DSS) stabilize transient PPIs prior to lysis and analysis. |
| Protease and Phosphatase Inhibitors | Added to cell lysis buffers to preserve the native state of proteins and their modifications during interaction studies. |
| Mass Spectrometry-Grade Reagents | Required for the sensitive and accurate identification of co-precipitated proteins by mass spectrometry. |
Implementation Guide
- Software and Tools: Utilize standardized cheminformatics toolkits like RDKit [104] for data curation. For ML-based predictions, common frameworks include Scikit-learn for classical models and TensorFlow/PyTorch for deep learning.
- Best Practices:
- Blinding: Where possible, use blinding to avoid bias during method evaluation and parameter tuning [102].
- Reproducibility: Document all software versions, parameters, and curation scripts. Use containerization (e.g., Docker) to ensure reproducible computational environments [102].
- Chemical Space Analysis: Validate that your benchmark dataset covers the chemical space relevant to your research (e.g., approved drugs, industrial chemicals) by plotting it against a reference space using techniques like PCA [104].
- Troubleshooting:
- Low Performance Across Methods: Re-evaluate the quality of the negative dataset and the relevance of the chosen features for the prediction task.
- High Variance in Results: Ensure the dataset is large and diverse enough. Implement stricter cross-validation schemes like LOPO [5].
Conclusion
Mastering protein-protein interaction assays is fundamental to advancing our understanding of cellular signaling and developing novel therapeutics. A strategic approach that combines foundational knowledge with carefully selected methodological tools, rigorous troubleshooting, and multi-layered validation is essential for generating reliable, biologically relevant data. The future of PPI analysis lies in the intelligent integration of established experimental methods with powerful new computational approaches, including deep learning models like graph neural networks and transformers. This synergy will enable researchers to navigate the complexity of signaling networks with unprecedented precision, accelerating the discovery of PPI-targeted therapeutics for cancer, inflammatory diseases, and beyond. As these technologies mature, they promise to transform our ability to decipher the dynamic interactomes that govern cellular fate and function.
References
- 1. Recent advances in deep learning for protein-protein interaction [https://pmc.ncbi.nlm.nih.gov/articles/PMC12168265/]
- 2. New insights into protein–protein interaction modulators in ... [https://www.nature.com/articles/s41392-024-02036-3]
- 3. A comprehensive dataset of protein-protein interactions ... [https://www.nature.com/articles/s41597-024-03233-z]
- 4. Revealing protein-protein interactions at the transcriptome ... [https://www.sciencedirect.com/science/article/pii/S1097276521005748]
- 5. Recent Advances and Application of Machine Learning for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12641681/]
- 6. Synthetic Phosphorylation Networks with Fluorescence and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12186673/]
- 7. Update on Structure and Function of SH2 Domains [https://www.mdpi.com/1422-0067/26/18/9060]
- 8. Understanding domain swapping in the c-Src SH3 domain ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12400191/]
- 9. CoDIAC: A comprehensive approach for interaction ... [https://pubmed.ncbi.nlm.nih.gov/41252491/]
- 10. The STRING database in 2025: protein networks with ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11701646/]
- 11. Solutions | Reaction Biology Protein Protein Interaction Assay [https://www.reactionbiology.com/resources/reading-room/blog/protein-protein-interaction-assays-illuminating-the-science-behind-linklight/]
- 12. The role of dynamic enzyme assemblies and substrate ... [https://www.nature.com/articles/s41467-018-04543-8]
- 13. Computational remodeling of an enzyme conformational ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10539519/]
- 14. Assay Guidance Manual - NCBI Bookshelf - NIH [https://www.ncbi.nlm.nih.gov/books/NBK53196/]
- 15. Microenvironmental effects can masquerade as substrate ... [https://www.sciencedirect.com/science/article/abs/pii/S0958166921001609]
- 16. – Turku Bioscience Centre Protein Interaction Assays [https://bioscience.fi/screening/services/protein-interaction-assays/]
- 17. Protein-Protein Interaction Detection: Methods and Analysis [https://pmc.ncbi.nlm.nih.gov/articles/PMC3947875/]
- 18. Protein Protein Interaction Assay Solutions [https://www.reactionbiology.com/resources/reading-room/blog/protein-protein-interaction-assays-illuminating-the-science-behind-linklight]
- 19. Pathway Analysis Incorporating Protein-Protein Interaction ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0162910]
- 20. Pathway Detection from Protein Interaction Networks and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3346698/]
- 21. The value of coimmunoprecipitation (Co-IP) assays in drug ... [https://pubmed.ncbi.nlm.nih.gov/40289752/]
- 22. Co-IP & Pull-Down for Protein–Protein Interaction Detection [https://www.creative-proteomics.com/resource/protein-protein-interaction-identification-method-co-ip-and-pull-down.htm]
- 23. Immunoprecipitation and co-immunoprecipitation protocol [https://www.abcam.com/en-us/technical-resources/protocols/immunoprecipitation]
- 24. Co-immunoprecipitation (Co-IP): The Complete Guide [https://www.antibodies.com/applications/co-immunoprecipitation]
- 25. Co-Immunoprecipitation (Co-IP) and Pull-Down [https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-assays-analysis/protein-interaction-analysis/co-immunoprecipitation-co-ip-pull-down.html]
- 26. Co-immunoprecipitation and semi-quantitative ... [https://www.sciencedirect.com/science/article/pii/S2666166721003518]
- 27. Integrated Membrane Yeast Two-Hybrid System for the ... [https://en.bio-protocol.org/en/bpdetail?id=5418&type=0]
- 28. Principle and Protocol of Yeast Two Hybrid System [https://www.creativebiomart.net/resource/principle-protocol-principle-and-protocol-of-yeast-two-hybrid-system-445.htm]
- 29. A Split-Ubiquitin Based Strategy Selecting for Protein ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5015938/]
- 30. Next-generation yeast-two-hybrid analysis with Y2H-SCORES ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8046355/]
- 31. Advantages and Disadvantages of Label Transfer in ... [https://www.mtoz-biolabs.com/advantages-and-disadvantages-of-label-transfer-in-protein-interaction.html]
- 32. Label Transfer Protein Interaction Analysis [https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/label-transfer-protein-interaction-analysis.html]
- 33. Protein Dynamics in Solution by Quantitative Crosslinking ... [https://www.sciencedirect.com/science/article/pii/S0968000418301865]
- 34. Rapid Mapping of Protein Interactions Using Tag‐Transfer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6348423/]
- 35. Quantitative cross-linking/mass spectrometry reveals subtle ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5140025/]
- 36. Unraveling cooperative and competitive interactions within ... [https://www.nature.com/articles/s41598-025-19264-4]
- 37. Biosensing applications of surface plasmon resonance- ... [https://www.sciencedirect.com/science/article/abs/pii/S0925400510006751]
- 38. Biosensor-Surface Plasmon Resonance: Label Free ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6774244/]
- 39. Protein-peptide Interaction by Surface Plasmon Resonance [https://bio-protocol.org/en/bpdetail?id=321&type=0]
- 40. Förster Resonance Energy Transfer [https://pmc.ncbi.nlm.nih.gov/articles/PMC4151567/]
- 41. Förster resonance energy transfer [https://en.wikipedia.org/wiki/F%C3%B6rster_resonance_energy_transfer]
- 42. Biolayer interferometry and its applications in drug ... [https://www.sciencedirect.com/science/article/pii/S0165993624002243]
- 43. Measuring Interactions Between Proteins and Small ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11335060/]
- 44. Protein analysis by dynamic light scattering [https://pubmed.ncbi.nlm.nih.gov/23166025/]
- 45. LinkLight™ Assay Technology [https://bioinvenu.com/?page_id=86]
- 46. LinkLight Protein Interaction Analysis Services [https://www.reactionbiology.com/services/target-specific-assays/gpcr-assays/linklight/]
- 47. Bio-Techne Announces Early Access Program for Next- ... [https://acdbio.com/bio-techne-announces-early-access-program-next-generation-spatial-protein-proximity-detection]
- 48. PLM-interact: extending protein language models to predict ... [https://www.nature.com/articles/s41467-025-64512-w]
- 49. PLIP 2025: introducing protein-protein interactions to the ... [https://pubmed.ncbi.nlm.nih.gov/40347107/]
- 50. Pathway Analysis: State of the Art - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC4681784/]
- 51. Integrative analysis of signaling and metabolic pathways ... [https://www.nature.com/articles/s41598-025-97623-x]
- 52. Ingenuity pathway analysis of α-synuclein predicts ... [https://www.frontiersin.org/journals/molecular-neuroscience/articles/10.3389/fnmol.2022.1029682/full]
- 53. Advances in Protein-Protein Interaction Prediction: A Deep ... [https://www.frontiersin.org/journals/bioinformatics/articles/10.3389/fbinf.2025.1710937/full]
- 54. A graph neural network model for inferring interindividual ... [https://www.nature.com/articles/s41598-025-23320-4]
- 55. DCMF-PPI: a protein-protein interaction predictor based on ... [https://bmcbioinformatics.biomedcentral.com/articles/10.1186/s12859-025-06272-4]
- 56. Transformers in Protein: A Survey [https://arxiv.org/html/2505.20098v2]
- 57. PLM-interact: extending protein language models to predict ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12559430/]
- 58. Where Have All the Interactions Gone? Estimating the ... [https://journals.plos.org/ploscompbiol/article?id=10.1371/journal.pcbi.0030214]
- 59. Immunoprecipitation Experimental Design Tips [https://www.cellsignal.com/applications/immunoprecipitation/ip-experimental-design-tips?srsltid=AfmBOorsWmPLO2oVpxvHCfGbdW2XoCqQpnYdESKrzO6jXfKsBuQhLz5x]
- 60. Co-immunoprecipitation: Principles and applications [https://www.abcam.com/en-us/knowledge-center/proteins-and-protein-analysis/co-immunoprecipitation]
- 61. Approaches for assessing and discovering protein ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3834224/]
- 62. Two protein/protein interaction assays in one go - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6053639/]
- 63. Protein interactions in human pathogens revealed through ... [https://www.nature.com/articles/s41564-024-01791-x]
- 64. Bacterial two-hybrid systems evolved: innovations for protein ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12369361/]
- 65. In vivo crosslinking and effective 2D enrichment for ... [https://www.nature.com/articles/s42004-025-01644-6]
- 66. Breaking barriers in crosslinking mass spectrometry with ... [https://www.nature.com/articles/s41467-025-64844-7]
- 67. A comparison and optimization of yeast two-hybrid systems [https://pmc.ncbi.nlm.nih.gov/articles/PMC5737776/]
- 68. A Genetic Strategy to Eliminate Self-Activator Baits Prior ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC544402/]
- 69. Yeast two-hybrid screening for proteins that interact with ... [https://www.nature.com/articles/s41598-019-52030-x]
- 70. An improved yeast two-hybrid approach for detection of ... [https://link.springer.com/article/10.1007/s11515-006-0008-9]
- 71. DoMY-Seq: A yeast two-hybrid–based technique for ... [https://www.sciencedirect.com/science/article/pii/S0021925820000095]
- 72. Recent progress and future challenges in structure-based ... [https://www.sciencedirect.com/science/article/pii/S1525001625002771]
- 73. How thoughtful experimental design can empower biologists ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12328601/]
- 74. Enhancing Biotech Research Through Design of Experiments [https://www.mabion.eu/science-hub/articles/enhancing-biotechnological-research-with-design-of-experiments/]
- 75. Narrowing the Gap Between In Vitro and In Vivo Genetic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6964707/]
- 76. An integrated approach to identify environmental ... [https://www.sciencedirect.com/science/article/pii/S0002929721003359]
- 77. Establishing an In Vivo Assay System to Identify Components ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0101661]
- 78. In Vivo Assay Guidelines - Assay Guidance Manual - NCBI [https://www.ncbi.nlm.nih.gov/books/NBK92013/]
- 79. Presenting Quantitative Data Graphically [https://courses.lumenlearning.com/waymakermath4libarts/chapter/presenting-quantitative-data-graphically/]
- 80. Presentation of Quantitative Data [https://ihatepsm.com/blog/presentation-quantitative-data]
- 81. The Assay Guidance Manual: A Guide for In Vitro and ... [https://www.promega.com/resources/pubhub/tpub_191-the-assay-guidance-manual/]
- 82. Protocol for establishing a protein-protein interaction ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9304681/]
- 83. Protocol for analyzing protein-protein interactions by split ... [https://www.sciencedirect.com/science/article/pii/S2666166724004933]
- 84. Computational Approaches for Pathway‐Centric Analysis of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12645814/]
- 85. Overview of Protein–Protein Interaction Analysis [https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-protein-protein-interaction-analysis.html]
- 86. Signal transduction at the single-cell level: Approaches to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5023475/]
- 87. Inhibition of Protein-Protein Interactions: Cell-Based Assays [https://www.ncbi.nlm.nih.gov/books/NBK464632/]
- 88. Signaling Pathway Analysis [https://www.creative-diagnostics.com/protein-array/signaling-pathway-analysis.html]
- 89. A human protein-protein interaction network [https://thebiogrid.org/77183/publication/a-human-protein-protein-interaction-network-a-resource-for-annotating-the-proteome.html]
- 90. The STRING database in 2025: protein networks with ... [https://pubmed.ncbi.nlm.nih.gov/39558183/]
- 91. Info - STRING functional protein association networks [https://string-db.org/cgi/info]
- 92. BioGRID | Database of Protein, Chemical, and Genetic ... [http://thebiogrid.org/]
- 93. Synthetic Protein Interaction Project [https://thebiogrid.org/project/12/synthetic-protein-interaction-project.html]
- 94. DIPS-Plus: The enhanced database of interacting protein ... [https://www.nature.com/articles/s41597-023-02409-3]
- 95. Protocols [https://www.cellsignal.com/learn-and-support/protocols?srsltid=AfmBOoowP4aSdQVGZNC60tZCJMPEhGNvyqO8mNemKb3MseCBBl51Ed2K]
- 96. Fundamentals of protein interaction network mapping - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4704491/]
- 97. Integrating in-silico and experimental validation ... [https://www.nature.com/articles/s41598-025-19931-6]
- 98. Maximizing binary interactome mapping with a minimal ... [https://www.nature.com/articles/s41467-019-11809-2]
- 99. Integrated web visualizations for protein-protein interaction ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4466863/]
- 100. Protein-Protein Interactions [https://cytoscape.org/cytoscape-tutorials/presentations/ppi-tools1-2017-mpi.html]
- 101. PIN - User guide - SFARI Gene [https://gene.sfari.org/user-guide/pin/]
- 102. Essential guidelines for computational method benchmarking [https://pmc.ncbi.nlm.nih.gov/articles/PMC6584985/]
- 103. Activity prediction benchmarking [https://www.desertsci.com/2025/06/27/activity-prediction-benchmarking/]
- 104. Comprehensive benchmarking of computational tools for ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-024-00931-z]