PCR vs. Culture for Mycoplasma Detection: A Modern Sensitivity Comparison for Researchers
This article provides a comprehensive comparison of Polymerase Chain Reaction (PCR) and traditional culture methods for detecting mycoplasma contamination, a critical concern in biopharmaceutical manufacturing and cell therapy.
PCR vs. Culture for Mycoplasma Detection: A Modern Sensitivity Comparison for Researchers
Abstract
This article provides a comprehensive comparison of Polymerase Chain Reaction (PCR) and traditional culture methods for detecting mycoplasma contamination, a critical concern in biopharmaceutical manufacturing and cell therapy. Tailored for researchers, scientists, and drug development professionals, it explores the foundational principles of each method, delves into their practical applications and protocols, addresses common troubleshooting and optimization challenges, and presents a rigorous validation and comparative analysis based on current scientific literature. The synthesis of this information aims to guide professionals in selecting the most appropriate, efficient, and sensitive detection strategy to ensure product safety and regulatory compliance.
Mycoplasma Detection Fundamentals: Why Sensitivity is Non-Negotiable in Biopharma
The Critical Impact of Mycoplasma Contamination on Cell Culture and Biological Products
Mycoplasma contamination represents one of the most significant yet insidious challenges in cell culture laboratories and biopharmaceutical manufacturing. These minute bacteria, lacking cell walls, persistently infect cell lines, affecting up to 35% of cultures worldwide, with some studies reporting contamination rates as high as 87% in certain settings [1] [2]. The critical impact of this contamination extends beyond compromised research data to include substantial economic losses and potential risks to patient safety when biological products are involved [2]. This guide provides a comprehensive comparison of detection methodologies, focusing on the sensitivity and applicability of PCR versus traditional culture methods, to support scientists, researchers, and drug development professionals in implementing robust contamination control strategies.
Understanding Mycoplasma Contamination
What Are Mycoplasmas?
Mycoplasmas are the smallest self-replicating organisms known, measuring only 0.1-0.3 μm in diameter [2] [3]. As members of the class Mollicutes, they are characterized by the complete absence of a cell wall, which contributes to their resistance to common antibiotics like penicillin and streptomycin and allows them to pass through standard 0.2 μm sterilization filters [1] [4]. Their minimal genome (600-2,200 kilobases) limits their metabolic capabilities, making them dependent on host organisms for essential nutrients [5] [4].
Prevalence and Common Contaminating Species
Despite their biological simplicity, mycoplasmas present a formidable challenge to cell culture integrity. Current estimates suggest that 15-35% of continuous cell lines worldwide suffer from mycoplasma contamination, with mammalian and avian cell lines showing particularly high susceptibility [1] [2]. More than 200 Mycoplasma species have been identified, but a select few account for the majority of contamination events:
Table 1: Major Mycoplasma Species Contaminating Cell Cultures
| Species | Primary Source | Approximate Frequency | Key Characteristics |
|---|---|---|---|
| M. orale | Human oral cavity | 20-40% of infections | Most common human-derived contaminant [2] |
| M. arginini | Bovine serum | 20-30% of infections | Frequently introduced via fetal bovine serum [6] [1] |
| M. hyorhinis | Swine | 10-40% of infections | Often introduced via trypsin of porcine origin [6] [1] |
| M. fermentans | Human | 10-20% of infections | Human-derived contaminant [6] |
| M. hominis | Human | 10-20% of infections | Human-derived contaminant [6] [2] |
| A. laidlawii | Bovine serum | 5-20% of infections | Common bovine-derived contaminant [6] [1] |
Consequences of Contamination
The effects of mycoplasma contamination on cell cultures and biological products are profound and multifaceted. These microorganisms compete with host cells for essential nutrients, including amino acids, nucleotides, sugars, and lipids, leading to altered cellular metabolism, reduced proliferation rates, and morphological changes [5] [2]. At the molecular level, mycoplasma contamination can cause chromosomal aberrations, affect gene expression profiles, modify plasma membrane antigens, and disrupt various cellular functions, ultimately compromising the validity of experimental data [2] [7].
In biopharmaceutical manufacturing, the consequences extend to economic losses and potential patient risks. Mycoplasma contamination can necessitate batch rejection, result in production shutdowns, and require exhaustive decontamination procedures. Most alarmingly, undetected mycoplasmas in biological products can cause sepsis and disseminated infections in patients, which may prove fatal [2].
Detection Methodologies: A Comparative Analysis
Several methodologies have been developed for mycoplasma detection, each with distinct principles, advantages, and limitations. The choice of method depends on various factors, including required sensitivity, turnaround time, equipment availability, and regulatory considerations.
Table 2: Mycoplasma Detection Methods Comparison
| Method | Principle | Time to Result | Advantages | Limitations |
|---|---|---|---|---|
| Microbial Culture | Growth on specialized agar and broth media | 28 days on average [2] | Regulatory gold standard; can detect viable organisms | Time-consuming; cannot detect non-cultivable species [4] |
| DNA Staining (DAPI/Hoechst) | Fluorescent staining of extranuclear DNA | 1-2 days (including 5-7 day indicator cell culture) [7] | Direct visualization; relatively simple procedure | Lower sensitivity; subjective interpretation [6] [8] |
| Enzymatic (MycoAlert) | Detection of microbial ATP generation | <30 minutes [9] | Rapid; simple protocol; quantitative results | Lower sensitivity than PCR; may give false negatives [9] |
| PCR | Amplification of mycoplasma DNA sequences | 2-3 hours [3] | High sensitivity and specificity; rapid results | Requires DNA purification in standard protocols [9] |
| qPCR | Real-time amplification and detection | 1-2 hours [9] | Quantitative; high sensitivity; no electrophoresis needed | Requires specialized equipment; potential inhibition issues [9] |
Experimental Evidence: Sensitivity Comparisons
Multiple studies have directly compared the sensitivity of different mycoplasma detection methods. A comprehensive evaluation of 30 cell lines from the National Cell Bank of Iran revealed striking differences in detection capabilities:
Table 3: Detection Sensitivity Across Methods - 30 Cell Line Study [6]
| Detection Method | Contamination Detection Rate | Sensitivity | Specificity | Accuracy |
|---|---|---|---|---|
| Microbial Culture | 33.33% (10/30) | Not specified | Not specified | Not specified |
| DNA Staining (DAPI) | 46.66% (14/30) | Not specified | Not specified | Not specified |
| Enzymatic (MycoAlert) | 53.33% (16/30) | Not specified | Not specified | Not specified |
| Conventional PCR | 56.66% (17/30) | 94.44% | 100% | 96.77% |
| Real-time PCR | 60% (18/30) | 100% | 100% | 100% |
A separate study of 40 cell lines confirmed these findings, showing mycoplasma contamination rates of 57.5% with molecular methods (PCR), 52.5% with enzymatic methods, and only 40% with microbial culture methods [4]. This consistent pattern across studies demonstrates the superior detection capability of nucleic acid amplification techniques.
PCR vs. Culture Methods: Detailed Experimental Protocols
Microbial Culture Method Protocol
The microbial culture method, recognized as the gold standard in pharmacopeias including European Pharmacopoeia Chapter 2.6.7 and United States Pharmacopoeia Chapter 63, involves the following steps [2] [4]:
Sample Preparation: Cell culture supernatants are collected after centrifuging at 200 × g for 5 minutes to remove cell debris. Samples should be cultured in antibiotic-free medium for at least 4-7 days before testing to enhance detection sensitivity [6] [4].
Inoculation: Aseptically transfer 0.1-0.2 mL of sample into:
- Liquid broth media (e.g., mycoplasma broth base)
- Solid agar plates (specialized mycoplasma agar) Incubate at 36°C ± 1°C under aerobic conditions with 5-10% CO₂ [4].
Subculture: After 3-7 days of incubation, subculture 0.1-0.2 mL from the broth medium onto fresh solid agar plates. Repeat this process weekly for 3-4 weeks to enhance the probability of detecting slow-growing strains [2].
Colony Examination: Periodically examine agar plates for characteristic "fried egg" colony morphology using microscopic examination at 50-100× magnification. The typical incubation period is 28 days before declaring a sample negative [2].
Quality Control: Include known positive and negative controls with each batch. Test all media components for sterility and growth promotion using reference mycoplasma strains [4].
PCR/qPCR Method Protocol
Molecular methods for mycoplasma detection typically target the highly conserved 16S ribosomal RNA gene, enabling broad species detection [6] [7]. The following protocol represents an optimized approach:
Sample Collection and Preparation:
- Collect 100-200 μL of cell culture supernatant (avoid cellular fraction)
- For direct qPCR (without DNA purification): Use 6 μL of supernatant directly in the reaction mix [9]
- For conventional qPCR: Extract DNA using commercial kits (e.g., QIAamp DNA Mini Kit) according to manufacturer's instructions [9]
Primer and Probe Design:
Reaction Setup:
- Prepare master mix according to kit specifications (e.g., PhoenixDx Mycoplasma Mix)
- Add template DNA (2-10 μL for purified DNA; 6 μL for direct qPCR)
- Include internal controls to monitor for PCR inhibition
- Include non-template controls to detect contamination [9]
Amplification Parameters (optimized for direct qPCR) [9]:
- Initial denaturation: 95°C for 2 minutes
- 40 cycles of:
- Denaturation: 95°C for 5 seconds
- Annealing/extension: 52°C for 20 seconds
- Total run time: ~65 minutes
Result Interpretation:
- For qPCR: Analyze amplification curves and Ct values
- Validate with melting curve analysis for SYBR Green-based assays
- Consider samples with Ct values <35 as potentially positive
- Confirm positive results by sequencing when necessary [7]
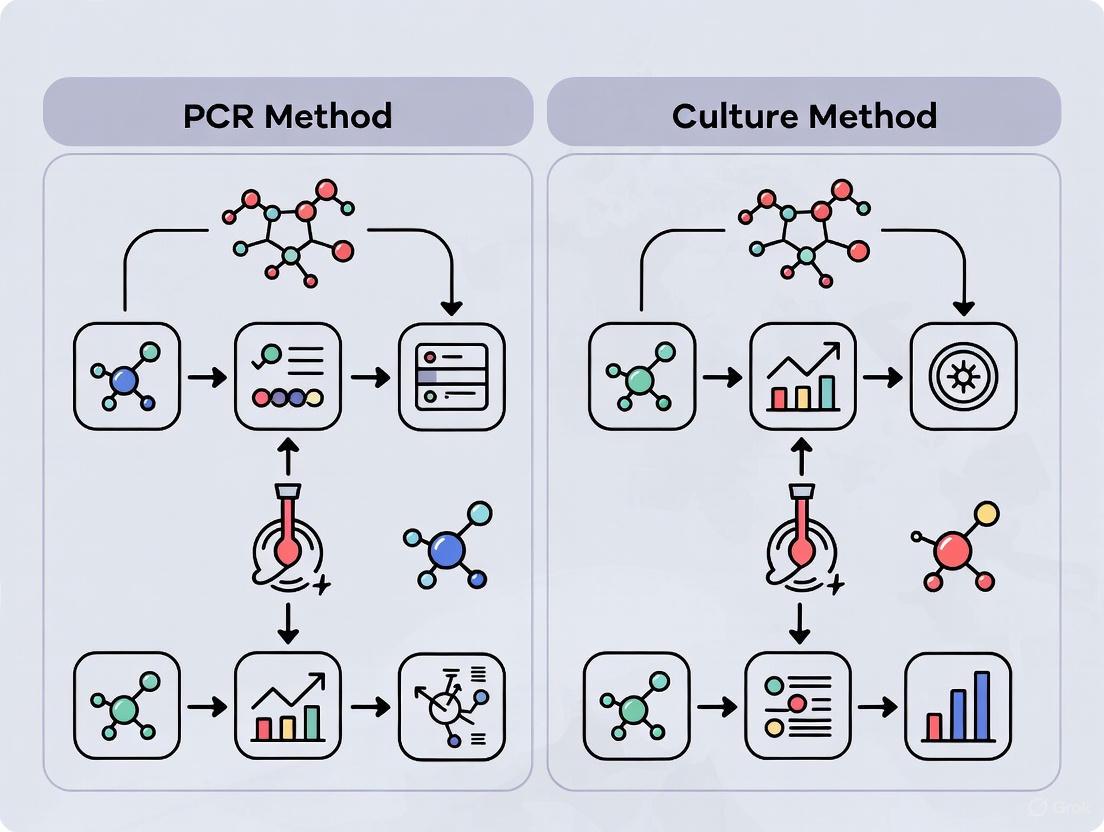
Figure 1: Workflow comparison between PCR-based and culture-based detection methods
Performance Data Analysis
Sensitivity and Detection Limits
The critical advantage of PCR-based methods lies in their enhanced sensitivity and lower detection limits compared to traditional techniques. Research demonstrates that well-optimized qPCR assays can achieve detection limits of 10 colony-forming units (CFU)/mL or lower, meeting the requirements set by pharmacopeial standards [2] [3]. Some studies report detection capabilities as low as 0.1 CFU/mL for certain mycoplasma strains, significantly surpassing the sensitivity of culture methods [2].
A direct comparison study demonstrated that optimized direct qPCR (without DNA purification) showed higher sensitivity than regular qPCR with purified DNA templates, with Ct values of 23.42 for direct qPCR versus 23.49 for regular qPCR when comparing equivalent sample volumes [9]. This enhanced sensitivity enables earlier detection of contamination, potentially preventing widespread laboratory contamination.
Detection of Non-Cultivable Species
Approximately 5-15% of mycoplasma contaminants belong to species that are difficult or impossible to culture using standard media [4]. These include certain strains of M. hyorhinis and Ureaplasma species, which may go undetected by culture methods but are readily identified by PCR-based approaches [4] [9]. The Ureaplasma species, which rely on urea hydrolysis rather than standard ATP generation, are particularly challenging for biochemical detection methods but can be detected with universal 16S rRNA-targeting PCR assays [9].
Time Efficiency and Workflow Integration
The dramatically shorter turnaround time of PCR-based methods represents a significant operational advantage. While culture methods require up to 28 days for definitive results, PCR and qPCR assays can provide reliable data within hours [2] [3] [9]. This difference is particularly crucial in biopharmaceutical manufacturing, where rapid release testing is essential for maintaining production schedules and minimizing inventory costs.
Figure 2: Time efficiency comparison across detection methodologies
Research Reagent Solutions
Implementation of reliable mycoplasma detection requires specific reagents and kits designed for optimal performance. The following table details essential materials and their applications in mycoplasma detection workflows:
Table 4: Essential Research Reagents for Mycoplasma Detection
| Reagent/Kits | Function/Application | Key Features | Regulatory Compliance |
|---|---|---|---|
| MycoAlert/MycoAlert Plus (Lonza) | Biochemical detection of mycoplasma contamination | Measures microbial ATP generation; results in <30 minutes; includes positive controls | Validated according to EP and USP guidelines [1] [4] |
| Mycoplasma Real-time PCR Detection Kits (Various manufacturers) | Molecular detection via qPCR | Detects 180+ mycoplasma species; includes internal controls; LOD: ≤10 CFU/mL | Compliant with EP 2.6.7, JP G3, and USP 63 [3] |
| PhoenixDx Mycoplasma Mix (Procomcure Biotech) | qPCR-based detection | Probe-based technology; includes internal control; optimized for direct PCR | Research use only [9] |
| Microsart RESEARCH Mycoplasma (Sartorius) | qPCR-based detection | Probe-based technology; includes internal control | Validated according to pharmacopeial guidelines [9] |
| Mycoplasma Elimination Reagents (e.g., Bio-Rad, Yeasen) | Treatment of contaminated cultures | Antibiotic mixtures targeting mycoplasma; minimal cytotoxicity to host cells | Research use only [3] [9] |
Regulatory Considerations and Industry Applications
Regulatory agencies worldwide, including the FDA and EMA, mandate mycoplasma testing for cell banks, viral seeds, unprocessed harvest fluids, and sometimes final products [2] [3]. The ICH Q5D guideline specifically addresses quality requirements for biotechnological/biological products, including mycoplasma testing [2].
While traditional culture methods remain the official compendial method in many pharmacopeias, PCR-based methods are increasingly accepted for in-process testing and rapid lot release. The FDA acknowledges that "PCR-based assays may be used to detect mycoplasma, provided that such an assay can be shown to be comparable to the agar and broth media procedure" [2]. This regulatory evolution reflects the growing recognition of PCR's superior sensitivity, specificity, and speed in detecting these problematic contaminants.
For cell and gene therapy products with short shelf lives, where the 28-day incubation period required for culture methods is impractical, qPCR-based methods are particularly valuable and often necessary [2].
The critical impact of mycoplasma contamination on cell culture systems and biological products necessitates robust, sensitive detection methods. While microbial culture remains the regulatory gold standard, PCR-based methods—particularly qPCR—demonstrate superior sensitivity (100% vs. 33-40% in comparative studies), faster turnaround times (hours versus weeks), and broader detection capabilities for non-cultivable species.
The experimental data presented in this guide consistently shows that molecular methods detect 15-25% more contaminated samples than traditional culture methods. This enhanced detection capability, combined with significantly reduced processing time, makes PCR-based approaches invaluable tools for maintaining cell culture integrity, ensuring research reproducibility, and safeguarding biopharmaceutical products.
For laboratories handling valuable or irreplaceable cell lines, implementing a complementary testing strategy that combines the specificity of culture methods with the sensitivity and speed of qPCR provides the most comprehensive protection against the detrimental effects of mycoplasma contamination.
In the field of clinical microbiology and biopharmaceutical product safety, the accurate detection of microbial contaminants like mycoplasma is paramount. For decades, culture-based methods have been regarded as the historical "gold standard" for this purpose, prized for their ability to confirm viable organisms. This guide provides a systematic comparison between these traditional techniques and modern molecular methods, such as Polymerase Chain Reaction (PCR), framing the discussion within the context of mycoplasma detection sensitivity research. The evaluation is grounded in current experimental data, detailing the protocols, performance metrics, and specific applications that define the current landscape of microbial testing. For researchers, scientists, and drug development professionals, understanding this comparative landscape is critical for selecting the appropriate method to ensure product safety, guide timely therapeutic decisions, and uphold rigorous scientific standards.
The core distinction between culture-based and PCR-based methods lies in their fundamental principles: one relies on microbial growth, and the other on nucleic acid amplification.
Culture-based methods depend on the ability of a viable mycoplasma cell to proliferate in specialized enriched broth and agar media over a defined period. This process confirms cellular viability and allows for subsequent analysis of the isolated organism. The United States Pharmacopeia (USP) method <63> is a key compendial method that requires a minimum 28-day incubation period to accommodate the slow growth of some mycoplasma species [10]. This lengthy process is a significant bottleneck in time-sensitive applications like cell and gene therapy release testing.
In contrast, PCR-based methods function by amplifying specific DNA sequences unique to mycoplasma species. This molecular technique detects the presence of mycoplasma genetic material, providing results in a fraction of the time required by culture. However, a primary limitation of standard PCR is its inability to distinguish between DNA from live and dead cells, which can lead to false positives in certain scenarios [8] [11]. To address the limitations of both approaches, hybrid approaches have emerged. The hybrid PCR-based method incorporates a brief enrichment culture step (e.g., 3 days) in a mycoplasma-supportive broth to amplify viable organisms, followed by PCR detection. This strategy minimizes matrix interference from the sample, improves sensitivity, and maintains the ability to detect viable contaminants, all while delivering results in ≤ 8 days [10]. Another innovative approach is culture-based viability PCR, which uses species-specific quantitative PCR (qPCR) both before and after a sample incubation in growth media. A decrease in the cycle threshold (CT) value after incubation indicates the proliferation of viable organisms, combining the sensitivity of qPCR with a confirmation of viability [11].
Table 1: Core Principles of Mycoplasma Detection Methods
| Method Type | Fundamental Principle | Key Outcome | Time to Result |
|---|---|---|---|
| Culture-Based | Growth of viable organisms in enriched media [10]. | Confirmation of viable, cultivable cells. | ≥28 days [10]. |
| Standard PCR | Amplification of species-specific DNA sequences [8]. | Detection of mycoplasma DNA (live and dead). | A few hours [8]. |
| Hybrid PCR | Short enrichment culture followed by PCR amplification [10]. | Detection of viable cells with reduced matrix interference. | ≤ 8 days [10]. |
| Viability PCR | qPCR measurement before and after incubation in media [11]. | Confirmation of viable cells via DNA increase from proliferation. | Several days [11]. |
Experimental Data and Performance Comparison
Recent studies directly comparing these methods provide quantitative data on their diagnostic performance. A critical factor influencing sensitivity is the sample type. A 2025 study on detecting Mycoplasma pneumoniae (MP) in children with respiratory infections demonstrated that the choice of sample source significantly impacts results. With real-time PCR (RT-PCR) of oropharyngeal swabs as a reference, the sensitivity of a commercially available PCR test on oropharyngeal samples was 96.2%, significantly superior to the 74.9% sensitivity of a multiplex PCR test on nasopharyngeal samples [12]. This finding underscores that sample type can be as important as the chosen technology.
The limitations of culture-based methods become particularly evident in complex testing environments. For instance, in a study on environmental monitoring in healthcare settings, culture-based viability PCR outperformed traditional culture. It detected viable S. aureus in 73% of samples that had detectable DNA, whereas traditional culture after broth enrichment detected it in only 19% of those same samples [11]. This demonstrates that even when culture is enhanced with enrichment steps, it may fail to detect viable pathogens that more sensitive molecular methods can identify.
In the context of high-value products like CAR T-cells, the hybrid PCR method has been validated as a suitable alternative to the USP <63> culture method. A case study demonstrated that this method could consistently recover and detect various mycoplasma species, including the traditionally non-cultivable M. hyorhinis, in high cell-density products (1-5 × 10^6 cells/mL) and was non-inferior to the USP <63> test [10]. This shows that advanced PCR methods can meet the rigorous standards required for product release testing while offering a significantly faster turnaround.
Table 2: Experimental Performance Comparison in Clinical and Product Testing
| Application Context | Methodology | Key Performance Finding | Reference Standard |
|---|---|---|---|
| Respiratory Infection (M. pneumoniae) | PCR (Oropharyngeal Sample) | Sensitivity: 96.2% (92.3–98.4%) [12]. | RT-PCR on oropharyngeal swabs [12]. |
| Respiratory Infection (M. pneumoniae) | PCR (Nasopharyngeal Sample) | Sensitivity: 74.9% (67.9–81.0%) [12]. | RT-PCR on oropharyngeal swabs [12]. |
| Environmental Monitoring (S. aureus) | Culture-Based Viability PCR | Viable detection: 73% (8/11) of qPCR-positive samples [11]. | Species-specific qPCR and culture [11]. |
| Environmental Monitoring (S. aureus) | Traditional Culture (after enrichment) | Viable detection: 19% (5/26) of total samples [11]. | Culture on TSA agar [11]. |
| CAR T-Cell Product Release | Hybrid PCR (with enrichment) | Non-inferior to USP <63> for mycoplasma detection [10]. | USP <63> culture method [10]. |
Detailed Experimental Protocols
To ensure reproducibility and provide a clear technical understanding, this section outlines the standard protocols for two key methods: the compendial culture-based assay and the emerging hybrid PCR assay.
Compendial Culture-Based Method (USP <63>)
The USP <63> method is a rigorous, prolonged process designed to detect even low levels of viable mycoplasma [10].
- Sample Inoculation: The test article is inoculated into two different types of liquid enrichment broths that support the growth of a wide range of mycoplasma species.
- Enrichment Culture: The inoculated broths are incubated for a minimum of 14 days at 36±1°C. One broth is typically incubated aerobically, and the other under anaerobic conditions to accommodate the growth requirements of different mycoplasma species.
- Subculture and Observation: After 14 days of incubation, a portion of each broth culture is subcultured onto solid agar plates designed to support mycoplasma growth. These agar plates are then incubated for an additional 14 days and examined periodically for the appearance of characteristic mycoplasma colonies, often identified by their distinctive "fried-egg" morphology.
- Result Interpretation: The entire process requires at least 28 days to complete. The presence of mycoplasma colonies on the agar plates at any point confirms a positive result. The method's key advantage is its direct confirmation of viability and cultivability.
Hybrid PCR-Based Detection Method
This protocol, used for testing high cell-density products, combines enrichment with PCR for a faster, yet sensitive, result [10].
- Sample Normalization: The test article, such as a CAR T-cell product, may be normalized to a standardized cell density (e.g., 1 × 10^6 cells/mL) using its own spent media to minimize matrix interference.
- Liquid Enrichment: The normalized sample is introduced into a mycoplasma-supportive broth and incubated for 3 days. This step allows any viable mycoplasma present to proliferate, amplifying the signal and diluting out potential PCR inhibitors from the sample matrix.
- Nucleic Acid Extraction: Following the enrichment period, a small aliquot of the broth culture is taken. DNA is extracted from this aliquot using a commercial kit, such as the QIAamp DNA Mini Kit, to purify the genetic material for analysis.
- PCR Amplification and Detection: The extracted DNA is analyzed using a validated PCR assay, which employs species-specific primers to amplify target mycoplasma DNA. The detection can involve fluorescent probes (like QProbe PCR) to identify the presence of amplified DNA, providing a positive or negative result for mycoplasma contamination.
- Result Interpretation: Results can be obtained in ≤ 8 days total. A positive signal indicates the presence of viable mycoplasma, as the enrichment step would not support the replication of dead organisms.
The Scientist's Toolkit: Essential Research Reagents and Materials
The successful execution of these detection methods relies on a suite of specialized reagents and materials. The following table details key solutions and their functions in the experimental workflows.
Table 3: Essential Research Reagent Solutions for Mycoplasma Detection
| Item Name | Function / Application | Example Use Case |
|---|---|---|
| Universal Transport Medium (UTM) | Preserves sample integrity during collection and transport [12]. | Collecting nasopharyngeal swabs for respiratory pathogen PCR panels [12]. |
| Mycoplasma Supportive Broth | Enriched liquid medium that promotes the growth of viable mycoplasma [10]. | Liquid enrichment phase in both compendial culture and hybrid PCR methods [10]. |
| QIAamp DNA Mini Kit | Silica-membrane-based system for purification of high-quality DNA from complex samples [12]. | Extracting mycoplasma DNA from clinical swabs or enriched culture broth for PCR [12]. |
| Selective Agar Plates | Solid growth media containing agents to inhibit competing flora and support mycoplasma colony formation [11]. | Subculturing and isolating specific pathogens like C. difficile or observing mycoplasma colonies [11]. |
| Species-Specific Primers & Probes | Short nucleic acid sequences designed to bind and amplify unique genetic targets of a pathogen [12]. | Enabling specific detection of M. pneumoniae CARDS toxin gene or other species in PCR assays [12]. |
| PowerUp SYBR Green Master Mix | A ready-to-use PCR reagent containing a fluorescent dye that binds to double-stranded DNA [11]. | Performing quantitative PCR (qPCR) for viability testing or pathogen quantification [11]. |
| Nucleospin Food Kit | Optimized for extracting genomic DNA from complex, difficult food matrices [13]. | Purifying DNA from food samples (e.g., lettuce, chicken) for metagenomic analysis of pathogens [13]. |
The data and protocols presented clearly illustrate that the designation of a "gold standard" is highly context-dependent. Culture-based methods remain indispensable for their ability to provide unequivocal evidence of viable, cultivable organisms, which is why they are enshrined in compendial standards like USP <63> [10]. Their major limitations are the extended time-to-result (≥28 days) and lower sensitivity compared to molecular methods, as they can miss low-level or non-cultivable contaminants [11].
PCR-based techniques offer a powerful alternative with superior speed and sensitivity, capable of detecting a wider range of mycoplasma species, including those that are difficult to culture [8]. Their primary limitation is the inability to distinguish between live and dead cells based on DNA detection alone [11]. However, the development of hybrid methods like culture-based viability PCR and enrichment PCR effectively bridges this gap. By incorporating a brief culture step, these integrated approaches leverage the strengths of both worlds: the viability confirmation of culture and the speed and sensitivity of PCR [10] [11].
For researchers and drug development professionals, the choice of method should be guided by the specific application. In time-critical scenarios like cell and gene therapy product release, hybrid PCR methods offer a validated, rapid, and sensitive alternative. For environmental surveillance or diagnosing complex infections, viability PCR can provide a more accurate assessment of contamination risk. Ultimately, the evolving toolkit for mycoplasma detection demonstrates that the future lies not in a single "gold standard," but in selecting and validating the right tool—or combination of tools—for the scientific question at hand.
The Rise of Nucleic Acid Amplification Techniques (NATs) as Rapid Alternatives
The following table provides a comparative overview of key Nucleic Acid Amplification Techniques (NAATs), highlighting their core principles, advantages, and limitations relative to traditional culture methods.
| Technique | Principle | Key Advantage over Culture | Primary Limitation | Typical Turnaround Time |
|---|---|---|---|---|
| Polymerase Chain Reaction (PCR) | Thermal cycling for DNA amplification [14] | High sensitivity and specificity [15] | Requires thermal cycler; potential for false positives from non-viable pathogen DNA [15] | Hours to days [15] |
| Digital PCR (dPCR) | End-point PCR with partitioning for absolute quantification [16] | Exceptional sensitivity and absolute quantification without standard curves [16] | Requires specialized droplet system; pre-designed panels may not cover all pathogens [16] | ~4.8 hours [16] |
| Loop-Mediated Isothermal Amplification (LAMP) | Isothermal amplification with strand-displacing polymerase and multiple primers [14] | Rapid, simple, and resistant to inhibitors; suitable for point-of-care use [14] [17] | Complex primer design [18] | 30-60 minutes [14] |
| Nucleic Acid Sequence-Based Amplification (NASBA) | Isothermal, transcription-based amplification for RNA [14] [19] | High analytical sensitivity for RNA; detects viable pathogens [14] [19] | Target length limitation (~100-250 nucleotides) [14] | <2 hours [19] |
| Strand Displacement Amplification (SDA) | Isothermal amplification using a restriction enzyme and strand-displacing polymerase [14] | Can be performed at high temperatures [14] | Inability to efficiently amplify long target sequences [14] | <1 hour [14] |
| Blood Culture (Traditional Method) | Growth and proliferation of microorganisms in enriched media [16] | Gold standard; allows for antibiotic susceptibility testing [16] | Time-consuming; low sensitivity; affected by prior antibiotic use [16] [15] | 24-48 hours to several days [16] [15] |
For decades, culture-based methods have been the cornerstone for detecting pathogens like mycoplasma, providing a gold standard that allows for direct observation of microbial growth. However, these methods are hampered by prolonged incubation times (24-48 hours or more) and significantly reduced sensitivity, especially in patients who have already received antibiotics [16] [15]. The critical need for rapid and accurate diagnosis in clinical management and drug development has catalyzed the rise of Nucleic Acid Amplification Techniques (NAATs). These molecular methods offer a paradigm shift by detecting pathogen-specific genetic material with superior speed and sensitivity, enabling early intervention and supporting antimicrobial stewardship [16]. This guide objectively compares the performance of major NAATs against culture methods, with a specific focus on evidence relevant to mycoplasma detection sensitivity.
Comparative Performance Data: Sensitivity, Specificity, and Speed
Direct Comparison: dPCR vs. Blood Culture
A recent 2025 retrospective study of 149 patients with suspected bloodstream infections provides compelling data on the performance of a modern NAAT compared to culture [16] [20].
Key Experimental Findings:
- Sensitivity: dPCR identified 63 pathogenic strains across 42 positive specimens, while blood culture detected only 6 strains from 6 specimens [16].
- Detection Range: dPCR demonstrated a wide dynamic range, quantifying pathogen concentrations from 25.5 to 439,900 copies/mL [16].
- Turnaround Time: The average detection time for dPCR was 4.8 hours, a significant improvement over the 94.7 hours required for blood culture [16].
- Polymicrobial Infections: dPCR identified 14 cases of polymicrobial infections, including instances of double, triple, quadruple, and even quintuple infections, showcasing its capacity to detect complex cases often missed by culture [16].
Meta-Analysis of Diagnostic Accuracy for a Parasitic Infection
A structured review and meta-analysis evaluated NAATs for diagnosing Schistosoma mansoni infections, providing pooled performance data across multiple studies and techniques [21].
Pooled Diagnostic Accuracy for Human Samples [21]:
- Overall Sensitivity: 89.79% (95% CI: 83.92% - 93.67%)
- Overall Specificity: 87.70% (95% CI: 72.60% - 95.05%)
- Diagnostic Odds Ratio (DOR): 37.73 (95% CI: 21.79 - 65.33)
This analysis also compared different NAATs, finding that LAMP demonstrated the highest sensitivity, followed by PCR-ELISA, conventional PCR, and qPCR, illustrating the variable performance profiles of different amplification techniques [21].
Detailed Experimental Protocols
To ensure experimental reproducibility, this section outlines the standard methodologies for key techniques cited in this guide.
1. Sample Collection and Preparation:
- Collect venous blood into EDTA tubes using standard aseptic procedures.
- Centrifuge at 1,600 × g for 10 minutes to separate plasma.
- Extract plasma DNA using a commercial nucleic acid purification kit and an automated system.
2. dPCR Reaction Setup:
- Use a pre-designed, multi-channel dPCR kit capable of detecting a panel of pathogens.
- Prepare the reaction mix by adding 15 μL of extracted DNA to a master mix containing fluorescent probes and primers.
- Vortex and centrifuge the reaction mixture.
3. Droplet Generation and Amplification:
- Load the reaction solution into a droplet generator to create thousands of individual partitions.
- Transfer the droplet cartridge to a thermal cycler and run the PCR amplification as per the manufacturer's instructions.
4. Detection and Analysis:
- Scan the cartridge using a chip scanner.
- Analyze droplets across six fluorescence channels (FAM, VIC, ROX, CY5, CY5.5, A425) using dedicated software (e.g., Gene PMS).
- Determine the absolute quantification (copies/mL) based on the ratio of positive to negative droplets.
1. Nucleic Acid Extraction:
- Extract total nucleic acids from respiratory specimens (e.g., throat swabs, bronchoalveolar lavage) using a commercial kit (e.g., NucliSens basic kit).
- Store extracted nucleic acids at -80°C if not used immediately.
2. NASBA Amplification with Molecular Beacons:
- Prepare an amplification mixture containing:
- Primers specific to the M. pneumoniae 16S rRNA gene.
- A molecular beacon probe (e.g., Mycobeacon01) with a fluorophore (FAM) and a quencher (Dabsyl).
- Three enzymes: Avian Myeloblastosis Virus Reverse Transcriptase (AMV-RT), RNase H, and T7 RNA polymerase.
- Incubate the reaction isothermally at 41°C for 90-120 minutes in a real-time fluorescence reader.
3. Real-Time Detection:
- Monitor fluorescence in real-time. The molecular beacon hybridizes to the amplified RNA, separating the fluorophore from the quencher and producing a fluorescent signal.
- A positive result is indicated by a fluorescence signal that exceeds a predetermined threshold.
Workflow and Technological Integration
The fundamental difference between traditional, isothermal, and emerging integrated diagnostic workflows is illustrated below.
Comparison of Diagnostic Workflows
The Frontier: Integration with CRISPR-Cas Systems and Microfluidics
The next generation of NAATs focuses on integrating amplification with detection systems in a single, seamless platform.
- One-Pot NAAT-CRISPR Assays: Combining isothermal NAATs (like RPA or LAMP) with CRISPR-Cas systems (e.g., Cas12, Cas13) in a single tube merges high sensitivity with single-nucleotide specificity. The primary challenge is the biochemical incompatibility between amplification and CRISPR components, which is being solved via spatial separation (physical compartments) and temporal separation (controlled, sequential activation) strategies [22].
- Digital Microfluidics (DMF): DMF platforms, particularly those based on electrowetting-on-dielectric (EWOD), manipulate discrete droplets on a planar electrode array. This technology enables the automation of the entire NAAT workflow—from nucleic acid extraction and purification to amplification and detection—in a miniaturized, programmable system with minimal human intervention, making it ideal for point-of-care testing [18].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of NAATs relies on a suite of specialized reagents and equipment. The following table details key solutions required for the experiments cited in this guide.
| Item | Function | Example from Protocols |
|---|---|---|
| dPCR System & Reagents | Partitions samples for absolute nucleic acid quantification. | Pilot Gene Technology droplet dPCR system & kits [16]. |
| NASBA Amplification Module | Provides enzymes and buffer for isothermal RNA amplification. | NucliSens basic kit amplification module (bioMérieux) [19]. |
| Molecular Beacons | Fluorescently-quenched probes for real-time, sequence-specific detection in NASBA. | Mycobeacon01 for M. pneumoniae detection [19]. |
| Nucleic Acid Extraction/Purification Kit | Isolates high-quality DNA/RNA from complex clinical samples. | Auto-Pure10B System kits; NucliSens basic kit extraction module [16] [19]. |
| Strand-Displacing DNA Polymerase | Essential for isothermal methods like LAMP and SDA; displaces DNA strands without heat denaturation. | Bacillus stearothermophilus (Bst) DNA polymerase [14]. |
| CRISPR-Cas Enzymes & crRNA | Provides high-specificity detection and signal transduction for integrated assays. | Cas12a or Cas13a nucleases with custom guide RNA [22]. |
The evidence demonstrates that Nucleic Acid Amplification Techniques have unequivocally emerged as rapid, sensitive, and specific alternatives to traditional culture methods. While culture remains vital for antibiotic susceptibility testing, NAATs like dPCR, LAMP, and NASBA offer transformative advantages in speed and detection capability, which are critical for timely diagnosis and treatment decisions. The ongoing integration of these techniques with CRISPR-based detection and automated microfluidic platforms heralds a new era of point-of-care molecular diagnostics, promising to further redefine the boundaries of sensitivity, specificity, and accessibility in clinical and research laboratories.
Key Mycoplasma Species of Concern in Manufacturing and Clinical Settings
Mycoplasma contamination represents a critical challenge in both biomedical research and pharmaceutical manufacturing. As the smallest self-replicating organisms, mycoplasmas lack cell walls and possess minimal genomes, making them difficult to detect using conventional microbiological methods while rendering them resistant to common antibiotics like penicillin [23]. These parasitic organisms can persist as covert contaminants in cell culture systems, often reaching high densities (10⁵–10⁸ organisms/mL) without causing visible turbidity or cytopathic effects [24]. The presence of mycoplasmas can significantly alter cellular parameters, metabolic functions, and experimental outcomes, compromising research integrity and potentially leading to unsafe biological products [25].
The shift from traditional culture-based detection to modern molecular methods represents a significant advancement in mycoplasma monitoring. This guide provides a comprehensive comparison of polymerase chain reaction (PCR) versus culture methods for mycoplasma detection sensitivity, offering researchers and manufacturing professionals evidence-based insights for selecting appropriate detection strategies. By examining key species of concern, analytical performance metrics, and experimental validation protocols, this article serves as an essential resource for quality control in biological manufacturing and clinical diagnostics.
Key Mycoplasma Species of Concern
Mycoplasma species demonstrate distinct tropisms for specific hosts and anatomical sites, with their distribution and clinical significance varying considerably between manufacturing and clinical contexts [23]. Understanding these species-specific patterns is fundamental to effective contamination control.
High-Prevalence Manufacturing Contaminants
In industrial bioprocessing and cell culture laboratories, a limited number of mycoplasma species account for the overwhelming majority of contamination events. Extensive surveys indicate that approximately 95% of identified cell culture contaminations are caused by just five species: Mycoplasma arginini, M. fermentans, M. hyorhinis, M. orale, and Acholeplasma laidlawii [24]. The sources of these contaminants are typically laboratory personnel (oral mycoplasmas) or contaminated animal sera and reagents used in cell culture systems [25].
Table 1: Key Mycoplasma Species in Manufacturing and Clinical Settings
| Species | Primary Context | Significance/Source | Growth Characteristics |
|---|---|---|---|
| Acholeplasma laidlawii | Manufacturing | Common contaminant of bovine serum; 95% of cell culture contaminants | Less fastidious, can grow in serum-free media |
| Mycoplasma arginini | Manufacturing | Bovine origin; common cell culture contaminant | Fastidious growth requirements |
| Mycoplasma fermentans | Manufacturing & Clinical | Human origin; cell culture contaminant & potential human pathogen | Fastidious growth requirements |
| Mycoplasma hyorhinis | Manufacturing | Porcine origin; common contaminant from trypsin | Fastidious growth requirements |
| Mycoplasma orale | Manufacturing | Human oral flora; common personnel-sourced contaminant | Fastidious growth requirements |
| Mycoplasma pneumoniae | Clinical | Human respiratory pathogen; causes atypical pneumonia | Strict aerobe; requires special media |
| Mycoplasma hominis | Clinical | Genitourinary tract infections, reproductive failure | Fastidious growth requirements |
| Ureaplasma spp. | Clinical | Urogenital infections, infertility, neonatal morbidity | Requires urea; 2-5 days for culture |
| Mycoplasma genitalium | Clinical | Sexually transmitted infections, urethritis | Extremely fastidious; up to 8 weeks for culture |
Clinically Significant Species
In clinical diagnostics, different mycoplasma species emerge as significant pathogens. Mycoplasma pneumoniae is a well-established cause of community-acquired atypical pneumonia, particularly in children and young adults [26]. Genitourinary tract infections and reproductive complications are frequently associated with Ureaplasma species, Mycoplasma hominis, and Mycoplasma genitalium, with these organisms linked to infertility, chorioamnionitis, preterm delivery, and neonatal morbidity [27]. The detection of these clinical pathogens presents unique challenges due to their fastidious growth requirements and the extended incubation periods needed for culture confirmation.
Comparative Detection Methods: PCR vs. Culture
The evolution of mycoplasma detection methodologies has progressed from traditional culture-based approaches to increasingly sophisticated molecular techniques. Understanding the relative capabilities and limitations of these methods is essential for effective laboratory testing strategy.
Culture-Based Detection
Traditional culture methods represent the historical gold standard for mycoplasma detection and are currently mandated by regulatory authorities for product release testing of biologics [28]. These methods require inoculation of specimens onto both solid agar and into liquid enrichment media specifically formulated to support the growth of diverse mycoplasma species, followed by prolonged incubation periods of up to 28 days [28] [25]. The culture approach identifies viable organisms through characteristic "fried-egg" colony formation on solid media or metabolic color changes in broth cultures [29].
Despite its established position in regulatory frameworks, culture-based detection faces significant limitations. The extended time-to-result (often 28 days) renders it incompatible with biological products featuring short shelf lives [28]. Additionally, certain fastidious mycoplasma species exhibit stringent nutritional requirements that may not be met by standard culture media, potentially leading to false-negative results [29]. The method also demands specialized expertise in media preparation and colony recognition, introducing technical variability between laboratories.
PCR-Based Molecular Detection
PCR-based methods have emerged as powerful alternatives to culture, offering revolutionary improvements in detection speed, sensitivity, and practical utility. These nucleic acid amplification techniques target conserved genomic regions, such as the 16S rRNA gene or the 16S-23S rRNA intergenic spacer region, enabling rapid detection—often within 1-3 hours—compared to weeks for cultural methods [29] [30].
The exceptional sensitivity of modern PCR assays allows detection of fewer than 5 mycoplasma genomes per microliter of sample [30], with quantitative PCR (qPCR) demonstrating capability to detect as few as 10⁻¹ copy numbers under optimal conditions [29]. This sensitivity significantly surpasses that of traditional methods. Furthermore, PCR assays can be designed with comprehensive species coverage, simultaneously detecting all mycoplasma species commonly encountered in manufacturing and clinical contexts [30].
Table 2: Performance Comparison of Mycoplasma Detection Methods
| Parameter | Culture Methods | PCR-Based Methods | Experimental Evidence |
|---|---|---|---|
| Time to Result | 21-28 days [28] | 1-3 hours [30] | Multiplex PCR detected pathogens in 75 minutes [26] |
| Analytical Sensitivity | 10-100 CFU/mL for regulatory compliance [28] | <5 genome copies/μL [30]; 10⁻¹ copies for qPCR [29] | LOD of 50 copies for RT-PCR; 1-copy sensitivity observed [24] |
| Species Coverage | Limited to cultivable species; misses fastidious strains | Comprehensive; detects >143 Mycoplasma species [29] | Novel universal primers covered 143 species vs. 5-6 by culture [29] |
| Ability to Detect Multiple Species | Limited due to competition in culture | Excellent; identifies polymicrobial infections [31] | mPCR detected 2-4 pathogens in 19.8% of samples vs. 0.5% by culture [26] |
| Regulatory Status | Gold standard (USP <63>, Ph. Eur. 2.6.7) [28] | Alternative method requiring validation [28] | European Pharmacopoeia recognizes PCR if LOD ≤10 CFU/mL [28] |
Experimental Data and Performance Comparison
Side-by-Side Method Evaluations
Recent multicenter studies provide compelling direct comparisons between PCR and culture methodologies. A comprehensive evaluation of five commercial molecular assays demonstrated that PCR-based methods consistently detected mycoplasma contamination with sensitivity comparable or superior to culture-based approaches [28]. The Biofire Mycoplasma assay exhibited the highest sensitivity, followed closely by MycoSEQ and MycoTOOL kits [28].
Notably, a systematic comparison of PCR-based and bioluminescent assays for mycoplasma detection in cell cultures revealed no statistically significant difference in performance between the two rapid methods [25]. Both techniques successfully identified contaminated samples with high reliability, though researchers noted that eliminating antibiotic use during pre-culture was critical for achieving accurate results with either methodology [25].
Multiplex PCR Applications
The capacity to detect multiple pathogens simultaneously represents a significant advantage of PCR-based methods. In lower respiratory tract infection diagnostics, a multiplex PCR assay identified polymicrobial infections in 19.8% of samples (144/728), whereas culture detected multiple pathogens in only 0.5% (4/728) [26]. This striking discrepancy highlights the limitation of culture methods in recognizing complex infection profiles, potentially leading to incomplete diagnoses and suboptimal therapeutic interventions.
Similar findings emerged from a study of complicated urinary tract infections, where PCR identified polymicrobial infections in 43.52% of cases compared to 31.95% by culture [31]. Patients with undetected polymicrobial infections by culture experienced significantly higher clinical failure rates (33.33%) compared to those with concordant polymicrobial detection (22.22%), underscoring the clinical implications of detection methodology selection [31].
Experimental Protocols and Validation
Validation Framework for PCR Assays
Implementing PCR-based mycoplasma detection requires rigorous validation to ensure analytical performance meets regulatory standards. The European Pharmacopoeia and Japanese Pharmacopoeia specify that molecular methods must demonstrate a limit of detection (LOD) of ≤10 CFU/mL compared to agar and broth culture methods to serve as suitable alternatives [28].
A comprehensive validation approach should include:
- Analytical Sensitivity (LOD) Determination: Testing serial dilutions of quantitated mycoplasma stocks with a minimum of 24 replicates at each dilution to establish the minimal detectable concentration [24].
- Specificity Assessment: Evaluating cross-reactivity with phylogenetically related bacteria, common cell culture contaminants, and host cell DNA to ensure assay specificity [29].
- Robustness Testing: Assessing performance across different sample matrices including cell culture supernatants, serum, and biological products [24].
- Inhibition Studies: Incorporating exogenous controls to identify potential PCR inhibition in complex sample types [24].
Nucleic Acid Extraction and Amplification
Effective DNA extraction represents a critical component of reliable PCR detection. Methods utilizing column-based purification with DNase treatment effectively remove contaminating DNA that could lead to false-positive results [24]. Many commercial kits now offer the option of direct testing of cell culture supernatants without initial DNA extraction, significantly reducing processing time while maintaining sensitivity [30].
Primer design targeting the 16S rRNA gene provides broad species coverage for common contaminants, while targeting the 16S-23S rRNA intergenic spacer region offers enhanced discriminatory power for species differentiation [29]. Advanced detection chemistries, including dual-labeled fluorescent probes, provide additional specificity through sequence-specific hybridization [24].
Figure 1: Standard workflow for PCR-based mycoplasma detection showing significant time reduction compared to 28-day culture methods.
The Scientist's Toolkit: Research Reagent Solutions
Implementing robust mycoplasma detection requires specific reagents and systems designed for optimal performance in either manufacturing or clinical contexts. The following table summarizes essential solutions for establishing reliable testing protocols.
Table 3: Essential Research Reagents for Mycoplasma Detection
| Reagent/Solution | Function | Application Notes | Representative Products |
|---|---|---|---|
| Universal Primer Sets | Amplifies conserved genomic regions (16S rRNA, 16S-23S ISR) across multiple species | Enables detection of >143 Mycoplasma species with single primer set [29] | Custom-designed primers; MycoScope primer set [30] |
| Commercial PCR Kits | Provides optimized reagent mixtures for specific detection | Validated for sensitivity (<5 genomes/μL) and species coverage [30] | MycoSEQ (Life Technologies), MycoTOOL (Roche), VenorGEM (Minerva Biolabs) [28] |
| Culture Media Systems | Supports growth of fastidious Mycoplasma species | Essential for compendial methods; requires quality control of components | Hayflick's broth/agar, SP4 medium, A7 agar [28] [27] |
| Nucleic Acid Extraction Kits | Isulates DNA while removing inhibitors | Column purification with DNase treatment reduces false positives [24] | Qiagen column purification systems [24] |
| Positive Control Templates | Validates assay performance and sensitivity | Plasmid standards with target sequences; quantitated mycoplasma stocks [24] | pAlaidlawii, pMpneumoniae plasmid controls [24] |
| Enzymatic Master Mixes | Provides reaction components for amplification | Optimized for different Mycoplasma groups (Orale, Pneumoniae, Laidlawii) [24] | Group-specific master mixes (MMO, MMP, MML) [24] |
The comprehensive comparison between PCR and culture methods for mycoplasma detection reveals a consistent pattern of advantages favoring molecular approaches in most operational scenarios. PCR-based methods demonstrate superior sensitivity, dramatically reduced turnaround times (hours versus weeks), and enhanced capability to identify polymicrobial contamination that frequently eludes culture-based detection [31] [26]. The exceptional analytical sensitivity of modern qPCR assays, capable of detecting fewer than 5 mycoplasma genomes per microliter, provides an effective tool for early contamination detection before substantial impacts on cell systems or manufactured products occur [30].
Despite the performance advantages of molecular methods, traditional culture retains importance in regulatory frameworks and specific applications where viability determination is essential. The evolving regulatory landscape continues to recognize the value of both approaches, with current guidelines accepting properly validated PCR methods as alternatives to compendial culture-based testing [28]. Implementation decisions should consider specific operational requirements, including testing throughput, product shelf-life constraints, regulatory obligations, and technical expertise.
For most research and manufacturing environments, PCR-based detection offers compelling advantages that align with the need for rapid, sensitive, and comprehensive mycoplasma screening. The expanding availability of commercial kits with extensive validation data supports broader implementation across diverse laboratory settings. As molecular technologies continue to advance, further improvements in automation, multiplexing capability, and quantitative precision will likely strengthen the position of PCR-based methods as primary tools for ensuring mycoplasma control in both manufacturing and clinical contexts.
Protocols in Practice: Implementing PCR and Culture Methods for Robust Detection
The detection of Mycoplasma species remains a critical concern in both clinical diagnostics and biopharmaceutical quality control. For decades, culture-based methods have served as a traditional approach for pathogen detection. However, evolving diagnostic needs have revealed significant limitations inherent in these traditional techniques. This guide provides a detailed, objective comparison between conventional culture methods and modern PCR-based alternatives for mycoplasma detection, offering experimental data and methodologies to inform researchers, scientists, and drug development professionals in their diagnostic selection process.
Analytical Comparison: Culture vs. PCR for Mycoplasma Detection
The following table summarizes key performance metrics and characteristics of culture versus PCR methods for mycoplasma detection, synthesized from multiple experimental studies.
Table 1: Comprehensive Comparison of Culture and PCR Methods for Mycoplasma Detection
| Parameter | Culture Method | PCR-Based Methods | Experimental Context & Citations |
|---|---|---|---|
| Total Turnaround Time | 2-5 days for Ureaplasma spp. and M. hominis; Up to 8 weeks for M. genitalium [27]. As little as 1-3 weeks for M. pneumoniae [32]. | < 8 hours for standard multiplex PCR [27]. As little as 2 minutes to 1 hour with high-speed microfluidic platforms [33]. | Direct comparison from clinical specimen evaluation [27]. High-speed PCR data from technology review [33]. |
| Analytical Sensitivity (Limit of Detection) | Viable organisms required; inhibited by antibiotics [34]. | 8.8 - 10.8 CFU for multiplex PCR [27]. 46 copies/mL for automated real-time PCR [32]. 6.3 pg DNA (~8.2x103 genomic copies) for a specialized four-primer PCR [35]. | Limits determined via serial dilution experiments with quantified controls [27] [32] [35]. |
| Diagnostic Sensitivity | Lower; 21/85 specimens positive in a fertility clinic study [27]. | Higher; 28/85 specimens positive (including 11 culture-negative samples) in the same study [27]. "Significantly less false-negative" results compared to other methods [36]. | Based on a head-to-head clinical comparison of 85 patient specimens [27]. |
| Diagnostic Specificity & PPV/NPV | Considered the reference in some studies (though imperfect). | Specificity: 96%, PPV: 94%, NPV: 93% (vs. culture) [27]. | Calculated against a composite "true-positive" definition (positive by culture or dual PCR targets) [27]. |
| Key Requirements/Complexity | Complex Media Requirements: Specialized media essential (e.g., A7 agar), CO2 incubation, microscopic examination for "fried-egg" colonies [27] [32]. Expertise in culture techniques needed. | Primer Design & Thermal Cycling: Requires precise primer design, DNA polymerase, thermal cycler. Less dependent on operator skill for interpretation [37] [35]. | Methodological descriptions from experimental protocols [27] [37] [32]. |
| Ability to Detect VBNC States | Fails to detect Viable But Non-Culturable (VBNC) pathogens [34]. | Capable of detecting microbial DNA from VBNC organisms [34]. | VBNC state documented for pathogens like E. coli and K. pneumoniae [34]. |
| Impact of Biofilms | Poor detection of bacteria in biofilm state [34]. | Effective detection of bacterial DNA within biofilms [34]. | Biofilm-adapted variants often become culture-impaired [34]. |
Detailed Experimental Protocols
Culture Protocol for Genital Mycoplasmas
The following methodology was used in a direct comparison study with PCR [27].
- Specimen Collection & Transport: Cervical/vaginal swabs transported in 2SP medium. Urine samples concentrated via centrifugation (1,600 × g for 30 min) prior to testing.
- Culture Medium: Inoculation onto specialized A7 agar plates, which incorporate a direct test for urease to differentiate Ureaplasma species from other Mycoplasmatales.
- Incubation Conditions: Plates incubated at 37°C in 5% CO2 for up to 5 days.
- Detection & Identification: Daily microscopic examination for the appearance of typical mycoplasma colonies. Differentiation of species based on urease activity and colony morphology.
Multiplex PCR Protocol for Genital Mycoplasmas
This protocol details the multiplex PCR used in the comparative study [27].
- DNA Extraction: Bacterial DNA from 100 µL of specimen was isolated using a lysis buffer, followed by phenol-chloroform-isoamyl alcohol extraction, isopropanol precipitation, and resuspension in RNase-DNase-free water.
- Primer Targets:
- Ureaplasma spp.: urease gene (429 bp product)
- M. genitalium: 140-kDa adhesion protein gene (282 bp product)
- M. hominis: 16S rRNA gene (334 bp product)
- PCR Reaction: Hot-start PCR in 50-µL reactions containing:
- 0.2 mM dNTPs
- 10 mM Tris, 3 mM MgCl2
- 25 pmol of each unlabeled forward primer and biotin-labeled reverse primer
- 1.25 U of Gold Taq DNA polymerase
- Thermal Cycling Profile:
- Initial denaturation: 95°C for 10 min
- 35 cycles of:
- Denaturation: 95°C for 15 s
- Annealing/Extension: 60°C for 60 s
- Final extension: 72°C for 5 min
- Product Detection: Biotinylated PCR products were detected via Enzyme-Linked Oligosorbent Assay (ELOSA) using horseradish peroxidase-labeled probes specific to each target.
A Universal PCR Protocol for Mycoplasma Testing in Cell Cultures
A recent study designed a robust PCR method for routine mycoplasma screening in cell cultures, utilizing ultra-conserved primers [35].
- Primer Design: Primers were bioinformatically designed against highly conserved 16S rRNA regions, providing coverage for 92% of all species across the six orders of the class Mollicutes.
- Four-Primer PCR: The reaction includes:
- Myco-primers: Target the conserved mycoplasma 16S rRNA sequence (producing a 166-191 bp product).
- Uc48-primers: Target a universal eukaryotic DNA sequence (producing a 105 bp product) serving as an internal control for the presence of amplifiable eukaryotic cell DNA.
- Sensitivity: The reported limit of detection for this assay is 6.3 pg of M. orale genomic DNA, equivalent to approximately 8.21 × 103 genomic copies [35].
Workflow Visualization: Culture vs. PCR
The following diagram illustrates the significant differences in steps, time, and complexity between the culture and PCR methodologies for mycoplasma detection.
The Scientist's Toolkit: Essential Research Reagents
The following table details key reagents and their functions required for implementing the PCR-based detection methods described in this guide.
Table 2: Essential Reagents for PCR-Based Mycoplasma Detection
| Reagent / Solution | Function / Purpose | Specific Examples / Notes |
|---|---|---|
| Specialized Primers | Bind to complementary sequences of the target mycoplasma DNA for specific amplification. | Primers targeting urease gene (Ureaplasma), 140-kDa protein (M. genitalium), 16S rRNA (M. hominis) [27], or ultra-conserved 16S rRNA regions for broad detection [35]. |
| Thermostable DNA Polymerase | Enzyme that synthesizes new DNA strands by adding dNTPs to the primer-template hybrid. Withstands high temperatures of PCR cycling. | Gold Taq [27]. Hot-start versions (e.g., GoTaq G2 Hot Start) are common to reduce non-specific amplification [37]. |
| Deoxynucleoside Triphosphates (dNTPs) | The building blocks (A, T, C, G) used by the DNA polymerase to synthesize new DNA strands. | Typically used as a mixture at 0.2 mM concentration in the reaction [27]. |
| Reaction Buffer with MgCl₂ | Provides optimal chemical environment (pH, ionic strength) for polymerase activity. Mg²⁺ is a essential cofactor for the enzyme. | Often supplied with the polymerase. Concentration may require optimization (e.g., 3 mM MgCl₂) [27]. |
| DNA Lysis & Extraction Buffers | Break open cells and inactivate nucleases to release microbial DNA. Used to purify DNA from clinical samples. | NucliSens lysis buffer [27]. Commercial kits for automated extraction on platforms like MagNA Pure 96 [32]. |
| Positive Control DNA | Contains the target sequence to validate the PCR assay's performance and monitor for false negatives. | Can be a cloned amplicon in a plasmid, quantified precisely using methods like droplet digital PCR (ddPCR) [32]. |
| Detection Reagents | Used to visualize and confirm the amplified PCR product. | Biotin-labeled primers with enzyme-linked detection [27], fluorescent probes (TaqMan) for real-time PCR [32] [38], or intercalating dyes for gel electrophoresis. |
The experimental data and protocols presented in this guide objectively demonstrate the profound methodological advantages of PCR over culture for mycoplasma detection. The most striking differentiator is time: PCR reduces diagnostic turnaround from days or weeks to hours, enabling rapid clinical decision-making and streamlining research and quality control workflows [27] [32]. Furthermore, PCR demonstrates superior analytical and diagnostic sensitivity, capable of detecting organisms at low concentrations and in states (like VBNC and within biofilms) that evade culture entirely [27] [34] [36]. While culture maintains a historical role, the evidence strongly supports the adoption of PCR-based methodologies as the more efficient, sensitive, and reliable standard for mycoplasma detection in modern scientific and clinical practice.
Polymerase chain reaction (PCR) has revolutionized molecular diagnostics by enabling rapid, sensitive detection of pathogens that are difficult to culture, such as Mycoplasma pneumoniae. This guide explores the complete PCR workflow, with a specific focus on how its performance compares to conventional culture methods for mycoplasma detection. Understanding this workflow is crucial for researchers and drug development professionals seeking to implement optimal diagnostic strategies for respiratory pathogens and other infectious agents. We will examine each step of the PCR process, supported by experimental data comparing its sensitivity, turnaround time, and clinical utility against traditional culture-based approaches.
The PCR Workflow: A Step-by-Step Guide
The PCR process transforms a clinical sample into a detectable result through a series of carefully controlled steps. The entire pathway, from sample collection to result interpretation, can be visualized in the following workflow:
Step 1: Sample Collection
The PCR process begins with proper sample collection, which critically influences diagnostic accuracy. For respiratory pathogens like Mycoplasma pneumoniae, sampling site selection is particularly important. Recent comparative research demonstrates that oropharyngeal swabs show significantly better sensitivity (96.2%) compared to nasopharyngeal swabs (74.9%) for M. pneumoniae detection [12]. Samples are collected using specialized swabs and immediately placed in appropriate transport media to preserve nucleic acid integrity until processing.
Step 2: Nucleic Acid Extraction
Once samples reach the laboratory, nucleic acids (DNA or RNA) must be purified from the clinical specimen. This process involves:
- Cell Lysis: Breaking open cells and pathogens to release nucleic acids using chemical or mechanical methods
- Purification: Separating nucleic acids from proteins, lipids, and other cellular components
- Elution: Collecting purified nucleic acids in a suitable buffer for PCR amplification
Common extraction methods include column-based kits (e.g., QIAamp DNA Mini Kit) [12] [39] or automated extraction systems [40]. The quality and purity of extracted nucleic acids directly impact PCR efficiency and reliability.
Step 3: PCR Reaction Setup
The PCR reaction requires precise combination of several components:
- Template DNA: The extracted nucleic acids from the sample
- Primers: Short, specific DNA sequences that flank the target region
- Nucleotides (dNTPs): Building blocks for new DNA strands
- DNA Polymerase: Thermostable enzyme that synthesizes new DNA
- Buffer Solution: Optimal chemical environment for the reaction
- Probes (in qPCR): Fluorescently-labeled sequences for detection
These components are combined in a master mix to ensure reaction consistency across multiple samples.
Step 4: Thermal Cycling and Amplification
The PCR amplification process occurs in a thermal cycler that precisely controls temperature cycles. Each cycle consists of three core steps that are repeated 30-45 times:
This exponential amplification theoretically produces billions of copies of the target sequence from a single starting molecule.
Step 5: Detection and Analysis
Detection methods vary by PCR type:
- Gel Electrophoresis: Visualizing amplified DNA fragments in traditional PCR
- Fluorescence Detection: Real-time monitoring in qPCR using SYBR Green or target-specific probes
- Melting Curve Analysis: Differentiating amplification products by their melting temperatures [40]
Results are interpreted based on cycle threshold (Ct) values, presence/absence of specific amplification, or melting temperature profiles compared to appropriate controls.
PCR vs. Culture: Comparative Experimental Data
Sensitivity and Detection Rates
Multiple recent studies directly compare the performance of PCR versus culture methods for pathogen detection. The data reveal consistent patterns across different applications:
Table 1: Sensitivity and Detection Rate Comparison
| Pathogen/Application | PCR Sensitivity/Detection Rate | Culture Sensitivity/Detection Rate | Study Details |
|---|---|---|---|
| Mycoplasma pneumoniae (Oropharyngeal) | 96.2% sensitivity [12] | Not reported | 422 participants, RT-PCR reference |
| Mycoplasma pneumoniae (Nasopharyngeal) | 74.9% sensitivity [12] | Not reported | 422 participants, RT-PCR reference |
| Complicated UTI Pathogens | 43.52% polymicrobial detection rate [41] | 31.95% polymicrobial detection rate [41] | 773 patients, multicenter RCT |
| Mycoplasma pneumoniae (ddPCR) | 2.9 copies/reaction detection limit [39] | Culture rarely performed clinically [39] | 178 clinical samples |
Turnaround Time and Practical Considerations
Beyond sensitivity, practical implementation factors significantly impact diagnostic utility:
Table 2: Practical Performance Metrics
| Parameter | PCR Method | Culture Method | Clinical Implications |
|---|---|---|---|
| Turnaround Time | 1.5-49.68 hours [42] [40] | 104.4 hours [42] | Faster treatment decisions |
| Cost per Test | ~$5 (laboratory-developed tests) [40] | Typically higher | Significant savings in outbreak settings |
| Additional Capabilities | Resistance gene detection, quantification, multiplexing [12] [40] | Phenotypic susceptibility only | Comprehensive pathogen profiling |
PCR methods demonstrate clear advantages in detection speed, with turnaround times approximately 50-70% faster than culture methods. This temporal advantage enables more timely clinical decision-making and treatment optimization.
Advanced PCR Methodologies
Digital Droplet PCR (ddPCR)
Digital droplet PCR represents a significant technological advancement in nucleic acid detection. This method partitions a sample into thousands of nanoliter-sized droplets, with PCR amplification occurring in each individual droplet. This approach enables absolute quantification of target nucleic acids without requiring standard curves, offering enhanced precision for low-abundance targets [39].
For Mycoplasma pneumoniae detection, ddPCR has demonstrated a detection limit of 2.9 copies/reaction compared to 10.8 copies/reaction for real-time PCR [39]. This improved sensitivity is particularly valuable for monitoring treatment efficacy, as studies show significant correlation between bacterial load and disease severity.
Multiplex PCR Platforms
Multiplex PCR technologies enable simultaneous detection of multiple pathogens in a single reaction. Recent developments include fluorescence melting curve analysis (FMCA)-based systems capable of detecting six respiratory pathogens, including M. pneumoniae, with limits of detection between 4.94 and 14.03 copies/μL [40]. These platforms maintain high precision (intra-/inter-assay CVs ≤ 0.70% and ≤ 0.50%) while reducing cost per sample to approximately $5—86.5% cheaper than commercial kits [40].
Research Reagent Solutions
Successful PCR implementation requires specific laboratory reagents and materials. The following table outlines essential components for mycoplasma detection workflows:
Table 3: Essential Research Reagents for PCR-Based Mycoplasma Detection
| Reagent/Material | Function | Examples/Specifications |
|---|---|---|
| Specimen Collection Swabs | Sample acquisition | Nipro sponge swab TYPE L [12] |
| Transport Media | Preserve sample integrity during transport | Universal transport medium (UTM) [12] |
| Nucleic Acid Extraction Kits | Isolate DNA/RNA from clinical samples | QIAamp DNA Mini Kit [12] [39] |
| PCR Master Mix | Provide optimized reaction components | One Step U* Mix [40] |
| Specific Primers/Probes | Target amplification and detection | CARDS toxin gene primers [12], P1 gene primers [39] |
| Positive Controls | Verify assay performance | MP reference strain M129 (ATCC 29342) [12] |
| Thermal Cyclers | Temperature cycling for amplification | AriaMx Real-Time PCR System [12] |
The complete PCR workflow—from careful sample collection through nucleic acid extraction, amplification, and detection—offers significant advantages over traditional culture methods for mycoplasma detection and other diagnostic applications. Experimental data consistently demonstrate PCR's superior sensitivity, faster turnaround times, and enhanced capability to detect polymicrobial infections and resistance markers.
For researchers and drug development professionals, these technical advantages translate to improved clinical outcomes. PCR-guided therapy has demonstrated significantly better clinical outcomes (88.08% vs. 78.11%) compared to culture-guided approaches [42]. Furthermore, quantitative PCR methods enable correlation between bacterial load and disease severity, providing valuable insights for both diagnosis and therapeutic monitoring [39].
As PCR technologies continue to evolve with developments in digital PCR, multiplex platforms, and point-of-care applications, their role in clinical diagnostics and research will undoubtedly expand, offering increasingly sophisticated solutions for pathogen detection and characterization.
Polymersse Chain Reaction (PCR) technology has undergone a remarkable evolution since its inception, transforming from a manual, endpoint detection method to sophisticated, automated systems capable of precise quantification and high-throughput analysis. This evolution has been particularly significant in applied research and diagnostic fields, where the detection of contaminants like mycoplasma in cell cultures presents substantial challenges for both research integrity and biopharmaceutical safety. Traditional culture-based methods, while historically considered the "gold standard," require extensive incubation periods of up to several weeks, delaying critical decisions in cell line maintenance and drug development pipelines [6].
The emergence of advanced PCR formats has fundamentally addressed these limitations. Real-time quantitative PCR (qPCR) introduced the capability to monitor amplification as it occurs, providing not just detection but quantification of target nucleic acids. Further advancements led to digital PCR (dPCR), which offers absolute quantification without standard curves by partitioning samples into thousands of individual reactions [43]. Concurrently, automation and high-throughput platforms have revolutionized laboratory workflows, enabling processing of hundreds of samples with minimal manual intervention, thereby enhancing reproducibility and reducing contamination risks [44] [45]. This guide objectively compares these advanced PCR platforms, with experimental data framed within the critical context of mycoplasma detection sensitivity research.
Technical Comparison of Advanced PCR Platforms
The selection of an appropriate PCR platform depends on multiple factors, including required precision, throughput needs, and operational constraints. The table below provides a structured comparison of the dominant modern PCR systems based on 2025 market and performance data [46].
Table 1: Comparison of Key PCR Platforms for Research and Diagnostic Applications
| Platform | Best For | Key Technology & Features | Multiplexing Capacity | Typical Price Range |
|---|---|---|---|---|
| Roche LightCycler PRO | Overall Performance & Hybrid Workflows | Vapor chamber cooling (< ±0.2°C variance), IVD/Research modes, interchangeable 96/384-well blocks | Up to 7 optical channels | $35,000 – $55,000 |
| Bio-Rad CFX Opus 384 | Speed & High Throughput | Cloud connectivity (BR.io platform), optical shuttle, rapid scan time (<20 sec for full plate) | 4-target multiplexing | $40,000 – $55,000 |
| Stilla Nio+ | High-End Digital PCR | Crystal Digital PCR (droplet-based), absolute quantification without standard curves | 7-color multiplexing | $80,000 – $120,000+ |
| Thermo Fisher MiniAmp Plus | Budget-Conscious Labs & Routine PCR | Compact footprint, VeriFlex blocks for temperature optimization, 5-inch touchscreen | Standard endpoint PCR | $3,500 – $5,800 |
Performance Analysis and Selection Criteria
- Throughput and Efficiency: For core facilities and large-scale testing environments, the Bio-Rad CFX Opus 384 and similar 384-well systems are unparalleled. Their ability to process hundreds of samples in a single run, coupled with integrated cloud platforms for remote data management, dramatically increases laboratory efficiency [46]. This is crucial in applications like routine biobank screening for mycoplasma.
- Precision and Sensitivity: When the experimental question demands the highest possible accuracy, particularly for low-abundance targets or subtle copy number variations, digital PCR (dPCR) platforms like the Stilla Nio+ demonstrate superior performance. A 2024 study on respiratory viruses found that dPCR "demonstrated superior accuracy, particularly for high viral loads... and greater consistency and precision than Real-Time RT-PCR" [43]. This makes dPCR ideal for validating reference materials or quantifying minimal residual disease.
- Flexibility and Cost-Effectiveness: The Roche LightCycler PRO represents a balanced "all-in-one" solution, especially for labs that transition between assay development and clinical application. Its vapor chamber technology ensures exceptional temperature uniformity, reducing edge effects and improving data validity across the plate [46]. For basic amplification needs, budget-friendly options like the Thermo Fisher MiniAmp Plus provide reliable performance for genotyping and colony PCR.
Experimental Comparison: PCR vs. Culture for Mycoplasma Detection
Methodology for Comparative Studies
The superiority of PCR-based methods over traditional culture for mycoplasma detection is well-documented through standardized comparative studies. A typical experimental protocol involves testing a panel of cell lines with known contamination status using multiple methods in parallel [6] [47].
Sample Preparation:
- Collect supernatant from cell cultures grown in antibiotics-free medium for at least 4-7 days.
- For DNA-based methods (qPCR, dPCR), extract nucleic acids using commercial kits (e.g., QIAamp DNA Mini Kit, MagMax Viral/Pathogen kit). Some advanced qPCR assays can bypass DNA extraction, using cell culture medium directly [47].
- For culture methods, inoculate samples into both liquid and solid mycoplasma-specific media.
Testing Methods:
- Microbial Culture (Gold Standard): Inoculate samples into liquid broth (e.g., Hayflick's medium) and observe for color change due to metabolic activity. Subculture positive broths onto agar plates and incub anaerobically for up to 28 days. Colonies are identified by their characteristic "fried-egg" morphology [6].
- Real-Time PCR (qPCR): Perform using commercial kits (e.g., PromoKine) or laboratory-developed tests targeting the 16S rRNA gene. Use an internal control to monitor for PCR inhibition. The reaction is performed on platforms like the Bio-Rad CFX96 [6] [43].
- Digital PCR (dPCR): Partition the PCR mixture into thousands of nanoscale reactions on platforms like the QIAcuity or Stilla Nio+. After endpoint amplification, count the positive and negative partitions to calculate the absolute copy number of the target without a standard curve [43].
Table 2: Sensitivity Comparison of Mycoplasma Detection Methods from Experimental Studies
| Detection Method | Principle | Time to Result | Sensitivity | Specificity | Key Applications |
|---|---|---|---|---|---|
| Microbial Culture | Growth in specialized media | 2 to 4 weeks | ~10⁴ CFU/mL | 100% | Regulatory gold standard, required for product release [6] |
| Conventional PCR | Endpoint DNA amplification & gel detection | 6 to 8 hours | ~10³ to 10⁴ CFU/mL | 100% | Historical method for presence/absence testing [6] |
| Real-Time PCR (qPCR) | Fluorescence-based kinetic monitoring | 2 to 3 hours | <10 CFU/mL [47] | 100% [6] | Routine screening, rapid batch testing, high throughput [6] |
| Digital PCR (dPCR) | Absolute quantification by sample partitioning | 3 to 5 hours | Single copy detection | 100% (inferred) | Rare target detection, assay standardization, absolute quantification [43] |
Workflow Visualization
The following diagram illustrates the typical workflow for a comparative study evaluating different mycoplasma detection methods, highlighting the significant differences in time and complexity.
The Role of Automation and High-Throughput Systems
The integration of closed-system automation represents a paradigm shift in molecular diagnostics, directly addressing challenges of contamination, reproducibility, and staffing shortages in laboratories.
Contamination Control and Standardization: Automated systems, such as Cellares' Cell Shuttle or integrated QC platforms, utilize single-use consumable cartridges that keep patient material within a closed system from initial loading until harvest. This significantly reduces manual interventions and the associated risks of contamination—a critical advantage in sensitive mycoplasma testing and cell therapy manufacturing [44]. As noted in industry analysis, "the more intervention you have the more risk you might incorporate, and the more controls you need to exert to make sure that you reduce those risks" [44].
Addressing Workforce and Workflow Challenges: A 2025 survey of 400 laboratory professionals revealed that 89% agree automation is critical for keeping up with testing demand, with 95% viewing it as key to improving patient care [45]. These systems can consolidate up to 25 manual tasks, reducing hours of work to minutes and allowing skilled technologists to focus on complex data analysis and troubleshooting rather than repetitive pipetting.
Data Integrity and Compliance: Automated QC platforms seamlessly integrate instruments like cell counters, flow cytometers, and PCR systems with robotic liquid handlers. This automation streamlines the majority of in-process and release testing assays, from sample loading to automated data upload into Laboratory Information Management Systems (LIMS), creating reliable electronic batch records and enhancing regulatory compliance [44].
Essential Research Reagent Solutions
The performance of any PCR assay is fundamentally dependent on the quality and suitability of the reagents used. The following table details key components essential for establishing robust mycoplasma detection protocols.
Table 3: Essential Research Reagents for PCR-Based Mycoplasma Detection
| Reagent / Kit | Function | Application Notes |
|---|---|---|
| Nucleic Acid Extraction Kits (e.g., QIAamp DNA Mini Kit, MagMax Viral/Pathogen) | Isolates and purifies DNA from cell culture supernatants, removing PCR inhibitors. | Critical for sample prep; automated systems like KingFisher Flex or STARlet can standardize this step [43] [48]. |
| Multiplex Real-Time PCR Kits (e.g., Allplex Respiratory Panels, PromoKine Mycoplasma kits) | Contains optimized primer-probe mixes, enzymes, and buffers for specific target detection. | Enable simultaneous detection of multiple mycoplasma species or respiratory viruses in one reaction [43] [6]. |
| dPCR Reagent Kits & Nanoplates | Formulated for optimal partitioning and amplification in digital PCR systems (QIAcuity, Stilla Nio+). | Often platform-specific; require optimization of primer-probe concentrations to minimize cross-reactivity [43] [46]. |
| Mycoplasma Culture Media (Liquid Broth & Agar) | Supports the growth of fastidious mycoplasma organisms for the gold standard method. | Used for method validation and as a required reference standard in regulatory submissions [6]. |
| Internal Control Templates | Non-target nucleic acid sequence co-amplified with the sample. | Monitors for PCR inhibition and verifies extraction efficiency, ensuring result reliability [47]. |
The comparative data clearly establishes that advanced PCR formats—particularly real-time qPCR and dPCR—offer transformative advantages over traditional culture methods for mycoplasma detection. The dramatic reduction in time-to-result (from weeks to hours), coupled with significantly enhanced sensitivity (enabling detection of less than 10 CFU/mL), makes these molecular methods indispensable for modern biopharmaceutical quality control and research integrity [6] [47].
Looking forward, the convergence of digital PCR, closed-system automation, and artificial intelligence will further redefine the diagnostic landscape. dPCR is increasingly being adopted for applications requiring absolute quantification, such as monitoring viral load in cell therapies and liquid biopsy analysis [43] [49]. Simultaneously, the market is witnessing a rapid shift toward multiplexing and fully automated, integrated systems that streamline workflows from sample-in to answer-out [49] [45]. For researchers and drug development professionals, the strategic selection of a PCR platform must balance current throughput and precision needs with future scalability, leveraging these technological advancements to ensure both scientific rigor and patient safety.
Mycoplasma contamination represents a significant risk to the safety, efficacy, and quality of cell and gene therapy (CGT) products. These wall-less bacteria can alter cell metabolism, affect product characteristics, and potentially harm patients [50]. Regulatory authorities worldwide mandate mycoplasma testing as a required release test before these advanced therapies can be administered to patients [51]. For CGT products with short shelf-lives—often measured in hours or days rather than weeks—the traditional 28-day compendial culture method is entirely unsuitable [51] [52]. This case study objectively compares the performance of rapid polymerase chain reaction (PCR) methods against traditional culture-based alternatives for mycoplasma detection, focusing on the specific constraints imposed by short product shelf-lives.
Methodological Comparison: PCR vs. Culture-Based Detection
Core Principles and Mechanisms
Culture-Based Methods represent the historical "gold standard" for mycoplasma detection. This approach involves inoculating samples into both liquid broth and solid agar media that support the growth of a wide range of mycoplasma species. The liquid media is observed for color change indicating metabolic activity, while the solid agar is examined for the characteristic "fried-egg" colony morphology under microscopy. The major limitation of this method is the extended time required—at least 28 days—to confirm negative results, as some mycoplasma species grow very slowly [50] [47].
PCR-Based Methods utilize nucleic acid amplification technology to detect mycoplasma DNA directly from samples. These tests target specific genetic sequences present across multiple mycoplasma species, enabling detection without requiring bacterial growth. Modern PCR systems available for CGT manufacturing can detect over 130 mycoplasma species in less than one hour, providing a significant advantage for products with limited shelf-lives [51].
Performance Comparison: Sensitivity, Specificity, and Speed
Table 1: Direct Comparison of Mycoplasma Detection Methods
| Parameter | Culture-Based Methods | PCR-Based Methods | DAPI Staining |
|---|---|---|---|
| Time to Result | ≥28 days [51] [47] | ~1 hour to 3 hours [51] [8] | ~1-10 days (with indicator cells) [50] |
| Limit of Detection | Varies with medium preparation [50] | ≤10 CFU/mL [51] [47] | Lower sensitivity than PCR [8] |
| Species Coverage | Cultivable species only [50] | 130+ species [51] | Wide range but may miss low-level contamination [50] |
| Regulatory Status | Compendial method [47] | Accepted alternative method [52] | Often used with culture methods [50] |
| Suitable for Short Shelf-Life Products? | No | Yes | Potentially, but sensitivity concerns |
The data clearly demonstrates PCR's superior speed while maintaining excellent sensitivity. The BIOFIRE Mycoplasma test, for instance, offers a limit of detection (LOD) of ≤10 CFU/mL in approximately one hour, making it particularly suitable for autologous cell therapies that require rapid release testing before patient administration [51].
Experimental Data and Validation Protocols
Validation of a Rapid PCR Assay for CGT Products
Recent technological advancements have led to the development and validation of rapid PCR assays specifically designed for CGT applications. One study validated an internally-controlled Taqman-based real-time PCR assay that can detect less than 10 colony-forming units (CFU) of the most frequently encountered mycoplasma contaminants in mammalian cell cultures without the need for DNA extraction [47].
The experimental protocol involved:
- Sample Preparation: Cell culture medium was used directly without DNA extraction, simplifying the workflow.
- Amplification Technology: Taqman-based real-time PCR with internal controls to monitor for inhibition.
- Sensitivity Testing: The assay was validated to detect <10 CFU for common contaminant species.
- Specificity Assessment: No cross-reactivity with other bacteria or mammalian cells was observed [47].
This validation approach aligns with regulatory expectations for implementing alternative methods in cGMP manufacturing environments for CGT products [52].
Sample Processing Considerations for CGT Matrices
A critical challenge in mycoplasma testing for CGT products is the presence of mammalian cells in the sample, which can interfere with PCR detection. A double centrifugation protocol has been validated for sample preparation with a LOD of ≤10 CFU/mL [51]. However, this approach excludes mammalian cells, creating limitations for certain therapy types.
Recent developments address this constraint. New draft European pharmacopeia guidelines acknowledge the need for methods that can handle samples containing cells, prompting the development of modified protocols that maintain sensitivity while accommodating complex CGT matrices [51].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Research Reagent Solutions for Mycoplasma Detection
| Reagent/Equipment | Function | Example Products/Applications |
|---|---|---|
| Closed-System PCR Kits | Sample-to-answer mycoplasma detection with minimal hands-on time | BIOFIRE Mycoplasma test [51] |
| Nucleic Acid Extraction Kits | DNA purification from complex matrices | QIAamp DNA Mini Kit [12] |
| Cell Culture Media | Supports mycoplasma growth in culture methods | PPLO broth with supplements [50] |
| Fluorescent Stains | Direct visualization of mycoplasma DNA | DAPI, Hoechst dyes [50] |
| Validated Reference Strains | Method qualification and validation | M. hominis, M. fermentans, M. arginini [50] |
| Enzymatic Labeling Systems | Enhanced detection sensitivity | Nick translation with modified nucleotides [50] |
| Quality Control Materials | Monitoring assay performance | Internal controls, synthetic DNA standards [12] |
The toolkit highlights the importance of integrated systems that provide both the reagents and platform for rapid mycoplasma testing. Closed-system PCR tests are particularly valuable for CGT manufacturing as they reduce contamination risk and require minimal technical expertise to operate [51].
Regulatory Considerations for CGT Applications
The regulatory landscape for mycoplasma testing in CGT products has evolved significantly. While traditional culture methods were once strictly required, regulatory guidance now accepts validated rapid molecular methods, including real-time quantitative PCR-based tests [52].
Key regulatory considerations include:
- Method Validation: Demonstration of sensitivity, specificity, robustness, and reproducibility for the specific CGT matrix.
- Sample Selection: Choosing representative samples that accurately reflect potential contamination in the final product.
- Sample Preparation: Techniques that overcome matrix effects while maintaining sensitivity requirements [52].
Regulatory experts emphasize the importance of early engagement with health authorities when implementing rapid mycoplasma testing methods for CGT products with short shelf-lives [52].
For cell and gene therapy products with short shelf-lives, rapid PCR-based methods provide a scientifically sound and regulatory-accepted alternative to traditional culture-based mycoplasma testing. The compelling experimental data demonstrates that PCR delivers equivalent or superior sensitivity with results in hours rather than weeks, addressing the critical time constraints of these advanced therapies. While culture methods remain the historical gold standard, PCR technology has matured to offer validated, robust solutions that ensure product safety without compromising shelf-life. As the CGT field continues to expand, the implementation of rapid mycoplasma detection methods will be essential for scaling manufacturing and delivering these transformative treatments to patients in a timely manner.
Overcoming Detection Challenges: Sample Type, Inhibitors, and Optimal Protocols
A Comparative Guide for Molecular Diagnostics in Mycoplasma Detection
For researchers and drug development professionals, the choice between oropharyngeal and nasopharyngeal sampling is a critical pre-analytical variable that can dramatically impact the sensitivity of PCR-based Mycoplasma detection. While molecular methods like PCR have revolutionized diagnostic speed, their ultimate performance is inextricably linked to the quality and type of sample collected. This guide objectively compares sample types using recent experimental data, framing the findings within the broader thesis of PCR versus culture method sensitivity.
Quantitative Data Comparison: Oropharyngeal vs. Nasopharyngeal Swabs
Recent clinical studies provide compelling quantitative evidence for the superior sensitivity of oropharyngeal sampling for detecting Mycoplasma pneumoniae (MP) in respiratory infections.
Table 1: Diagnostic Performance of PCR for Mycoplasma pneumoniae by Sample Type
| Sample Type | Reference Test | Sensitivity (%) | Specificity (%) | Study Details | Citation |
|---|---|---|---|---|---|
| Oropharyngeal Swab | RT-PCR (residual extract) | 96.2 (92.3–98.4) | 100.0 (98.5–100.0) | 422 participants; Smart Gene Myco PCR test | [12] |
| Nasopharyngeal Swab | RT-PCR (residual extract) | 74.9 (67.9–81.0) | 99.2 (97.0–99.9) | 422 participants; Multiplex PCR test | [12] |
| Combined Oropharyngeal-Nasal (ON) Swab | Composite Standard | 94 (86–98) | - | 273 pediatric sample pairs; BioFire RP2.1 panel | [53] |
| Nasopharyngeal (NP) Swab | Composite Standard | 64 (61–75) | - | 273 pediatric sample pairs; BioFire RP2.1 panel | [53] |
The data unequivocally demonstrates that oropharyngeal samples yield significantly higher sensitivity than nasopharyngeal samples for detecting Mycoplasma pneumoniae [12]. The combined oropharyngeal-nasal (ON) swab, a less-invasive alternative, also shows a marked improvement in diagnostic yield over the traditional nasopharyngeal swab [53].
Detailed Experimental Protocols
Understanding the methodology behind this data is crucial for evaluating its validity and applicability.
Protocol: Head-to-Head Comparison in a Pediatric Cohort
This 2024 study directly compared nasopharyngeal and oropharyngeal swabs collected simultaneously from the same pediatric patients [12].
- Study Design & Sample Collection: The study involved 422 symptomatic children. For each participant, a physician collected both samples at the same time.
- Nasopharyngeal Sample: Collected using a swab and universal transport medium (UTM).
- Oropharyngeal Sample: Collected using a sponge swab and extraction buffer.
- PCR Testing & Analysis: The samples were tested in parallel using different commercially available PCR tests. To validate these results, residual DNA extracts from both sample types were also tested with an in-house real-time PCR (RT-PCR) targeting the CARDS toxin gene. The RT-PCR results from the oropharyngeal swab were used as the reference standard to calculate the sensitivity and specificity of the other tests. DNA load was also quantified [12].
- Key Findings: The sensitivity of the PCR test using oropharyngeal samples was over 20% higher than when using nasopharyngeal samples. The study concluded that oropharyngeal samples should be preferred for MP detection [12].
Protocol: Evaluation of a Combined Swab Method
This 2025 study evaluated a parent-collected combined oropharyngeal-nasal (ON) swab against a healthcare worker-collected nasopharyngeal (NP) swab in young children [53].
- Study Design & Sample Collection: The study was conducted in two phases. In the research phase, 139 matched sample pairs were collected from children (0-4 years old). In the implementation phase during a high MP incidence period, 219 additional pairs were collected.
- ON Swab: Parents/caregivers collected the swab by first swabbing the back of the throat (oropharynx), then the inside of both nostrils.
- NP Swab: Healthcare workers collected a standard nasopharyngeal swab.
- PCR Testing & Analysis: Samples were tested using the BioFire Respiratory Panel 2.1. A composite reference standard (positive result on either ON or NP swab) was used for sensitivity calculations. Acceptability was assessed via a caregiver questionnaire [53].
- Key Findings: The ON swab demonstrated significantly higher sensitivity for MP detection (94% vs. 64%) and was rated as significantly more acceptable by parents and caregivers [53].
Diagram 1: Experimental workflow and outcome comparison for different swab types in Mycoplasma pneumoniae detection.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Oropharyngeal and Nasopharyngeal Sampling
| Item | Function | Example Products & Specifications |
|---|---|---|
| Flocked Swabs | Sample collection from mucosal surfaces. Designed to release biological material efficiently. | Copan FLOQSwab [53]; Nipro sponge swab TYPE L [12] |
| Universal Transport Media (UTM) | Preserves viral and bacterial nucleic acids during transport and storage. | Copan UTM Nasopharyngeal Sample Collection Kit [12] |
| Nucleic Acid Extraction Kits | Isolates high-purity DNA from swab samples for downstream PCR analysis. | QIAamp DNA Mini Kit (Qiagen) [12] |
| PCR Assays & Platforms | Detects and identifies Mycoplasma DNA through nucleic acid amplification. | Smart Gene Myco (MIZUHO MEDY) [12]; BioFire RP2.1 (bioMérieux) [53]; SpotFire/FilmArray (bioMérieux) [12] |
| Real-Time PCR Systems | Quantifies DNA load and provides cycle threshold (Ct) values for detection. | AriaMx Real-Time PCR System (Agilent) [12] |
Broader Context: Sample Type in PCR vs. Culture Sensitivity
The emphasis on optimal sample type is a cornerstone of the paradigm shift from culture to PCR for mycoplasma detection.
- The Gold Standard's Blind Spot: Traditional culture methods, while specific, are slow (taking up to 28 days) and have high nutritional demands, making sample viability and fast processing critical [28] [47]. The choice of sample type is therefore equally, if not more, crucial for culture.
- PCR's Advantage and a Shared Pre-analytical Challenge: PCR's speed (results in hours) and ability to detect non-viable organisms reduce the dependency on perfect sample viability [54] [55]. However, as the data shows, PCR is not immune to pre-analytical errors. A poor-quality sample with low bacterial load will yield a false negative, regardless of the PCR assay's intrinsic sensitivity. The superior performance of oropharyngeal swabs is attributed to a higher concentration of M. pneumoniae in the throat compared to the nasopharynx [12].
- Impact of Antimicrobials: Prior antimicrobial treatment can reduce bacterial DNA load, further complicating detection. One study found a negative correlation between fluoroquinolone use and oropharyngeal DNA loads, highlighting that even the best sample type can be affected by pre-treatment [12].
In conclusion, within the thesis of PCR versus culture, the critical role of sample type remains a universal factor. The transition to highly sensitive molecular methods does not negate the need for optimal sampling; it reinforces it. For researchers and clinicians aiming to maximize the sensitivity of Mycoplasma pneumoniae detection, the evidence strongly supports the use of oropharyngeal or combined oropharyngeal-nasal swabs over traditional nasopharyngeal swabs.
Addressing PCR Inhibitors and Culture Fastidiousness for Reliable Results
Mycoplasma contamination represents a significant challenge in biopharmaceutical production and cell culture, potentially compromising product safety and experimental data. The detection of these fastidious microorganisms—notorious for their small size, lack of a cell wall, and complex nutritional requirements—hinges on two primary methodologies: traditional culture-based techniques and modern nucleic acid amplification tests (NAATs) like polymerase chain reaction (PCR). Each method grapples with distinct limitations; culture methods are hampered by the fastidious growth needs of Mycoplasma, while PCR is susceptible to inhibition from various substances. This guide provides an objective comparison of these detection methods, supported by experimental data, to assist researchers and drug development professionals in selecting and validating appropriate testing strategies for their specific applications.
Methodological Comparison: PCR vs. Culture
Experimental Protocol for Comparative Studies
A standardized approach for comparing Mycoplasma detection methods involves testing a panel of cultured Mollicutes species at defined concentrations. A representative proof-of-principle analysis follows this general protocol [28]:
- Strain Selection: A panel of type strains is selected based on international pharmacopeia standards (e.g., USP <63>, Ph. Eur. 2.6.7.) and their relevance as reported cell culture contaminants. Examples include Acholeplasma laidlawii, Mycoplasma fermentans, and Mycoplasma orale [28].
- Culture Conditions: Strains are cultured in enriched media such as Hayflick’s or SP4 broth and agar. Incubation proceeds at 35–37°C with 5% CO₂ for most species, or 30°C for Spiroplasma, until metabolic activity or colonies are observed [28].
- Sample Preparation: Cultured organisms are serially diluted in log-fold steps (e.g., from 1,000 CFU/mL to 1 CFU/mL) in a relevant biological matrix, such as cell therapy products like tumor-infiltrating lymphocytes (TILs), to assess matrix interference [28].
- Testing: Dilutions are tested in biological duplicates with multiple replicates per condition using both the compendial 28-day culture method and commercial PCR assays. The results are analyzed to determine limits of detection (LOD) and repeatability [28].
Quantitative Performance Data
The following table summarizes key performance characteristics of Mycoplasma detection methods, synthesizing data from comparative studies [28] [56] [57].
Table 1: Comparative Performance of Mycoplasma Detection Methods
| Feature | Traditional Culture Methods | PCR-Based Molecular Methods |
|---|---|---|
| Time to Result | Up to 28 days for compendial methods [28] [58] | Same day (a few hours) [58] [8] |
| Limit of Detection (LOD) | Varies by method; benchmark for molecular replacement is ≤10 CFU/mL [28] | Varies by assay; some kits meet the ≤10 CFU/mL standard, while others are less sensitive [28] |
| Sensitivity in Clinical Specimens | Detects ~33.9% of positive specimens in cUTI studies [57] | Detects ~52.3% of positive specimens in cUTI studies [57] |
| Ability to Detect Polymicrobial Contamination | Low (~3.6% of positive cultures) due to overgrowth by a single organism [57] | High (~46.2% of positive tests) [57] |
| Impact of Fastidious Nature | Significant; some species fail to grow on standard media, requiring specialized conditions [28] | Minimal; detection is based on genetic signature, not growth viability [8] |
| Impact of PCR Inhibitors | Not applicable | Significant; can cause false negatives, must be managed via sample purification, robust enzymes, or additives like BSA [59] [60] [61] |
| Regulatory Status | Gold standard (USP <63>); required for product release in some regions unless alternative methods are validated [28] [58] | Accepted alternative in Ph. Eur. and JP; considered an alternative method in USP requiring extensive validation [28] [58] [62] |
Understanding and Overcoming Key Challenges
The Challenge of Culture Fastidiousness
Mycoplasmas are fastidious because they lack many biosynthetic pathways and require highly enriched media for growth. This auxotrophy leads to extended incubation times—up to 28 days for compendial methods—making culture impractical for products with short shelf-lives, such as many advanced therapy medicinal products (ATMPs) [28] [58]. Furthermore, some species, like Mycoplasma hyorhinis and Mycoplasma pneumoniae, may fail to grow on standard media, necessitating the use of multiple culture conditions and increasing the complexity of the testing protocol [28].
The Challenge of PCR Inhibitors
PCR inhibitors are substances that prevent the amplification of nucleic acids, even when sufficient target DNA is present. They are the most common cause of amplification failure in PCR [59] [63]. These inhibitors can originate from the sample matrix (e.g., blood, cell culture components) or be introduced during sample processing and DNA extraction (e.g., phenol, ionic detergents) [59] [61] [63].
Mechanisms of Action: Inhibitors interfere with PCR through several mechanisms [59] [61] [63]:
- Interaction with DNA Polymerase: Direct binding or degradation of the enzyme (e.g., by proteases, heparin, or IgG).
- Depletion of Cofactors: Chelation of Mg²⁺ ions, which are essential for polymerase activity (e.g., by humic acids or EDTA).
- Interaction with Nucleic Acids: Binding to single or double-stranded DNA, preventing denaturation or primer annealing (e.g., by humic substances or polyphenols).
- Fluorescence Quenching: Interference with fluorescence detection in real-time PCR (e.g., by humic acid) [61].
Table 2: Common PCR Inhibitors and Their Sources
| Source | Common PCR Inhibitors |
|---|---|
| Biological Samples | Hemoglobin, lactoferrin, immunoglobulin G (IgG), heparin, urea, bile salts [59] [61] [63] |
| Cell Culture & ATMPs | Components from the culture medium, proteins, and other cellular debris [58] |
| Environmental Samples | Humic acids, fulvic acids, metal ions [61] [63] |
| Sample Processing | Ionic detergents (SDS, sarkosyl), organic solvents (phenol, ethanol), salts (KCl, NaCl) [59] [63] |
Solutions for Reliable PCR Detection
Several strategies can mitigate the effects of PCR inhibitors:
- Sample Purification: Commercial DNA extraction kits are designed to remove many inhibitors. Techniques like column chromatography (e.g., silica-based columns) or magnetic bead-based methods (e.g., Bioclone's BcMag Kit) can effectively purify DNA [59] [63].
- Use of Inhibitor-Tolerant Enzymes: Some DNA polymerases are engineered or discovered to be more resistant to specific inhibitors. Increasing the polymerase concentration can also help overcome mild inhibition [59] [61].
- Reaction Additives: Adding substances like bovine serum albumin (BSA) to the PCR reaction can bind to and neutralize certain inhibitors, particularly in samples derived from blood [59].
- Sample Dilution: Diluting the DNA extract reduces the concentration of inhibitors, but this also dilutes the target DNA and can reduce sensitivity [63].
- Quantifying Inhibition: A known amount of control template can be added to the sample reaction mixture. Comparing its amplification to a control reaction without inhibitors helps assess the level of inhibition in the sample [59] [63].
Workflow and Pathway Diagrams
The following diagram illustrates the logical workflow for selecting and validating a Mycoplasma detection method, highlighting the parallel challenges of fastidiousness and inhibition.
Research Reagent Solutions
The following table details key reagents and kits used in the featured experiments for reliable Mycoplasma detection.
Table 3: Essential Research Reagents for Mycoplasma Detection Studies
| Item | Function in Experiment |
|---|---|
| Hayflick's / SP4 Media | Enriched broth and agar required for the cultivation of fastidious Mycoplasma species [28]. |
| MycoSEQ Detection Kit (Thermo Fisher) | A SYBR Green-based qPCR kit for detecting Mycoplasma DNA; widely used with a track record of regulatory acceptance [28] [58] [62]. |
| MycoTOOL Detection Kit (Roche) | A real-time PCR kit for Mycoplasma detection; performance comparable to MycoSEQ in some studies [28]. |
| Biofire Mycoplasma Assay (bioMérieux) | A molecular assay noted for high sensitivity in comparative studies [28]. |
| Bovine Serum Albumin (BSA) | An additive to PCR reactions that can bind to and neutralize certain inhibitors, improving amplification reliability in complex matrices [59]. |
| Inhibitor-Tolerant DNA Polymerase (e.g., Q5 Taq) | Engineered or selected polymerases that maintain activity in the presence of common PCR inhibitors, crucial for direct PCR or dirty samples [60] [61]. |
| BcMag Inhibitor Removal Kit | Magnetic bead-based kit designed to selectively capture and remove PCR inhibitors from impure DNA samples prior to amplification [63]. |
The choice between PCR and culture for Mycoplasma detection involves a critical trade-off between speed and susceptibility to analytical challenges. Culture methods, while considered the gold standard, are inherently limited by the fastidious growth of Mycoplasma, leading to long turnaround times that are incompatible with modern, short-shelf-life biologics. PCR methods offer a rapid and sensitive alternative but require vigilant management of PCR inhibitors through robust purification protocols, reagent selection, and systematic validation. For researchers and drug development professionals, a deep understanding of these limitations and the implementation of the strategies outlined herein are essential for ensuring the reliability of Mycoplasma testing, the safety of cellular therapy products, and the integrity of scientific data.
The Impact of Prior Antimicrobial Treatment on Bacterial DNA Load and Detectability
The accurate and timely detection of bacterial pathogens is fundamental to effective patient management, particularly in serious infections such as sepsis. For decades, culture-based methods have served as the diagnostic gold standard, but they present significant limitations, including prolonged time-to-result and critical sensitivity to prior antimicrobial treatment [64] [27]. Molecular diagnostics, particularly polymerase chain reaction (PCR), offer a rapid alternative by detecting bacterial DNA rather than relying on bacterial viability and growth [64] [65].
A central question in molecular microbiology is how preceding antibiotic therapy influences the bacterial DNA load and its subsequent detectability by PCR. The prevailing concern has been that antibiotics, by causing bacterial cell death and disintegration, might reduce the total amount of intact, detectable DNA, potentially leading to false-negative results [64]. Conversely, some hypotheses suggest that the release of DNA from lysed cells could enhance detection. This guide objectively compares the performance of PCR-based methods against traditional culture in the context of prior antimicrobial treatment, synthesizing current experimental data to provide a clear framework for researchers and drug development professionals.
Comparative Performance Data: PCR vs. Culture Post-Antibiotic Treatment
Empirical studies consistently demonstrate that PCR-based methods maintain superior sensitivity compared to culture after antibiotic administration. The key differentiator is PCR's ability to detect DNA from non-viable bacteria, which culture cannot grow.
Table 1: Comparative Sensitivity of PCR and Culture After Antibiotic Treatment
| Pathogen / Experimental Model | Antibiotic Treatment | Culture Result | PCR-Based Method Result | Key Finding |
|---|---|---|---|---|
| S. aureus in human whole blood [64] | Vancomycin, Piperacillin (cell-wall active) | Growth inhibited; viable count reduced | Pathogen detection increased | Cell-wall active antibiotics improve DNA accessibility; no loss of DNA recovery. |
| S. aureus in human whole blood [64] | Ciprofloxacin, Clindamycin (non-cell-wall active) | Growth inhibited; viable count reduced | Pathogen detection unaffected | Concurrent antibiotic administration does not reduce PCR DNA recovery. |
| Clinical specimens for Genital Mycoplasmas [27] | Not specified (clinical samples) | 21 positive specimens | 28 positive specimens | PCR detected 11 additional positive specimens missed by culture. |
| E. coli in murine peritonitis model [66] | Ciprofloxacin (bactericidal) | Bacterial burden reduced (~2-4 log) | DNA detected, but associated with heightened TLR9-inflammatory response | Bactericidal antibiotics release pro-inflammatory bacterial DNA. |
The data reveals a critical consensus: while antibiotics rapidly reduce culturable bacterial units, the total bacterial DNA load remains detectable via PCR. In some cases, the action of cell-wall active antibiotics even enhances DNA detection, likely by breaking down cellular structures and making genomic DNA more accessible for extraction and amplification [64]. Furthermore, PCR demonstrates a higher clinical sensitivity, identifying pathogens in samples where culture fails due to prior treatment or difficult-to-grow organisms [27].
Detailed Experimental Protocols for Key Studies
To critically assess the data, understanding the underlying methodologies is essential. The following protocols are adapted from pivotal studies in the field.
Protocol 1: Investigating Antibiotic Impact on DNA Recovery from Blood
This protocol, derived from studies on S. aureus detection in whole blood, systematically evaluates the effect of different antibiotic classes [64].
- Bacterial Strains and Cultivation: Use reference (e.g., ATCC 29213) and clinical wild-type strains of S. aureus. Grow overnight cultures in lysogeny broth (LB).
- Whole Blood Preparation: Draw fresh human whole blood from healthy volunteers into sodium heparin or EDTA tubes. Buffer with HEPES and supplement with glucose and adenine.
- Antibiotic Pretreatment: Spike bacteria into supplemented whole blood and incubate for 4 hours to ensure logarithmic growth. Add antibiotics at clinically relevant concentrations (e.g., 15 μg/mL vancomycin, 1 μg/mL ciprofloxacin) and incubate for another 90 minutes.
- Pathogen Enrichment and DNA Extraction: Use a commercial pathogen enrichment kit (e.g., GINA Pathogen Enrichment).
- Selective Lysis: Add a hypotonic detergent buffer to lyse human blood cells selectively, leaving bacterial cells intact.
- Centrifugation: Pellet the intact bacteria.
- Bacterial Lysis and DNA Purification: Lyse the bacterial pellet by heating in an alkaline buffer and purify DNA using spin columns.
- DNA Quantification: Perform quantitative PCR (qPCR) to measure the recovery of pathogen DNA. Compare results to a non-antibiotic-treated control.
Protocol 2: Pre-Amplification for Ultra-Sensitive DNA Detection
For very low bacterial loads, a pre-amplification step can be incorporated to enhance sensitivity dramatically, as shown in studies on spiked whole blood [65].
- Sample Preparation: Filter 3 mL of spiked whole blood to concentrate bacterial cells.
- DNA Extraction: Isolate DNA using a method suitable for complex matrices like blood.
- Target-Specific Pre-Amplification:
- Primer Design: Use chimeric primers (Primer A) with a 3' end that is target-specific and a 5' end that is a universal sequence.
- First Amplification Cycles: Run initial cycles with only Primer A. The annealing and elongation occur at a low temperature (e.g., 43°C), generating amplicons with the universal sequence on both ends.
- Universal Amplification: Subsequent cycles use a universal primer (Primer B) that binds to the incorporated ends, enabling exponential, non-target-specific amplification of all tagged products.
- Downstream Detection: Analyze 2 μL of the pre-amplification product using a specific qPCR or microarray assay. This method can improve sensitivity by up to 100-fold, enabling detection down to 1 CFU/mL for some pathogens [65].
Mechanistic Insights: How Antibiotics Modulate DNA Availability and Immune Detection
The effect of antibiotics on bacterial DNA is not merely quantitative but also qualitative, influencing how DNA is presented to both diagnostic assays and the host immune system. The following diagram illustrates the divergent pathways triggered by bactericidal versus bacteriostatic antibiotics.
The diagram shows that bactericidal antibiotics directly kill bacteria, resulting in cell lysis and the release of substantial amounts of bacterial DNA. This DNA then becomes readily available for detection by PCR, which can explain the maintained or even improved sensitivity of molecular assays post-treatment [64]. However, this same process has a critical immunological consequence: the released DNA is sensed by the endosomal receptor Toll-like Receptor 9 (TLR9) in macrophages, triggering a potent pro-inflammatory response that can sometimes be detrimental in vivo [66]. In contrast, bacteriostatic antibiotics, which merely halt growth without large-scale killing, result in minimal DNA release. This leads to lower background inflammation but does not augment PCR sensitivity.
The Scientist's Toolkit: Essential Research Reagents and Solutions
Successful research in this field relies on a suite of specialized reagents and kits. The following table catalogues key solutions used in the featured experiments.
Table 2: Key Research Reagent Solutions for Investigating Bacterial DNA Detectability
| Reagent / Solution | Primary Function | Example Use Case & Notes |
|---|---|---|
| Pathogen Enrichment Kits (e.g., GINA Pathogen Enrichment) | Selective lysis of human cells & enrichment of intact bacteria via centrifugation. | Critical for removing PCR inhibitors (host DNA, hemoglobin) from blood samples prior to bacterial DNA extraction [64]. |
| DNA Extraction Kits (e.g., PowerWater DNA Isolation Kit, FastDNA SPIN Kit for Soil) | Isolation of total genomic DNA from complex sample matrices. | Kit choice significantly impacts DNA yield and observed microbial community; PowerWater is optimized for low-biomass aqueous samples [67]. |
| Pre-amplification Reagents (Chimeric Primers, Universal Primers) | Target-specific initial amplification followed by universal amplification to boost low-concentration DNA. | Enables detection sensitivity down to 1 CFU/mL of blood by amplifying specific markers above the detection threshold of downstream qPCR [65]. |
| qPCR/dPCR Master Mixes & Assays (Hydrolysis Probes, e.g., TaqMan) | Quantitative detection and quantification of specific bacterial DNA targets. | Probe-based assays offer superior specificity over intercalating dyes; dPCR is ideal for absolute quantification of rare targets [68] [69]. |
| Reference Materials & Controls (Genomic DNA from ATCC strains, Naïve host gDNA) | Assay development, calibration, and specificity testing. | Essential for validating that primers do not cross-react with host DNA and for establishing standard curves in qPCR [69]. |
The collective evidence firmly establishes that prior antimicrobial treatment does not diminish the detectability of bacterial DNA by PCR-based methods; in fact, it may enhance it. This positions molecular diagnostics as a superior tool in scenarios where patients have already received antibiotics, a common clinical reality. The critical advantage of PCR lies in its detachment from bacterial viability, allowing it to detect the "molecular shadow" of an infection even after antibiotics have eliminated culturable cells.
However, researchers must be cognizant of the biological context. The same bacterial DNA that facilitates robust PCR detection can also drive potentially damaging inflammatory responses via TLR9 signaling, a nuance highlighted by recent in vivo models [66]. Therefore, the choice between diagnostic methods is not merely technical but also interpretive. For pure detection sensitivity post-antibiotic treatment, PCR is unequivocally more reliable than culture. For a comprehensive understanding of the infection and host response, an integrated approach, considering both molecular and immunological data, is recommended. Future advancements in pre-analytical enrichment and pre-amplification will further solidify the role of PCR in the rapid and accurate diagnosis of bacterial infections.
Optimizing DNA Extraction and Amplification Conditions for Low-Level Contamination
The detection of low-level mycoplasma contamination represents a significant challenge in cell culture management and biopharmaceutical production. These minute, cell wall-less bacteria can persistently infect cell cultures, compromising research integrity and biotherapeutic safety without causing overt turbidity. Molecular techniques, particularly polymerase chain reaction (PCR), have emerged as powerful tools for mycoplasma detection due to their superior sensitivity and specificity compared to traditional methods like culture and staining techniques [8]. However, the accuracy of PCR-based detection is fundamentally dependent on the quality and quantity of extracted DNA, making optimization of pre-analytical procedures crucial for reliable results.
The integrity of DNA extracted from samples directly influences amplification efficiency, especially when targeting low-abundance contaminants. Effective DNA extraction must overcome numerous challenges, including the tough cytoplasmic membrane of mycoplasma, potential inhibitors in complex sample matrices, and the risk of DNA degradation during processing. This guide systematically compares DNA extraction and amplification methods, providing researchers with evidence-based protocols to enhance detection sensitivity for low-level mycoplasma contamination within the broader context of methodological comparisons between PCR and culture-based detection systems.
Comparative Analysis of DNA Extraction Method Performance
Key Performance Metrics for Extraction Methods
The efficiency of DNA extraction methods varies significantly based on sample type, microbial characteristics, and kit components. For mycoplasma detection, optimal methods must efficiently lyse the tough cytoplasmic membrane while preserving DNA integrity and minimizing co-extraction of PCR inhibitors. The table below summarizes the comparative performance of different extraction approaches based on yield, quality, and practical considerations:
Table 1: Performance Comparison of DNA Extraction Methods for Low-Level Contamination
| Extraction Method | Average DNA Yield | DNA Purity (A260/A280) | Inhibitor Removal | Processing Time | Cost per Sample |
|---|---|---|---|---|---|
| Silica Membrane Columns | Medium-High | 1.8-2.0 | High | 60-90 minutes | Medium |
| Magnetic Particle Methods | Medium | 1.7-2.0 | Very High | 45-60 minutes | Medium-High |
| Phenol-Chloroform | High | 1.6-1.9 | Medium | 120+ minutes | Low |
| Bead Beating + Kit | Very High | 1.7-2.0 | High | 90-120 minutes | Medium |
Recent studies demonstrate that optimized commercial kits consistently outperform traditional in-house methods for complex samples. Research on piggery wastewater surveillance revealed that optimized protocols using the QIAGEN QIAamp PowerFecal Pro DNA kit produced superior results for bacterial DNA recovery from challenging matrices compared to five alternative methods [70]. The incorporation of mechanical disruption through bead beating significantly enhanced DNA yield from tough-to-lyse bacteria, a finding directly applicable to mycoplasma detection where efficient lysis is critical.
Impact of Sample Type on Extraction Efficiency
Sample matrix composition dramatically affects DNA extraction efficiency. For mycoplasma detection in cell cultures, the presence of serum proteins, cellular debris, and media components can inhibit both lysis and downstream amplification. Clinical comparative studies have demonstrated that sample type selection alone can dramatically impact detection sensitivity. In a 2025 study comparing nasopharyngeal and oropharyngeal samples for Mycoplasma pneumoniae detection, oropharyngeal swabs showed significantly better sensitivity (96.2%) compared to nasopharyngeal samples (74.9%) using the same PCR methodology [12]. This highlights how sampling strategy must be considered alongside extraction optimization.
Complex matrices require specialized pre-processing steps to improve DNA recovery. For instance, forensic workflows successfully employ specialized pre-processing kits for challenging samples like bone and degraded tissues before extraction with systems like the Maxwell FSC [71]. Similarly, piggery wastewater research demonstrated that centrifugation-based sample preparation significantly improved DNA yield by concentrating microbial biomass while reducing dissolved inhibitors [70]. These principles apply directly to mycoplasma detection in complex cell culture supernatants or biological products.
Experimental Protocols for Optimized DNA Extraction and Amplification
Optimized DNA Extraction Protocol for Low-Biomass Samples
This protocol has been adapted from methodologies successfully employed for pathogen detection in complex matrices, incorporating specific optimizations for mycoplasma DNA recovery [72] [70]:
Reagents and Equipment:
- Lysis buffer with proteinase K (20 mg/mL)
- Commercially available DNA extraction kit (silica membrane or magnetic beads)
- Bead beating system with 0.1mm glass or ceramic beads
- Water bath or thermal mixer
- Microcentrifuge
- Nuclease-free water and consumables
Procedure:
- Sample Preparation: Concentrate 1-5 mL of cell culture supernatant by centrifugation at 14,000 × g for 30 minutes. Retain pellet and discard supernatant.
- Mechanical Lysis: Resuspend pellet in 200 μL of lysis buffer. Transfer to a bead beating tube containing 0.1mm glass beads. Process using a bead beater at maximum speed for 45 seconds, then place on ice for 1 minute. Repeat this cycle 3 times.
- Enzymatic Lysis: Add 20 μL of proteinase K (20 mg/mL) to the lysate. Mix thoroughly and incubate at 56°C for 1-2 hours with agitation at 300-400 rpm.
- DNA Purification: Follow manufacturer's instructions for your selected commercial DNA extraction kit with these modifications:
- Increase lysis incubation time to 10 minutes at 70°C
- Perform two wash steps with wash buffer with 5-minute incubation on ice before centrifugation
- Air-dry the column/magnetic beads for 10 minutes after final wash to ensure complete ethanol removal
- Elute in 50-100 μL of pre-warmed (70°C) nuclease-free water or elution buffer
- DNA Quantification and Storage: Quantify DNA using fluorometric methods (preferred) or spectrophotometry. Store at -20°C for short-term use or -80°C for long-term preservation.
Optimized PCR Amplification Protocol for Mycoplasma Detection
This protocol incorporates elements from highly sensitive detection methods validated in clinical studies [12] [73]:
Reaction Setup:
- 2X PCR master mix: 12.5 μL
- Forward primer (10 μM): 1 μL
- Reverse primer (10 μM): 1 μL
- Template DNA: 5 μL
- Nuclease-free water: to 25 μL total volume
Thermal Cycling Conditions:
- Initial denaturation: 95°C for 5 minutes
- 40-45 cycles of:
- Denaturation: 95°C for 30 seconds
- Annealing: 60-65°C for 45 seconds (optimize based on primer Tm)
- Extension: 72°C for 60 seconds
- Final extension: 72°C for 7 minutes
- Hold: 4°C indefinitely
Critical Optimization Steps:
- Include appropriate controls: no-template control, positive extraction control, and inhibition control
- For low-copy targets, increase cycle number to 45 while monitoring background signal
- Use touchdown PCR for difficult primers: decrease annealing temperature by 0.5°C every cycle for the first 10 cycles
- Add PCR enhancers such as betaine (1M final concentration) or DMSO (3-5%) for GC-rich targets
Visualization of Workflows and Method Selection
DNA Extraction and Amplification Workflow
Diagram 1: DNA Extraction and Amplification Workflow
Method Selection Decision Pathway
Diagram 2: Method Selection Decision Pathway
Research Reagent Solutions for Mycoplasma Detection
Table 2: Essential Research Reagents for Optimized Mycoplasma Detection
| Reagent/Category | Specific Examples | Function & Application | Performance Notes |
|---|---|---|---|
| DNA Extraction Kits | QIAamp PowerFecal Pro, DNeasy PowerLyzer PowerSoil, NucleoSpin Soil | Cellular lysis, inhibitor removal, DNA purification | PowerFecal Pro showed superior performance in complex matrices [70] |
| Automated Extraction Systems | Maxwell RSC 48, QIAcube Connect | Automated nucleic acid purification, reduced cross-contamination | Maxwell systems provide consistent yields from diverse forensic samples [71] |
| PCR Master Mixes | SureFast PLUS, Biopremier dtec-rt-PCR kits | Amplification with optimized buffer components | Commercial kits validated per ISO guidelines enhance reproducibility [74] |
| Sample Collection & Preservation | Universal Transport Medium (UTM), Extraction buffer with surfactants | Sample integrity maintenance, initial processing | Oropharyngeal swabs in specific buffer showed 96.2% sensitivity for M. pneumoniae [12] |
| Inhibitor Removal Additives | EDTA, Bovine Serum Albumin (BSA), PCR enhancers | Counteract PCR inhibitors, improve amplification | Bead beating with EDTA effectively accesses difficult samples while managing inhibition [72] |
Discussion: Integrating Extraction and Amplification Optimizations
The synergistic optimization of both DNA extraction and amplification conditions creates a comprehensive approach to low-level mycoplasma contamination detection. Evidence from clinical studies demonstrates that PCR-based methods generally offer superior sensitivity compared to culture-based approaches, particularly when optimized sample processing is implemented. In a direct comparison for Mycoplasma pneumoniae detection, PCR on oropharyngeal samples demonstrated 96.2% sensitivity compared to 74.9% for nasopharyngeal samples using the same platform, highlighting how sampling and processing jointly determine outcomes [12].
The integration of mechanical disruption methods, particularly bead beating, has proven essential for efficient lysis of tough bacterial membranes. As noted in optimization guides, "Bone is notoriously difficult… for obvious reasons—it's bone! Hard, mineralized, and resistant to lysis, bone presents a serious challenge to extract DNA from effectively. You often need a combo approach: chemical agents like EDTA to soften and demineralize, and powerful mechanical homogenization to physically break through the matrix" [72]. This principle applies equally to mycoplasma species with their tough cytoplasmic membranes.
Recent advances in PCR technology further enhance detection capabilities for low-level contamination. Methods like digital PCR (dPCR) provide absolute quantification without standard curves and demonstrate significantly higher sensitivity than traditional culture methods. In blood pathogen detection, dPCR identified 63 pathogenic strains across 42 positive specimens compared to only 6 strains via blood culture, with dramatically reduced detection time (4.8 hours versus 94.7 hours) [16]. Similarly, high-resolution melting (HRM) analysis offers rapid, closed-tube species differentiation based on melting temperature profiles, achieving strong concordance with sequencing results [73].
For laboratories implementing mycoplasma detection protocols, the systematic optimization of both pre-analytical (sample collection, DNA extraction) and analytical (amplification) steps is essential. The combination of appropriate sample processing, validated extraction methods, and sensitive amplification technologies creates a robust framework for detecting low-level mycoplasma contamination, ultimately supporting research integrity and product safety in biopharmaceutical applications.
Data-Driven Decisions: A Side-by-Side Comparison of Sensitivity, Speed, and Cost
Mycoplasma contamination represents a critical challenge in biological research and biopharmaceutical production, potentially compromising experimental results and the safety of biological products. The detection of these cell-wall-lacking prokaryotes is complicated by their small size (0.1–0.8 μm) and their inability to be visualized by standard microscopy or produce turbidity in culture media [6] [28]. This comparison guide provides an objective analysis of two primary detection methodologies: traditional culture-based techniques and modern polymerase chain reaction (PCR)-based methods. We present experimental data from multiple studies that directly compare the sensitivity, specificity, and practical applicability of these approaches, offering researchers evidence-based guidance for selecting appropriate mycoplasma detection strategies.
Quantitative Comparison of Detection Performance
Multiple studies have conducted head-to-head comparisons between PCR and culture methods, consistently demonstrating superior sensitivity for molecular approaches. The table below summarizes key performance metrics from these investigations:
Table 1: Direct comparison of mycoplasma detection methods across multiple studies
| Study Reference | Sample Type | Real-time PCR Sensitivity | Conventional PCR Sensitivity | Culture Method Sensitivity | Specificity |
|---|---|---|---|---|---|
| Kazemiha et al. (2015) [6] | 30 cell lines | 100% | 94.44% | 33.33% | 100% for both PCR methods |
| PCM et al. (1999) [75] | 92 pediatric patients with respiratory infection | 100% (all culture-positive cases detected) | Not reported | Reference standard | 100% |
| Recent Commercial Kits Study [28] | Cultured Mollicutes species | Varies by platform; some achieved ≤10 CFU/mL LOD | Not applicable | Requires 28-day incubation | Kit-dependent |
The stark contrast in detection capabilities is further evidenced by contamination rate findings. In one comprehensive analysis of 30 cell lines, mycoplasma contamination was detected in 60% of samples using real-time PCR, 56.66% using conventional PCR, but only 33.33% using microbial culture methods [6]. This demonstrates that culture-based approaches may fail to identify approximately half of all contaminated samples.
Experimental Protocols and Methodologies
Culture-Based Detection Protocol
The gold standard culture method follows pharmacopeia requirements (USP <63>, Ph. Eur. 2.6.7.) with this typical workflow [28]:
- Sample Collection: Approximately 15 mL of cell culture supernatant
- Inoculation: Simultaneous inoculation into both liquid broth and solid agar media
- Incubation: Extended incubation at 35–37°C for 28 days
- Subculture: Multiple blind passages to enhance detection of slow-growing species
- Visualization: Microscopic examination for characteristic "fried-egg" colonies on solid media
This method targets viable organisms through metabolic activity and colony formation, but its effectiveness is limited to cultivable species under artificial conditions [6].
PCR-Based Detection Protocols
PCR methods target conserved genomic regions across Mycoplasma species, primarily the 16S ribosomal RNA gene [6] [35]. A typical protocol includes:
- Sample Preparation: DNA extraction from 500 μL cell culture supernatant using commercial kits
- Primer Design: Selection of ultra-conserved regions covering >90% of Mycoplasma species [35]
- Amplification: Real-time PCR with genus-specific primers (e.g., 425 bp product targeting 16S rRNA)
- Detection: Fluorescence-based monitoring of amplification curves
- Analysis: Threshold-based determination of positive samples
Advanced digital droplet PCR (ddPCR) platforms have demonstrated even greater sensitivity, with limits of detection as low as 2.9 copies/reaction compared to 10.8 copies/reaction for conventional real-time PCR [39].
Table 2: Key research reagents for mycoplasma detection
| Reagent Category | Specific Examples | Application/Function |
|---|---|---|
| PCR Kits | PromoKine Mycoplasma Test Kit, MycoSEQ (Life Technologies), VenorGEM qOneStep (Minerva Biolabs) | Commercial kits for standardized detection |
| Culture Media | Hayflick's broth/agar, SP4 medium with arginine | Supports growth of fastidious mycoplasma species |
| Staining Reagents | DAPI, Hoechst dye | DNA staining for fluorescent microscopy detection |
| Enzymatic Assays | MycoAlert (Lonza) | Detects mycoplasma enzyme activity |
| Positive Controls | M. fermentans, M. orale, M. arginini, A. laidlawii | Validation of detection assay performance |
Visualizing Method Workflows
The fundamental differences between culture and PCR methodologies are illustrated in the following workflow diagrams:
Mycoplasma Detection Method Workflows
Detection Rate Comparison Across Studies
Mechanisms Underlying Performance Differences
The significant disparity in detection sensitivity between PCR and culture methods stems from fundamental biological and technical factors:
Biological Limitations of Culture Methods
- Viability Dependency: Culture methods detect only viable, cultivable organisms, missing non-viable or non-cultivable species [76]
- Growth Requirements: Many mycoplasma species have fastidious growth needs that artificial media cannot replicate [6]
- Slow Growth: The extended generation time of mycoplasmas necessitates prolonged incubation (up to 28 days) [28]
- Species Selectivity: Culture media favor certain species while inhibiting others, creating detection biases [6]
Technical Advantages of PCR Methods
- Direct DNA Detection: PCR identifies mycoplasma-specific genetic sequences regardless of viability [35]
- Conserved Target Regions: Amplification of ultra-conserved 16S rRNA regions enables broad species detection [35]
- Rapid Turnaround: Most PCR methods provide results within hours versus weeks for culture [77]
- Automation Potential: High-throughput processing enables screening of multiple samples simultaneously [28]
The comprehensive evidence from multiple direct-comparison studies demonstrates the unequivocal superiority of PCR-based methods over traditional culture techniques for mycoplasma detection. The consistent finding of 100% sensitivity for PCR versus 33–40% for culture methods across different sample types and experimental conditions highlights the critical need for molecular approaches in settings where detection reliability is paramount. While culture methods maintain value for specific applications requiring viability determination, PCR-based techniques offer researchers and biopharmaceutical professionals significantly enhanced detection capabilities, faster results, and broader species coverage—all essential factors for ensuring the integrity of biological products and the validity of scientific research.
Mycoplasma contamination poses a significant threat to biotherapeutic product quality and patient safety, making rigorous testing essential for cell and gene therapy manufacturers [78]. For decades, the 28-day culture-based method has been the compendial standard for mycoplasma detection. However, this prolonged timeframe is incompatible with the short shelf life of many advanced therapies, where patients are waiting for infusion and time is critical to effective treatment [79] [80].
The emergence of rapid polymerase chain reaction (PCR) technologies, capable of delivering results in approximately one hour, presents a transformative alternative [81]. This guide objectively compares these two methodologies, providing experimental data on their performance, and explores the paradigm shift occurring in quality control testing for biopharmaceutical manufacturing.
Methodological Comparison: PCR vs. Culture
The fundamental differences between rapid PCR and traditional culture methods extend far beyond the obvious discrepancy in time-to-result.
Traditional Culture-Based Method
The culture-based method, as specified in pharmacopoeias like USP <63>, requires a 28-day agar and broth cultivation process [82] [78]. This method relies on the growth of mycoplasma in complex media, followed by visual observation for characteristic signs of growth. The lengthy process is necessary because mycoplasmas are slow-growing and lack a cell wall, making them difficult to detect through conventional microbiological techniques [78]. The method's requirement for specialized expertise and its inability to provide timely results for short-lived biologics has been a long-standing challenge for the industry [82].
Rapid PCR-Based Detection
PCR-based methods detect mycoplasma through amplification of specific genetic targets, such as the 16S rRNA region, which is conserved across mycoplasma species [30]. Modern automated systems, like the BIOFIRE FILMARRAY 2.0 Industry System, integrate extraction, amplification, and detection into a closed "molecular lab in a pouch" [81]. This system can detect over 130 species of mycoplasma and provides sample-to-answer results in approximately one hour with just two minutes of hands-on time, requiring no specialized molecular biology expertise [79] [81] [80].
Table 1: Core Characteristics of Mycoplasma Detection Methods
| Feature | Culture-Based Method | Rapid PCR Method |
|---|---|---|
| Time to Result | 28 days [82] [78] | ~1 hour [79] [81] |
| Hands-on Time | Significant | ~2 minutes [81] |
| Expertise Required | Specialized training | Minimal technical training [79] |
| Throughput | Low | Medium to High (multiple instruments can be linked [79]) |
| Detection Principle | Microbial growth in culture media | Nucleic acid amplification [30] |
| Key Advantage | Compendial standard; does not require specialized equipment | Speed, ease of use, and suitability for products with short shelf-lives [80] |
Performance and Sensitivity Data
While culture has been the historical gold standard, recent studies demonstrate that modern PCR assays can meet or exceed its sensitivity while providing results dramatically faster.
Sensitivity and Limit of Detection (LOD)
Regulatory guidelines for mycoplasma testing require a method capable of detecting ≤10 CFU/mL to replace culture-based standards [82]. A comprehensive 2023 study compared five commercial molecular assays and found that the BIOFIRE Mycoplasma assay was the most sensitive, followed by the MycoSEQ and MycoTOOL kits, which were comparable [82]. The BIOFIRE system achieves this high sensitivity through specialized sample preparation protocols, including a 10 mL release test protocol that concentrates the sample via centrifugation to reach a validated LOD of ≤10 CFU/mL [79] [80]. Another kit, the MycoScope, offers high-sensitivity detection of less than 5 mycoplasma genomes per microliter of sample [30].
Comparative Detection Capabilities
The same 2023 study demonstrated that not all commercial PCR assays were capable of meeting the stringent ≤10 CFU/mL LOD, highlighting the importance of rigorous validation when selecting a rapid method [82]. Furthermore, studies have shown that PCR-based detection is not hindered by contaminating organisms that often complicate culture-based methods, providing more reliable detection in complex samples [83].
Table 2: Quantitative Performance Comparison of Representative Methods
| Method | Claimed LOD | Time to Result | Mycoplasma Species Detected |
|---|---|---|---|
| Culture (USP <63>) | ≤10 CFU/mL (Required standard [82]) | ≥28 days [82] [78] | Broad range, but limited by cultivability |
| BIOFIRE Mycoplasma | ≤10 CFU/mL (validated [79] [80]) | ~1 hour [81] | >130 species [79] |
| MycoSEQ Plus | ≤10 CFU/mL (validated [78]) | Hours [78] | >200 species (in silico analysis [78]) |
| MycoScope | <5 genomes/μL [30] | <3 hours [30] | All species common in cell culture [30] |
Experimental Protocols in Practice
The implementation of rapid PCR methods requires specific protocols to ensure they meet regulatory sensitivity requirements for product release testing.
Protocol for Rapid 1-Hour PCR Testing
For the BIOFIRE system, two standard protocols have been developed to align with regulatory guidelines [80]:
- 10 mL Single Centrifugation Protocol: This protocol is suitable for final product release testing. It uses a larger sample volume concentrated by centrifugation to achieve the required ≤10 CFU/mL sensitivity [79] [80].
- Low Volume Protocol (~1.7 mL): Designed for manufacturers with limited test article availability, this protocol also achieves the necessary sensitivity for release testing [80].
In both protocols, the sample is loaded into a pre-hydrated, closed pouch that contains all necessary reagents for automated PCR. The system then performs extraction, amplification, and detection automatically, providing a "Mycoplasma Detected" or "Mycoplasma Not Detected" result in about one hour [79] [81].
Protocol for 28-Day Culture-Based Testing
The compendial culture method is a dual-media process involving both agar and broth cultures, incubated for 28 days [82] [78]. Samples are inoculated into the media and observed for growth throughout the incubation period. This process requires significant laboratory space, dedicated equipment, and trained microbiologists to interpret results, which are based on visual observation of characteristic mycoplasma colonies or broth turbidity [78].
Diagram 1: A simplified workflow comparing the rapid PCR and traditional culture-based methods for mycoplasma detection, highlighting the significant difference in process complexity and time.
The Scientist's Toolkit: Essential Research Reagents
Implementing robust mycoplasma testing requires specific reagents and systems. The following table details key solutions for effective detection.
Table 3: Key Research Reagent Solutions for Mycoplasma Detection
| Product/System | Function | Key Features |
|---|---|---|
| BIOFIRE FILMARRAY 2.0 Industry System [79] [81] | Automated nucleic acid extraction, amplification, and detection. | Closed-pouch system; detects >130 species; ~1 hr turnaround; 21 CFR Part 11-ready software. |
| MycoSEQ Mycoplasma Detection System [82] [78] | Real-time PCR-based detection kit. | Covers >200 species; LOD of ≤10 CFU/mL; uses TaqMan chemistry. |
| MycoScope Mycoplasma PCR Detection Kit [30] | PCR-based detection kit for use with existing lab equipment. | Detects <5 genomes/μL; targets 16S rRNA; results in <3 hours; no DNA extraction needed. |
| Vero E6 Cells [84] | Cell line for viral culture, used in some mycoplasma culture methods. | Used in specialized culture protocols requiring eukaryotic cells. |
| Mycoplasma Agar and Broth Media [78] | Culture media for the compendial 28-day growth method. | Complex media formulations required for the growth of fastidious mycoplasma. |
The data and comparisons presented in this guide clearly illustrate a significant shift in mycoplasma testing paradigms. Rapid PCR methods, capable of delivering results in about one hour, are no longer just research tools but are mature, validated technologies ready to address the critical needs of modern biopharmaceutical manufacturing, particularly for cell and gene therapies [81] [80].
While the 28-day culture method remains a compendial standard, its practical limitations are undeniable. The emergence of PCR systems that meet the stringent ≤10 CFU/mL sensitivity requirement provides manufacturers with a powerful tool to enhance patient safety, reduce costs, and streamline production workflows without compromising quality [82] [78]. The choice for researchers and drug development professionals is not merely about selecting a test, but about embracing a more efficient, data-driven approach to quality control that aligns with the accelerated pace of advanced therapeutic development.
The demonstration of a Limit of Detection (LOD) below 10 Colony Forming Units per milliliter (CFU/mL) has long been a critical benchmark for microbiological assays. For years, traditional culture methods were the primary means of achieving this sensitivity. However, advanced molecular techniques, particularly Polymerase Chain Reaction (PCR) and its derivatives, now consistently demonstrate the capability to meet and even exceed this stringent detection threshold. This guide objectively compares the experimental data and methodologies that validate PCR's performance against traditional culture-based alternatives, with a specific focus on mycoplasma detection in biopharmaceutical and clinical contexts.
Quantitative LOD Comparison: PCR vs. Culture Methods
Extensive research and regulatory updates confirm that PCR-based methods achieve LODs on par with the gold standard culture methods, with the added advantage of significantly faster results.
Table 1: Summary of LOD and Performance Metrics for Various Detection Methods
| Detection Method | Application Context | Reported LOD | Key Performance Advantage | Turnaround Time | Citation |
|---|---|---|---|---|---|
| qPCR (Mycoplasma) | Cell Culture/Biologics | <10 CFU / <100 Genomic Copies (GC)/mL | Recognized as equivalent to culture by European Pharmacopoeia | Hours | [85] [86] [47] |
| Digital PCR (dPCR) | Bloodstream Infections | 25.5 - 439,900 copies/mL | Higher sensitivity and wider detection range than blood culture | 3-6 hours | [16] |
| Gram Probe RT-PCR | Pleural Fluid (Bacterial) | 10 CFU/mL | Higher positive rate (17.78%) vs. culture (11.67%) | A few hours | [87] |
| Culture (Gold Standard) | Mycoplasma Testing | 10 CFU/mL | High specificity | 3-5 days (bacteria) to several weeks (mycoplasma) | [16] [47] |
The data reveals a clear trend: while culture methods maintain a defined LOD of 10 CFU/mL, modern PCR techniques reliably meet this benchmark and offer transformative improvements in speed. The recent update to the European Pharmacopoeia chapter 2.6.7 formally recognizes this equivalence for mycoplasma testing, stating that nucleic acid amplification techniques (NAT) like PCR are "fully equivalent to traditional culture assays" with a defined LOD of "10 CFU/ml or less than 100 genomic copies per ml (100 GC/ml)" [85] [86].
Experimental Protocols for Validating PCR's LOD
The claim of PCR achieving an LOD of <10 CFU is not made lightly; it is backed by rigorous, standardized experimental validation. The following protocols detail how this sensitivity is demonstrated in scientific studies.
Protocol 1: Mycoplasma qPCR Assay Validation
This protocol, based on a study describing a highly sensitive Taqman-based real-time PCR assay, outlines the process for validating a PCR method for mycoplasma detection in cell cultures [47].
- Objective: To validate a qPCR assay for detecting mycoplasma in cell culture with an LOD of less than 10 CFU.
- Sample Preparation: The assay is designed to test cell culture medium directly, eliminating the need for DNA extraction, which streamlines the process and reduces potential losses [47].
- Assay Design: A Taqman-based real-time PCR format is used. This employs sequence-specific fluorescent probes, ensuring high specificity and reducing false positives compared to dye-based methods [47] [88].
- Sensitivity (LOD) Determination: The assay is challenged with serial dilutions of the most frequently encountered mycoplasma contaminants. The LOD is established as the lowest concentration at which the PCR can consistently detect the target, which was demonstrated to be below 10 CFU [47].
- Specificity Testing: The assay's primers and probes are tested against a panel of mycoplasma species to ensure broad detection coverage and against host cell DNA (e.g., from CHO or human cells) to confirm no cross-reactivity [88].
Protocol 2: Digital PCR (dPCR) for Blood Pathogen Detection
This protocol is derived from a comparative clinical study evaluating dPCR against blood culture, highlighting the procedural differences that lead to dPCR's superior sensitivity [16].
- Objective: To compare the sensitivity and detection range of dPCR with routine blood culture for identifying pathogens in blood samples.
- Study Design: A retrospective analysis of 149 patients with suspected infections. Blood samples from each patient were subjected to both dPCR and blood culture simultaneously [16].
- Sample Processing for dPCR:
- Plasma Separation: Whole blood samples are collected in EDTA tubes and centrifuged to separate plasma.
- DNA Extraction: Plasma DNA is extracted using a commercial nucleic acid purification kit and an automated system.
- dPCR Analysis: The extracted DNA is added to a pre-designed multi-channel dPCR panel. The mixture undergoes droplet generation, PCR amplification, and droplet analysis using a scanner across six fluorescence channels to identify multiple microorganisms simultaneously [16].
- Sample Processing for Blood Culture:
- Incubation: Two sets of blood culture samples (aerobic and anaerobic) are incubated in an automated system like BacT/ALERT 3D.
- Subculture and Identification: Positive samples are gram-stained, subcultured on agar plates, and identified using a system like Vitek 2 Compact [16].
- Outcome Measurement: The study compared the number of positive specimens, species of pathogens identified, and the time to result for both methods. dPCR detected 63 pathogenic strains in 42 specimens, far exceeding the 6 strains found by culture, with a mean turnaround time of 4.8 hours versus 94.7 hours for culture [16].
The experimental workflow for a typical dPCR-based pathogen detection study, as outlined in Protocol 2, can be visualized as follows:
The Scientist's Toolkit: Essential Research Reagent Solutions
Successfully implementing a PCR-based detection assay with a low LOD requires specific, high-quality reagents and controls.
Table 2: Key Reagents for PCR-based Mycoplasma Detection
| Reagent / Solution | Critical Function | Example & Regulatory Context |
|---|---|---|
| qPCR Kit | Provides primers, probes, and master mix for specific target amplification. | Venor Mycoplasma qPCR: A ready-to-use, EP 2.6.7-compliant kit that uses reverse transcriptase PCR to detect both DNA and RNA, covering >130 mollicutes [85] [86]. |
| Quantitative Reference Standards | Enables absolute quantification and verification of assay sensitivity in a specific product matrix. | 100GC Mycoplasma Standards: Provide traceable, non-infectious reference material with 100 genomic copies/vial, crucial for the inhibitory substances test required by EP 2.6.7 [85] [86]. |
| Internal Control | Rules out false negatives caused by PCR inhibitors in the sample. | Included in commercial kits like Venor Mycoplasma qPCR; its amplification confirms the reaction is functioning correctly [86] [47]. |
| External Positive Control | Verifies that the entire testing process, from sample processing to detection, is working optimally. | Defined controls with a concentration close to the LOD (e.g., 10 CFU or 100 GC) are mandated by the updated EP 2.6.7 [86]. |
The collective evidence from regulatory updates and peer-reviewed research solidifies the position of PCR-based methods as a viable and often superior alternative to culture for achieving an LOD of <10 CFU. While culture remains the historical gold standard, PCR offers equivalent sensitivity with the transformative benefits of a drastically reduced turnaround time—from weeks or days to mere hours. This acceleration is critical in settings like biopharmaceutical manufacturing and clinical diagnostics, where rapid results directly impact product release, patient treatment decisions, and ultimately, public health outcomes. The ongoing harmonization of global pharmacopoeias around PCR testing marks a significant milestone, empowering researchers and drug development professionals to adopt these rapid, reliable, and sensitive molecular techniques with confidence.
Mycoplasma contamination poses a significant threat to the integrity of cell cultures, potentially compromising research data and biopharmaceutical products. Analytical specificity—a method's ability to accurately detect target organisms without cross-reacting with non-target species—is paramount for reliable mycoplasma detection. This guide objectively compares the specificity and species coverage of various mycoplasma detection methods, with particular emphasis on evaluating cross-reactivity profiles. The analysis is framed within the broader context of comparing PCR versus culture methods, providing researchers and drug development professionals with experimental data to inform their selection of detection methodologies.
Comparative Analysis of Detection Method Specificity
Methodologies and Cross-Reactivity Profiles
The specificity of mycoplasma detection methods varies significantly based on their underlying mechanisms. Culture methods, historically considered the gold standard, rely on the growth of mycoplasma colonies on specialized agar media, typically showing high specificity for cultivable species but potentially missing fastidious strains that grow poorly in vitro [29]. DNA staining techniques (e.g., DAPI or Hoechst) utilize fluorescent dyes that bind to A-T rich regions in DNA, providing direct visualization of contamination but potentially staining non-mycoplasma bacterial DNA or cellular debris, leading to false positives [50] [89]. Enzymatic methods detect specific mycoplasma enzymes like adenosine phosphorylase, but cross-reactivity with other bacteria (e.g., Bacillus subtilis, Escherichia coli) has been reported, reducing specificity [29].
Immunoassays (ELISA and immunostaining) employ antibodies against mycoplasma antigens or specific enzymes such as elongation factor TU (EF-TU). While offering species-specific identification, their range is limited by antibody specificity, potentially missing contaminating species not targeted by the antibodies used [89]. PCR-based methods amplify conserved genomic regions (typically 16S rRNA or 16S-23S rRNA ISR) using genus-specific primers. Their specificity depends on primer design, with potential cross-reactivity if primers anneal to non-target sequences, though well-designed assays can minimize this risk [35] [29].
Species Coverage Performance
Table 1: Species Coverage of Mycoplasma Detection Methods
| Detection Method | Species Coverage | Coverage Limitations | Key Experimental Findings |
|---|---|---|---|
| Microbial Culture | Cultivable species only | Limited to species that grow on specific media; misses fastidious strains | Detected only 33.33% of contaminations in 30 cell lines compared to molecular methods [6] |
| DNA Staining (DAPI/Hoechst) | Broad in principle but limited by sensitivity | Cannot differentiate mycoplasma species; may stain other bacterial DNA [50] | Detected 46.66% of contaminations in comparative study; subjective interpretation [6] |
| Enzymatic (MycoAlert) | Multiple common species | Limited by enzyme presence across all species; cross-reactivity with some bacteria [29] | Detected 53.33% of contaminations; performance varies by contaminating species [6] |
| Conventional PCR | 90%+ with well-designed primers | Primers must target truly conserved regions across Mollicutes class | 94.44% specificity; 100% PPV; covers >60 species with optimized primers [6] [90] |
| Real-time PCR | 90%+ with well-designed primers | Similar to conventional PCR but with probe-based additional specificity | 100% specificity, sensitivity, and accuracy; detected 60% of contaminations in 30 cell lines [6] |
| Universal PCR (16S-23S ISR) | 143 Mycoplasma species validated | Minimal off-target binding with carefully validated primers | 100% specificity for 16 species tested; no cross-reactivity with common cell culture contaminants [29] |
Experimental Data on Cross-Reactivity
Table 2: Cross-Reactivity Testing of PCR-Based Methods
| Detection Method | Non-Target Organisms Tested | Cross-Reactivity Results | Experimental Conditions |
|---|---|---|---|
| Universal PCR (16S-23S ISR) | Bacteria: S. pullorum, E. coli, S. aureus, P. fragi; Fungus: C. albicans; Viruses: PRV, PCV2, PPV | No cross-reactivity observed with any non-target organisms [29] | Testing conducted with purified nucleic acids from each organism using optimized primer sets |
| Four-Primer PCR Assay | Non-mycoplasma bacteria: Brachyspira, Micrococcoides, Nitriliruptor, Oligella, Peptococcus | 97% on-target specificity; 3% off-target binding at strain level [35] | Bioinformatics analysis of 25,796 database entries followed by experimental validation |
| Internally-Controlled qPCR | Mammalian cells; common bacterial contaminants | No cross-reactivity with mammalian DNA or bacterial contaminants [47] | Assay designed with species-specific probes and internal controls |
| ATCC PCR Service | Bacterial and mammalian genomic DNA | No cross-reactivity observed; specific for Mycoplasma, Acholeplasma, Spiroplasma, Ureaplasma [90] | Proprietary primer sets targeting 16S rRNA with touchdown PCR regimen |
Experimental Protocols for Specificity Validation
Primer Design for Maximum Species Coverage
A 2023 study established a robust protocol for designing primers with comprehensive species coverage. Researchers identified highly conserved 16S rRNA mycoplasma-specific regions using the NCBI Bacterial 16S Ribosomal RNA RefSeq Targeted Loci Project (Accession PRJNA33175) containing 25,796 entries at the time of analysis. This bioinformatics approach identified one fitting primer pair for global mycoplasma detection, matching 198 out of 216 mycoplasma species (92% coverage) [35].
The experimental validation involved a four-primer PCR format that simultaneously amplified a eukaryotic control sequence (105 bp) and mycoplasma target sequence (166-191 bp). This design allowed verification of successful PCR in all samples while specifically detecting mycoplasma DNA in contaminated samples. The primers were validated against 16 cell lines, successfully identifying contaminated samples without false positives [35].
Cross-Reactivity Testing Protocol
To validate assay specificity, researchers typically test against a panel of non-target organisms that might be present in experimental samples. A comprehensive 2025 study evaluated their universal PCR, ERA, and qPCR assays against 16 different Mycoplasma species, 4 representative bacteria (S. pullorum, E. coli, S. aureus, P. fragi), 1 common fungal contaminant (C. albicans), and 3 relevant viruses (PRV, PCV2, PPV) [29].
The testing protocol involved:
- Extraction of purified nucleic acids from each non-target organism
- Amplification using the same experimental conditions optimized for mycoplasma detection
- Analysis of amplification products through gel electrophoresis (PCR), fluorescence detection (qPCR), or lateral flow strips (ERA)
- Verification of no detectable signal when compared to positive mycoplasma controls
This rigorous validation confirmed 100% specificity with no cross-reactivity observed with any non-target organisms [29].
Limit of Detection (LOD) Determination
The four-primer PCR assay established its limit of detection using serial dilutions of M. orale DNA mixed with a constant amount of mycoplasma-free genomic DNA from twelve cell lines to simulate actual testing conditions. The detection limit was determined to be 6.3 pg or 8.21×10³ genomic copies, demonstrating high sensitivity within a complex background of eukaryotic DNA [35].
Another study evaluating universal PCR, ERA, and qPCR assays reported detection limits of 10¹ copies for PCR, 10⁰ copies for ERA, and an impressive 10⁻¹ copies for qPCR when testing against a Spiroplasma 16S-23S spacer fragment plasmid [29].
PCR-Based Method Workflow
Research Reagent Solutions
Table 3: Essential Research Reagents for Mycoplasma Detection Experiments
| Reagent/Chemical | Function | Application Examples |
|---|---|---|
| Primers (16S rRNA targeted) | Amplify conserved mycoplasma sequences | Universal primer pairs targeting 16S rRNA for broad species coverage [35] [90] |
| Primers (16S-23S ISR targeted) | Amplify intergenic spacer region with high discriminatory power | Improved species coverage across 143 Mycoplasma species [29] |
| DNA Polymerase | Catalyzes DNA synthesis during PCR | Thermostable polymerase used in proprietary PCR mixes [90] |
| dNTPs | Building blocks for DNA amplification | Component of PCR master mixes for mycoplasma DNA amplification [90] |
| Fluorescent Probes (TaqMan) | Enable real-time detection of amplified DNA | qPCR assays with species-specific probes [47] |
| Agarose | Matrix for electrophoretic separation of DNA fragments | Visualization of PCR products (e.g., 500bp band for MycoScope kit) [30] |
| DNA Staining Dyes (DAPI/Hoechst) | Fluorescent staining of mycoplasma DNA | Direct visualization of contamination by fluorescence microscopy [50] [89] |
| Culture Media (PPLO Broth) | Supports growth of mycoplasma for culture methods | Microbial culture as gold standard detection [6] [50] |
| Antibodies (Anti-EF-TU) | Specific recognition of mycoplasma antigens | Immunostaining for mycoplasma detection [89] |
| Nucleotide Analogs | Enzymatic incorporation for enhanced detection | Modified nucleotides for nick translation in immunofluorescence assays [50] |
The analytical specificity of mycoplasma detection methods varies significantly, with PCR-based approaches demonstrating superior species coverage and minimal cross-reactivity compared to traditional methods. Well-designed PCR assays targeting conserved regions like 16S rRNA or 16S-23S ISR can achieve coverage exceeding 90% of known mycoplasma species with virtually no cross-reactivity to common cell culture contaminants. The experimental data presented enables researchers to select appropriate detection methods based on their specific requirements for species coverage, specificity, and analytical rigor, ultimately supporting the integrity of cell-based research and biopharmaceutical production.
Integrating Rapid PCR Methods into Regulatory and Compendial Frameworks (USP, Ph. Eur.)
The detection of Mycoplasma contamination in biopharmaceuticals, cell therapies, and clinical diagnostics has historically relied on 28-day culture-based methods that pose significant challenges for products with short shelf lives [28]. The recent harmonization of major pharmacopeias, including the United States Pharmacopeia (USP), European Pharmacopoeia (Ph. Eur.), and Japanese Pharmacopoeia (JP), has established a new regulatory framework that recognizes nucleic acid amplification techniques (NAT) as equivalent to traditional culture methods [86] [91]. This paradigm shift enables laboratories to implement rapid PCR-based testing while maintaining compliance with global regulatory standards. The revised Ph. Eur. chapter 2.6.7 (Edition 12.2), which becomes mandatory in April 2026, specifically defines sensitivity requirements for NAT methods at ≤ 10 CFU/mL or < 100 genomic copies/mL, introducing genomic copies as a standardized unit optimized for molecular methods [86] [91]. This guide provides a comprehensive comparison of PCR versus culture methods for mycoplasma detection sensitivity, offering experimental data and protocols to support implementation within these updated regulatory frameworks.
Regulatory Framework Harmonization
Global Pharmacopeia Alignment
The recent regulatory updates represent a significant achievement in global harmonization, creating consistent requirements across international pharmacopeias. This alignment reduces the validation burden for laboratories operating in multiple regulatory jurisdictions and facilitates more straightforward method implementation.
Table 1: Comparative Analysis of Updated Pharmacopeia Requirements for Mycoplasma Testing
| Parameter | Ph. Eur. 2.6.7 (12.2) | USP <63> & <77> | JP 18 G3 |
|---|---|---|---|
| NAT Status | Equivalent to culture methods | Alternative method requiring validation | Equivalent to culture methods |
| Required LOD | ≤ 10 CFU/mL or < 100 GC/mL | ≤ 10 CFU/mL | ≤ 10 CFU/mL or < 100 GC/mL |
| Reference Unit | Genomic copies (GC) | CFU | Genomic copies (GC) |
| Detection Scope | Culturable & non-culturable mycoplasmas | Culturable mycoplasmas | Culturable & non-culturable mycoplasmas |
| Sample Type | Cells & supernatant | Not specified | Cells & supernatant |
| Validation Requirement | In user's product matrix | Comparability to culture | In user's product matrix |
| Internal Control | Mandatory | Not specified | Mandatory |
| Effective Date | April 2026 | Ongoing | Ongoing |
Key Changes in Ph. Eur. 2.6.7
The revised Ph. Eur. chapter 2.6.7 introduces several critical updates that laboratories must address [86] [91]:
- NAT-Specific Framework: NAT methods now have a dedicated regulatory framework and are considered equivalent to culture-based methods, not merely alternatives.
- Standardized Units: The introduction of genomic copies (GC) as a standardized unit optimized for NAT methods, with a defined GC:CFU ratio below 10 to ensure traceability and consistency.
- Expanded Detection Scope: Requirements to detect both culturable and non-culturable mycoplasmas, addressing a significant limitation of culture-based methods.
- Comprehensive Sampling: Testing should include both cells and supernatant whenever possible, as mycoplasmas can adhere to or reside within cells.
- Enhanced Validation: Mandatory method validation in the user's own product matrix, even when using commercially validated kits, to confirm absence of inhibitory substances and ensure assay sensitivity under real conditions.
- Control Requirements: Implementation of mandatory controls including an internal control to rule out inhibition, an external positive control with defined GC or CFU content close to the limit of detection, and a negative control without target sequence.
Comparative Sensitivity Analysis: PCR vs. Culture Methods
Clinical Detection Performance
Recent clinical studies have demonstrated significant advantages of PCR-based methods for Mycoplasma pneumoniae detection. A 2025 study comparing sampling methods for PCR detection in children with respiratory symptoms revealed substantially different sensitivity profiles [12].
Table 2: Clinical Sensitivity Comparison of Mycoplasma pneumoniae Detection Methods
| Testing Method | Sample Type | Sensitivity | Specificity | Reference Standard |
|---|---|---|---|---|
| Multiplex PCR | Nasopharyngeal | 74.9% (67.9–81.0%) | 99.2% (97.0–99.9%) | Oropharyngeal RT-PCR |
| Smart Gene Myco | Oropharyngeal | 96.2% (92.3–98.4%) | 100.0% (98.5–100.0%) | Oropharyngeal RT-PCR |
| RT-PCR | Nasopharyngeal | 32.2% positive | N/A | Oropharyngeal RT-PCR |
| RT-PCR | Oropharyngeal | 43.4% positive | N/A | Oropharyngeal RT-PCR |
This comprehensive study involving 422 participants found that the sensitivity of MP detection was significantly better in oropharyngeal samples than in nasopharyngeal samples (96.2% vs. 74.9%), supporting the use of oropharyngeal swabs for optimal detection [12]. The research also identified a negative correlation between fluoroquinolone use and oropharyngeal DNA loads, highlighting the impact of prior antimicrobial treatment on detection sensitivity.
Commercial Assay Performance Comparison
A systematic evaluation of five commercial molecular assays provides critical performance data for laboratories selecting appropriate testing platforms [28]. This proof-of-principle analysis tested assays against 10 cultured Mollicutes species at four log-fold dilutions (1,000 CFU/mL to 1 CFU/mL) in biological duplicates.
Table 3: Analytical Performance of Commercial Mycoplasma Detection Assays
| Commercial Assay | Limit of Detection | Repeatability | Meeting ≤10 CFU/mL LOD | TIL Matrix Interference |
|---|---|---|---|---|
| Biofire Mycoplasma | Most sensitive | Acceptable | Yes | No interference |
| MycoSEQ | Comparable to MycoTOOL | Acceptable | Yes | No interference |
| MycoTOOL | Comparable to MycoSEQ | Acceptable | Yes | No interference |
| VenorGEM qOneStep | Less sensitive | Acceptable | No | No interference |
| ATCC Universal | Less sensitive | Acceptable | No | No interference |
The study demonstrated that not all commercial assays were capable of meeting the ≤10 CFU/mL LOD required to replace culture-based methods according to European and Japanese pharmacopeia standards [28]. The Biofire Mycoplasma assay showed the highest sensitivity, followed by MycoSEQ and MycoTOOL which demonstrated comparable performance. Importantly, no assay interference was observed when testing in the presence of tumor infiltrating lymphocytes (TILs), supporting utility in cellular therapy products.
Experimental Protocols and Methodologies
Clinical Sampling and PCR Protocol
The high-sensitivity clinical detection protocol from the 2025 study illustrates optimal sampling and processing techniques [12]:
Sample Collection:
- Oropharyngeal samples collected using Nipro sponge swab TYPE L
- Nasopharyngeal samples collected using UTM Nasopharyngeal Sample Collection Kit
- All samples collected by trained physicians (board-certified pediatricians or resident physicians)
- Paired samples from each individual collected simultaneously by the same physician
Storage Conditions:
- Samples frozen within one day after examination
- Stored at -80°C (hospital) or -18°C (clinic)
- Transported with freezer box (no thawing observed)
DNA Extraction and PCR:
- DNA extracted using QIAamp DNA Mini Kit
- RT-PCR conducted using AriaMx Real-Time PCR System
- Target: CARDS toxin gene
- Primers: Forward 5'-TTTGGTAGCTGGTTACGGGAAT-3', Reverse 5'-GGTCGCCACGAATTTCATATAAG-3'
- Probe: 5'-FAM-TGTACCAGAGCACCCCAGAAGGGCT-BHQ1-3'
- Quantification: Synthetic DNA standards (106 to 101 copies/test)
- Quality control: Positive controls (MP reference strain M129) and negative extraction controls
Commercial Assay Evaluation Protocol
The comparative study of commercial assays utilized rigorous methodology to ensure reproducible results [28]:
Bacterial Strains and Growth:
- Ten Mollicutes type strains selected based on pharmacopeia standards and reported cell culture contaminants
- Cultured in Hayflick's broth and agar or SP4 medium with arginine as needed
- Incubation at 37°C with 5% CO₂ for most species, 30°C for Spiroplasma ixodetis
- No more than 15 passages from initial inoculation as per USP <63> requirements
LOD Determination:
- Log-fold dilutions prepared (1,000 CFU/mL to 1 CFU/mL)
- Biological duplicates with three replicates per condition (n=6)
- Testing performed by two different analysts
- Additional testing in presence of tumor infiltrating lymphocytes (TILs)
Validation Criteria:
- Ability to detect ≤10 CFU/mL as required by EP and JP
- Repeatability across replicates and analysts
- Absence of matrix inhibition with TILs
Implementation Workflow and Signaling Pathways
The transition from culture-based to PCR-based mycoplasma detection requires a systematic approach to ensure regulatory compliance while maintaining detection sensitivity. The following workflow diagram illustrates the critical decision points and validation requirements.
Essential Research Reagent Solutions
Successful implementation of PCR-based mycoplasma detection requires specific reagents and reference materials that meet updated pharmacopeial standards. The following table details essential components for establishing a compliant testing framework.
Table 4: Essential Research Reagents for Mycoplasma Detection
| Reagent Category | Specific Products | Function & Application | Regulatory Compliance |
|---|---|---|---|
| qPCR Assays | Venor Mycoplasma qPCR | Detection of >130 mollicute species via RT-qPCR | EP 2.6.7, USP <63>, JP 18 G3 |
| Gel-Based PCR | Venor Mycoplasma gelPCR | Conventional PCR with gel detection | EP 2.6.7, USP <63>, JP 18 G3 |
| Digital PCR | Venor Mycoplasma dPCR | Absolute quantification without standard curve | EP 2.6.7, USP <63>, JP 18 G3 |
| Quantitative Standards | 100GC Mycoplasma Standards | Precise quantitative reference material (100 GC/vial) | EP 2.6.7 GC requirement |
| CFU-Based Standards | 10CFU & 100CFU Standards | Culture-based reference materials | Bridging NAT and culture methods |
| Extraction Systems | Venor Mycoplasma Extraction Kit | Nucleic acid extraction using magnetic bead technology | Validated with Venor assays |
| Internal Controls | Manufacturer-specific | Inhibition control for false-negative identification | EP 2.6.7 mandatory requirement |
The Venor Mycoplasma qPCR represents a fully-aligned solution with the updated regulatory framework, featuring reverse transcriptase-based detection of both DNA and RNA to enhance analytical sensitivity, and coverage of more than 130 mollicute species including all pharmacopoeial reference strains [86] [91]. The 100GC Mycoplasma Standards provide traceable quantitative reference material essential for verifying assay sensitivity in product-specific matrices and fulfilling the new requirement for external positive controls with defined concentration [91].
The integration of rapid PCR methods into regulatory frameworks for mycoplasma detection represents a significant advancement in testing capabilities for biopharmaceuticals, cell therapies, and clinical diagnostics. The harmonization of USP, Ph. Eur., and JP requirements establishes a consistent global standard that enables laboratories to implement sensitive, rapid molecular methods while maintaining regulatory compliance. Experimental data demonstrates that properly validated PCR methods can exceed the sensitivity of traditional culture-based approaches while providing results in hours rather than weeks. The critical success factors for implementation include careful assay selection based on demonstrated LOD performance, comprehensive validation in product-specific matrices, and robust quality control incorporating mandatory internal and external controls as specified in the updated Ph. Eur. chapter 2.6.7. As the April 2026 effective date approaches, laboratories should proactively transition to compliant methods and reagents to ensure uninterrupted testing operations and maintain the highest standards of product quality and patient safety.
Conclusion
The comparative analysis unequivocally demonstrates that PCR-based methods offer a superior alternative to traditional culture for mycoplasma detection, providing exceptional sensitivity (up to 100%), specificity, and drastically reduced turnaround times—from weeks to hours. This paradigm shift is particularly crucial for the advancing fields of biologics, cell, and gene therapies, where product shelf-lives are short and sample volumes are limited. The future of mycoplasma testing lies in the widespread adoption and regulatory acceptance of rapid, automated, and highly sensitive molecular methods like real-time PCR and fully integrated NAT systems. Embracing these technologies is essential for enhancing product safety, streamlining manufacturing processes, and accelerating the delivery of innovative therapies to patients. Future directions will likely involve greater integration of artificial intelligence for data analysis and the continued evolution of point-of-care molecular diagnostics.
References
- 1. Prevention and Detection of Mycoplasma Contamination in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3584481/]
- 2. Mycoplasma Contamination Risks in Biomanufacturing - Mabion [https://www.mabion.eu/science-hub/articles/mycoplasma-contamination-in-biologics-manufacturing/]
- 3. Mycoplasma tests and the Impact of Contamination - Yeasen [https://www.yeasenbio.com/blogs/cell/concept-of-mycoplasma-and-the-impact-of-contamination]
- 4. Sensitivity of biochemical test in comparison with other ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4158010/]
- 5. How Does Mycoplasma Contamination Affect Cell Culture? [https://www.eppendorf.com/us-en/lab-academy/life-science/cell-biology/how-does-mycoplasma-contamination-affect-cell-culture/]
- 6. Real-time PCR assay is superior to other methods for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4960155/]
- 7. Assessing mycoplasma contamination of cell cultures by ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0172358]
- 8. Mycoplasma Detection Kits Compared: PCR vs. ELISA vs. ... [https://synapse.patsnap.com/article/mycoplasma-detection-kits-compared-pcr-vs-elisa-vs-dapi-staining]
- 9. Direct qPCR is a sensitive approach to detect Mycoplasma ... [https://bmcresnotes.biomedcentral.com/articles/10.1186/s13104-019-4763-5]
- 10. Hybrid PCR-based Mycoplasma Detection for Product- ... [https://www.sciencedirect.com/science/article/abs/pii/S1465324925005158]
- 11. Culture-based viability PCR: strategies to harness ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12281229/]
- 12. PCR sensitivity for Mycoplasma pneumoniae detection in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12345179/]
- 13. Integrating Metagenomic and Culture-Based Techniques to ... [https://www.mdpi.com/2304-8158/14/3/352]
- 14. Nucleic acid amplification: Alternative methods of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3831736/]
- 15. Which is a Better Lab Test, PCR or Culture? [https://americanpharmacogenomicsassociation.com/li-newsletter/pcr-or-culture/]
- 16. Comparative analysis between digital PCR and blood ... [https://www.frontiersin.org/journals/medicine/articles/10.3389/fmed.2025.1615409/full]
- 17. Comparison of PCR with Other Nucleic Acid Amplification ... [https://www.mybiosource.com/learn/comparison-of-pcr-with-other-nucleic-acid-amplification-techniques/]
- 18. Nucleic acid amplification tests in digital microfluidics [https://www.nature.com/articles/s41378-025-00977-5]
- 19. Detection of Mycoplasma pneumoniae by Real-Time ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC193797/]
- 20. Comparative analysis between digital PCR and blood ... [https://pubmed.ncbi.nlm.nih.gov/41090121/]
- 21. Nucleic acid amplification techniques for the detection of ... [https://www.sciencedirect.com/science/article/pii/S1201971221006949]
- 22. Recent Advances and Applications in Integrating Nucleic ... [https://www.sciencedirect.com/science/article/abs/pii/S0165993625003243]
- 23. The Genera Mycoplasma and Ureaplasma [https://veteriankey.com/the-genera-mycoplasma-and-ureaplasma/]
- 24. Assay Validation for Rapid Detection of Mycoplasma ... [https://www.bioprocessintl.com/assays/assay-validation-for-rapid-detection-of-mycoplasma-contamination]
- 25. Performance of PCR-based and Bioluminescent assays for ... [https://www.sciencedirect.com/science/article/abs/pii/S0167701215300439]
- 26. Multicenter evaluation of fast multiplex PCR for detection ... [https://www.frontiersin.org/journals/cellular-and-infection-microbiology/articles/10.3389/fcimb.2025.1643991/full]
- 27. Comparison of Multiplex PCR Assay with Culture for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC387538/]
- 28. Comparison of Five Commercial Molecular Assays for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9945500/]
- 29. Development and validation of universal PCR, basic ERA ... [https://www.sciencedirect.com/science/article/abs/pii/S0882401025003948]
- 30. Sensitive Detection of Cell Culture Mycoplasma Infection [https://www.rapidmicrobiology.com/news/sensitive-detection-of-cell-culture-mycoplasma-infection]
- 31. The Clinical Validity and Utility of PCR Compared to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12029264/]
- 32. Implementation of a Laboratory-Developed Test for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12300490/]
- 33. A roadmap to high-speed polymerase chain reaction (PCR) [https://www.sciencedirect.com/science/article/pii/S0956566323007728]
- 34. PRIMARY CARE | Limitations of Culture [https://microgendx.com/primary-care-limitations-of-culture/]
- 35. A PCR protocol to establish standards for routine mycoplasma testing... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10192841/]
- 36. Specifity and sensitivity of polymerase chain reaction (PCR ... [https://www.sciencedirect.com/science/article/pii/002217599390279G]
- 37. PCR Amplification | An Introduction to PCR Methods [https://www.promega.com/resources/guides/nucleic-acid-analysis/pcr-amplification/]
- 38. PCR-based diagnostics for infectious diseases [https://pmc.ncbi.nlm.nih.gov/articles/PMC7106425/]
- 39. Absolute quantification of Mycoplasma pneumoniae in ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2023.1177273/full]
- 40. Development and clinical validation of a novel multiplex ... [https://www.nature.com/articles/s41598-025-16434-2]
- 41. The Clinical Validity and Utility of PCR Compared to ... [https://www.mdpi.com/2076-2607/13/4/949]
- 42. Clinical utility of PCR compared to conventional culture ... [https://www.sciencedirect.com/science/article/pii/S0732889324004255]
- 43. Comparative Performance of Digital PCR and Real-Time RT ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12474457/]
- 44. PDA 2025: How Automation Assures Product Quality for ... [https://www.biopharminternational.com/view/pda-2025-how-automation-assures-product-quality-for-next-gen-biopharma]
- 45. Experts See 17 Laboratory Trends Dominating 2025 [https://clpmag.com/diagnostic-technologies/experts-see-17-laboratory-trends-dominating-2025/]
- 46. Best PCR Systems of 2025 : Top Picks for Research, Clinical & Industry... [https://www.labx.com/resources/best-pcr-systems-of-2025-top-picks-for-research-clinical-industry-labs/4817]
- 47. A highly sensitive internally-controlled real-time PCR assay ... [https://www.sciencedirect.com/science/article/pii/S104510561930137X]
- 48. Comparison of different detection methods for Mycoplasma ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7893926/]
- 49. Digital PCR and Real-time PCR(qPCR) 2025-2033 Analysis [https://www.marketreportanalytics.com/reports/digital-pcr-and-real-time-pcrqpcr-314646]
- 50. A New Sensitive Method for the Detection of Mycoplasmas ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6952905/]
- 51. Mycoplasma Release Testing Cell and Gene Therapy ... [https://www.biomerieux.com/corp/en/education/resource-hub/pharma-quality-control/scientific-posters/mycoplasma-release-testing-cell-and-gene-therapy-product-samples.html]
- 52. Mycoplasma testing regulatory guidance and strategies for ... [https://www.insights.bio/cell-and-gene-therapy-insights/webinars/332/Mycoplasma-testing-regulatory-guidance-and-strategies-for-cGMP-cell-and-gene-therapy-manufacturing]
- 53. Combined oropharyngeal nasal (ON) swabs for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12502526/]
- 54. Comparison of sample types and diagnostic methods for in ... [https://www.sciencedirect.com/science/article/abs/pii/S0378113516304825]
- 55. Comparison of culture and a multiplex probe PCR for ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0173422]
- 56. Mycoplasma detection in cell cultures: a comparison of four ... [https://pubmed.ncbi.nlm.nih.gov/11204859/]
- 57. The Essential Role of PCR and PCR Panel Size in ... [https://www.mdpi.com/1422-0067/24/18/14269]
- 58. A practical approach for gmp-compliant validation of real ... [https://pubmed.ncbi.nlm.nih.gov/34362616/]
- 59. Polymerase chain reaction inhibitors [https://en.wikipedia.org/wiki/Polymerase_chain_reaction_inhibitors]
- 60. Improving sensitivity of single tube nested PCR to detect ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7002839/]
- 61. PCR inhibition in qPCR, dPCR and MPS—mechanisms and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7072044/]
- 62. Mycoplasma Testing Implementation and Validation [https://www.thermofisher.com/us/en/home/life-science/bioproduction/contaminant-and-impurity-testing/mycoplasma-detection/compliance.html]
- 63. PCR Inhibitors: Effects on PCR Reactions and Removal ... [https://bioclone.net/technology-pcr-inhibitors/?srsltid=AfmBOooS9FiVloS5MDq-2wCIFAxlgf2sd-Cq5hLTMPV2GRVQXKVnRHZX]
- 64. Influence of antibiotic treatment on the detection of S. ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6683330/]
- 65. Enhancing Bacterial DNA Detection in Blood Samples ... [https://www.mdpi.com/2079-6382/13/2/161]
- 66. Bactericidal antibiotic treatment induces damaging ... [https://www.nature.com/articles/s41467-024-54497-3]
- 67. Investigation of Detection Limits and the Influence of DNA ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2018.02140/full]
- 68. Polymerase Chain Reaction Chips for Biomarker Discovery ... [https://www.mdpi.com/2072-666X/16/3/243]
- 69. Recommendations for Method Development and ... [https://link.springer.com/article/10.1208/s12248-023-00880-9]
- 70. Optimization of DNA extraction methods from pig farm ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12551764/]
- 71. Two Decades of Maxwell - PID Volume 12 Fall 2025 [https://promega.foleon.com/profiles-in-dna/pid-volume-12-fall-2025/two-decades-of-maxwell]
- 72. How to Work with Challenging or Limited Genomic ... [https://blog.omni-inc.com/blog/how-to-work-with-challenging-or-limited-genomic-samples-updated-for-2025]
- 73. Optimization of the real-time PCR platform method using ... [https://www.nature.com/articles/s41598-025-16972-9]
- 74. Development of real-time PCR methods for quality control ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12058689/]
- 75. Comparison of PCR, Culture, and Serological Tests for ... - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC84154/]
- 76. Cross comparison of rapid mycoplasma detection platforms [https://www.sciencedirect.com/science/article/abs/pii/S1045105609001742]
- 77. How Pure is Your Cell Culture Broth? Comparing ... [https://bitesizebio.com/9071/how-pure-is-your-cell-culture-broth-comparing-mycoplasma-detection-kits/]
- 78. Mycoplasma Testing for Biopharmaceutical Manufacturing [https://www.thermofisher.com/blog/life-in-the-lab/revolutionizing-mycoplasma-testing-for-biopharmaceutical-manufacturing/]
- 79. EASY 1 HOUR MYCOPLASMA DETECTION IN CELL ... [https://www.biomerieux-industry.com/pharma-quality-control/resources/white-papers/easy-1-hour-mycoplasma-detection-cell-therapy-samples]
- 80. AUTOMATED NUCLEIC ACID AMPLIFICATION ASSAY ... [https://www.sciencedirect.com/science/article/pii/S1465324925001185]
- 81. 1-Hour In-House Mycoplasma Testing for Cell & Gene Therapies [https://www.biomerieux.com/corp/en/our-offer/industry-products/biofire-mycoplasma-cell-and-gene-therapy.html]
- 82. Comparison of Five Commercial Molecular Assays ... - PubMed [https://pubmed.ncbi.nlm.nih.gov/36688643/]
- 83. Comparison of PCR versus Culture for Detection ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3811193/]
- 84. Clinical comparison and agreement of PCR, antigen, and viral ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9300048/]
- 85. Contamination Control Compass Edition #10/2025 [https://news.minerva-biolabs.com/newsletter/2025/10/]
- 86. New EP 2.6.7 Revision: Compliant Mycoplasma Testing [https://www.rapidmicrobiology.com/news/ep-2-6-7-revision-compliant-mycoplasma-testing-for-biopharma-and-atmps]
- 87. Rapid and Sensitive Identification of Bacterial Infection ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4784618/]
- 88. Simple, specific, rapid, and pharmacopoeia-compliant ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12441703/]
- 89. Why Test for Mycoplasma in My Cell Culture? [https://www.goldbio.com/blogs/articles/testing-for-mycoplasma?srsltid=AfmBOopc17NUzX_NHvCB2_61F6tfN46TbzEy3qbf4k_zf0iu-s9VF2s-]
- 90. Mycoplasma Testing Service [https://www.atcc.org/services/cell-authentication/mycoplasma-testing]
- 91. Update EP 2.6.7 – What will Change for You [https://news.minerva-biolabs.com/informative-articles/2025/11]