Microglia Isolation Showdown: Magnetic Bead vs. Percoll Gradient for Purity, Yield, and Phenotype
This article provides a comprehensive comparative analysis of two predominant microglia isolation techniques—immunomagnetic bead separation and Percoll density gradient centrifugation.
Microglia Isolation Showdown: Magnetic Bead vs. Percoll Gradient for Purity, Yield, and Phenotype
Abstract
This article provides a comprehensive comparative analysis of two predominant microglia isolation techniques—immunomagnetic bead separation and Percoll density gradient centrifugation. Tailored for researchers and drug development professionals, it delves into the foundational principles, step-by-step protocols, and critical factors influencing cellular yield, purity, and the preservation of in vivo phenotypes. We synthesize recent evidence to guide method selection, troubleshoot common pitfalls, and discuss the implications of isolation strategy on downstream applications, from transcriptomics to high-throughput drug screening, ultimately aiming to enhance reproducibility and translational relevance in microglial research.
Understanding Microglia Isolation: Core Principles and Critical Challenges
The Critical Need for Pure Microglia Populations in Neurological Research
Microglia, the resident immune cells of the central nervous system, play critical roles in brain development, homeostasis, and neurological disorders. Their functions range from synaptic pruning and clearance of cellular debris to the production of inflammatory mediators in response to injury or disease. The isolation of highly pure microglia populations is therefore essential for conducting precise and controlled experiments that allow researchers to focus on specific cellular functions and gene expression without interference from other cell types. Results can be inconsistent or misleading without proper isolation due to mixed cell populations, potentially compromising the validity of research findings, particularly in the study of complex neurological conditions such as Alzheimer's disease, Parkinson's disease, and other neurodegenerative disorders [1] [2] [3].
This guide provides an objective comparison of two fundamental microglia isolation techniques—magnetic bead isolation and Percoll gradient centrifugation—evaluating their performance based on purity, yield, viability, and technical requirements to inform researchers' methodological selections.
Why Microglia Purity Matters in Research
Microglia represent approximately 5-20% of the glial cell population in the central nervous system [4]. When studying their unique functions in health and disease, contamination by other cell types—particularly astrocytes, oligodendrocytes, and pericytes—can significantly confound experimental results. Primary microglia maintain functionality and structural integrity without genetic modification, unlike immortalized cell lines which may accumulate mutations over time and fail to exhibit adult phenotype and behavior [1].
Species and age considerations are particularly important in microglia research. Significant differences exist between rodent and human microglia in their biochemical responses to pharmacological substances [3] [5]. Furthermore, aged microglia exhibit distinct characteristics compared to younger cells, including elevated baseline inflammation, differential gene transcription, and altered phagocytic ability—a phenomenon known as "inflammaging" [4]. These differences highlight the importance of selecting appropriate source materials and isolation methods that yield pure populations representative of the physiological state being studied.
Isolation Methodologies: Direct Comparison
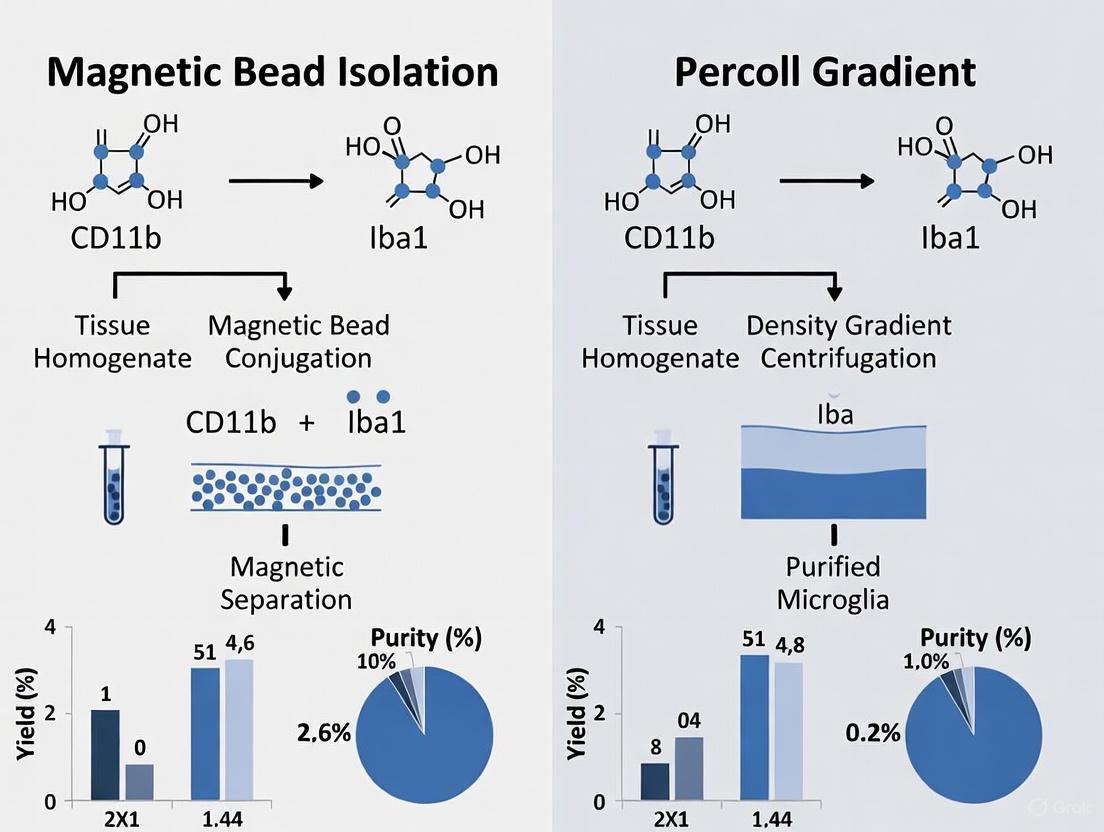
Magnetic Bead Isolation (MACS)
Magnetic bead separation uses antibodies conjugated to magnetic particles that recognize specific cell surface markers (e.g., CD11b for microglia). When exposed to a magnetic field, labeled cells are retained while unlabeled cells pass through [6] [7].
Protocol Overview:
- Prepare single-cell suspension from brain tissue through enzymatic digestion and mechanical disruption [1] [6]
- Incubate cell suspension with anti-CD11b magnetic beads (10 μL per 1×10⁷ cells) at 4°C for 15 minutes [6]
- Wash cells to remove unbound beads by centrifugation at 300 × g for 10 minutes at 4°C [6]
- Apply cell suspension to MS column through 70 μm cell strainer [6]
- Wash column 3 times with 500 μL MACS buffer [6]
- Elute CD11b⁺ cells by removing column from magnetic field and flushing with buffer [6]
This method can be adapted for both positive selection (directly isolating microglia) and negative selection (depleting non-target cells) approaches [1] [8]. When isolating multiple cell types from the same tissue, a tandem protocol can be implemented using CD11b for microglia, ACSA-2 for astrocytes, and a non-neuronal antibody cocktail for neuronal purification [1].
Percoll Gradient Centrifugation
Percoll gradient centrifugation is a density-based separation technique that exploits differences in the buoyant densities of various brain cell types. Percoll consists of colloidal silica particles coated with polyvinylpyrrolidone (PVP), creating an inert, low-osmolality medium ideal for separating fragile cells without damage [1] [9].
Protocol Overview:
- Create discontinuous or continuous density gradients using isotonic Percoll solutions [9]
- Layer the single-cell brain suspension on top of the gradient
- Centrifuge at appropriate speed and duration (varies by protocol)
- Collect microglia from the appropriate density interface (typically at lower density regions)
- Wash cells to remove residual Percoll particles [1]
Percoll's unique properties include low osmolality (<20 mOsm at density ~1.13 g/ml) and minimal interaction with cell surfaces, making it particularly suitable for separating osmotically sensitive cells like microglia while maintaining high viability [9].
Performance Comparison: Experimental Data
The table below summarizes key performance metrics for magnetic bead and Percoll gradient isolation methods based on published experimental data:
Table 1: Performance Comparison of Microglia Isolation Methods
| Parameter | Magnetic Bead Isolation | Percoll Gradient |
|---|---|---|
| Purity | >95% for human microglia [3]; ~97.2% for iPSC-derived microglia [2] | High purity, but may have slight myeloid cell contamination [10] |
| Viability | >85% [10] | Maintains high viability due to low osmolality and inert properties [9] |
| Yield | ~1×10⁶ viable cells per adult mouse brain [4] | Varies by protocol; generally high recovery rates |
| Processing Time | ~5 days for complete protocol [6]; faster than FACS for single or multiple samples [10] | Rapid processing; avoids time-consuming immunolabeling steps [1] |
| Technical Complexity | Moderate; requires specific antibodies and magnetic equipment [7] | Moderate; requires gradient preparation and centrifugation optimization |
| Cost | Higher due to antibody and specialized column requirements [1] | Lower; avoids expensive immunoreagents [1] |
| Cell Function Post-Isolation | Maintains phagocytic ability, cytokine secretion, and inflammatory responses [3] | Maintains normal cellular functions; Percoll has minimal effect on biological activities [9] |
| Special Considerations | Potential for slight myeloid cell contamination [10]; antibody-dependent specificity [7] | Circumvents enzymatic digestion which might affect cell viability [1] |
Technical Workflow Comparison
The following diagram illustrates the key steps and decision points in both isolation workflows:
Research Reagent Solutions
The table below outlines essential reagents and materials required for implementing these microglia isolation techniques:
Table 2: Essential Research Reagents for Microglia Isolation
| Reagent/Material | Function/Purpose | Example Specifications |
|---|---|---|
| Anti-CD11b Magnetic Beads | Immunomagnetic labeling of microglia via surface antigen recognition | Species-specific antibodies conjugated to iron oxide particles [6] [7] |
| Percoll Solution | Density gradient medium for buoyant density-based separation | Colloidal silica coated with PVP, 10-30 nm particle size [9] |
| Enzymatic Digestion Cocktail | Tissue dissociation to create single-cell suspensions | Papain (2.5 U/mL) + DNase (10 U/mL) [5] or trypsin-based formulations [1] |
| MACS Buffer | Maintenance of cell viability during magnetic separation | Typically PBS-based with EDTA and fetal bovine serum [6] |
| Cell Culture Media | Support cell survival and growth post-isolation | Serum-supplemented DMEM or specialized microglial media with growth factors [4] [3] |
| Separation Columns | Immobilization of magnetically labeled cells | MS or LS columns compatible with magnetic separators [6] [8] |
| Centrifugation Equipment | Density gradient separation and post-processing washes | Capable of precise speed control (e.g., 300 × g for washing) [6] |
Both magnetic bead isolation and Percoll gradient centrifugation offer effective pathways to obtain high-purity microglia populations, yet each presents distinct advantages and limitations. Magnetic bead separation provides exceptional purity (>95%) and is ideal for studies requiring highly specific microglia populations, particularly when combined with well-validated surface markers like CD11b. Conversely, Percoll gradient separation offers a cost-effective alternative that avoids potential antibody-induced cell activation and maintains high cell viability through its gentle, inert properties.
The selection between these methods should be guided by specific research requirements, including desired purity levels, available budget, technical expertise, and downstream applications. Studies focusing on transcriptomic profiling or sensitive functional assays may benefit from the superior purity of magnetic bead isolation, while larger-scale preparations or experiments seeking to minimize antibody exposure might prefer density gradient approaches. As microglia research continues to evolve, methodological selections should align with the specific physiological or pathological contexts being studied, particularly considering the growing recognition of microglial diversity across brain regions, developmental stages, and disease states.
Immunomagnetic bead separation stands as a pivotal technique in modern life science research, enabling the precise isolation of specific cell populations from complex mixtures. This technology hinges on a elegantly simple yet powerful core principle: antibody-driven specificity. The process utilizes superparamagnetic beads conjugated with highly specific antibodies that target unique surface markers on cells of interest. When exposed to a magnetic field, these labeled cells are retained while unbound cells are washed away, yielding a purified population. This article explores the fundamental operation of this technology and provides a direct comparison with the traditional Percoll gradient method, with a specific focus on microglia isolation for neurological research. The critical distinction lies in the separation mechanism—immunomagnetic separation relies on biological affinity through antibody-antigen recognition, whereas density gradient methods like Percoll exploit physical properties such as cell size and density [1] [11].
Table 1: Core Mechanism Comparison of Cell Separation Techniques
| Feature | Immunomagnetic Bead Separation | Percoll Gradient Separation |
|---|---|---|
| Separation Principle | Biological affinity (antibody-antigen binding) | Physical properties (cell density and size) |
| Basis for Separation | Expression of specific surface markers (e.g., CD11b) | Natural density differences between cell types |
| Key Reagents | Antibody-coated magnetic beads | Density gradient medium (Percoll) |
| Specificity | High (targets predefined cell populations) | Lower (separates broad cell classes) |
| Complexity | Higher (requires specific antibodies and protocols) | Lower (protocols are generally simpler) |
Performance Comparison: Microglia Isolation
When isolating microglia from neural tissue, the choice between immunomagnetic separation and Percoll gradients involves significant trade-offs in purity, yield, viability, and procedural requirements. The following comparative data, synthesized from multiple experimental studies, provides a clear performance overview to guide methodological selection.
Table 2: Performance Comparison for Microglia Isolation from Mouse Brain
| Performance Metric | Immunomagnetic Bead Separation | Percoll Gradient Separation | Supporting Experimental Context |
|---|---|---|---|
| Purity (CD11b+ cells) | High (>95%) [12] | Variable; requires flow cytometry refinement [13] | Purity confirmed via flow cytometry with microglia-specific markers [12]. |
| Cell Yield | Lower (protocol-dependent) [13] | Higher [13] [11] | One study reported yields of ~0.5-1.0 million cells per mouse brain using a optimized Percoll protocol [13]. |
| Cell Viability | High (>95% with Percoll myelin removal) [12] | High [11] | Viability highly dependent on myelin removal step; 30% Percoll proved superior to sucrose [12]. |
| Procedure Duration | ~4-5 hours (can be longer with MACS kits) [13] | ~3-4 hours [11] | Some commercial immunomagnetic kits can take up to 12 hours [13]. |
| Key Advantage | High specificity and purity; targets specific subtypes (e.g., CD11b+) | Simplicity; avoids expensive antibodies; preserves naive cell state | Magnetic separation allows sequential isolation of multiple cell types from one sample [1]. |
| Key Limitation | Higher cost; potential for antibody-induced activation | Lower specificity; can co-isolate other myeloid cells | Isolated microglia may start to change morphology shortly after purification [1]. |
Experimental Protocols in Practice
Detailed Protocol: Immunomagnetic Separation of Microglia
The following established protocol yields highly pure, viable microglia with phenotypes that accurately reflect their in vivo state [12].
- Brain Harvest and Dissociation: Following perfusion with ice-cold PBS, dissect the brain and remove the meninges. Chop the tissue into small pieces and subject it to enzymatic digestion using a commercial neural tissue dissociation kit (e.g., Miltenyi Biotec) containing a blend of enzymes like papain and DNase. This is performed at 37°C for approximately 35 minutes with mechanical dissociation, for instance using a GentleMACS Octo Dissociator with heaters [13] [12].
- Myelin Removal: Resuspend the resulting single-cell suspension in 30% Percoll solution and centrifuge at 700 × g for 10 minutes. This critical step pellets the cells while leaving the less dense myelin fragments in the supernatant, which is discarded. The cell pellet is then washed with HBSS [12].
- Immunomagnetic Labeling and Separation: Incubate the cells with a PE-conjugated anti-CD11b antibody for 10 minutes at 4°C. After washing, add anti-PE magnetic beads and incubate for another 15 minutes. Pass the cell suspension through a magnetic column placed in a strong magnetic field. The CD11b+ microglia are retained in the column, while the negative fraction (effluent) flows through. Removing the column from the magnetic field allows for the elution of the highly purified microglia [12].
Detailed Protocol: Percoll Gradient Separation of Microglia
This density-based method is valued for its simplicity and effectiveness, particularly for isolating microglia from both young and adult mice [13] [11].
- Tissue Dissociation: Perform mechanical and enzymatic dissociation of the brain tissue as described in the immunomagnetic protocol to obtain a single-cell suspension [13].
- Gradient Preparation and Centrifugation: Prepare two distinct Percoll solutions: a 70% Percoll stock and an isotonic working solution (e.g., 30% SIP). Carefully layer the 30% Percoll/cell suspension over a pre-formed 70% Percoll cushion in a centrifuge tube. Centrifuge the gradient at a specified speed and time (e.g., without brakes to avoid disruption). This step separates cells into distinct layers based on their buoyant densities [13].
- Microglia Harvesting: After centrifugation, microglia typically form a dense band at the interface between the two Percoll layers. Carefully aspirate this band using a Pasteur pipette. Transfer the harvested microglia to a new tube, wash with buffer to remove residual Percoll, and resuspend in culture medium [13].
Visualizing the Separation Mechanisms
The diagrams below illustrate the fundamental operational principles of each separation method.
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of these isolation techniques relies on a suite of specific reagents and tools.
Table 3: Essential Reagents for Microglia Isolation Protocols
| Reagent / Tool | Function / Role | Example Specifics |
|---|---|---|
| Anti-CD11b Antibodies | Primary marker for microglial cell surface; used for immunomagnetic selection or purity confirmation. | Conjugated to PE for use with anti-PE magnetic beads [12]. |
| Protein A/G Magnetic Beads | High-binding-capacity beads for attaching antibodies via Fc region. | Protein A or G beads show superior binding capacity compared to secondary antibody-coated beads [14]. |
| Percoll | Density gradient medium for separating cells based on buoyant density. | Used to create discontinuous gradients (e.g., 30% over 70%) [13]. |
| Neural Tissue Dissociation Kit | Enzyme blend for digesting extracellular matrix to create single-cell suspensions. | Commercial kits (e.g., Miltenyi) containing papain, DNase, etc. [13] [12]. |
| Magnetic Separation Columns | Placeholder for magnetic separation of bead-bound cells from the sample. | MS or LS columns (Miltenyi Biotec) used with a magnetic separator [12]. |
| Cell Culture Medium + Supplements | Maintains cell viability and health during and after isolation. | DMEM/F-12 with GlutaMAX, FBS, antibiotics, and sometimes M-CSF/GM-CSF [11]. |
Application in Experimental Research
The choice between immunomagnetic and Percoll separation extends beyond simple protocol selection; it fundamentally influences experimental design and data interpretation in microglia research.
Immunomagnetic separation is indispensable for studies requiring high cellular purity, such as transcriptomic analysis, where contamination from other neural cells can significantly skew results. Its ability to sequentially isolate multiple cell types (e.g., microglia via CD11b, followed by astrocytes via ACSA-2, and then neurons) from a single tissue sample is a powerful advantage for comprehensive studies [1]. However, researchers must be cognizant of the cost and the potential for antibody-mediated activation, which could alter the very cellular state under investigation.
Conversely, Percoll gradient separation offers a pragmatic solution for experiments requiring a more naive cellular state or where budget constraints are a significant factor. It is particularly well-suited for functional assays where high yield is prioritized over absolute purity, such as in phagocytosis or migration studies [11]. The primary trade-off is the potential co-isolation of other immune cells, like peripheral macrophages, which can complicate the interpretation of results unless stringent flow cytometric validation with specific markers (e.g., CX3CR1, Siglec-H) is performed post-isolation [13].
Immunomagnetic bead separation, with its foundation in antibody-driven specificity, provides an powerful and precise tool for isolating microglia. The experimental data clearly shows its superior performance in achieving high-purity isolates, which is often critical for downstream molecular analyses. The Percoll gradient method, while less specific, remains a highly valuable technique, offering excellent cell viability, higher yields, and greater simplicity at a lower cost. The decision between these two methods is not a matter of identifying a universal "best" technique, but rather of aligning methodological strengths with specific research goals. Scientists must weigh the critical need for purity against requirements for yield, cost-effectiveness, and the preservation of native cell states when designing their isolation strategy for microglia research.
The isolation of specific cell types from complex tissues is a fundamental requirement in biomedical research. Within the context of neuroscience, the study of microglia—the resident immune cells of the central nervous system—is crucial for understanding neuroinflammation, neurodegeneration, and brain homeostasis [1] [11]. Two predominant methods for isolating microglia have emerged: Percoll gradient centrifugation, which relies on density-based physical separation, and magnetic bead isolation (often referred to as MACS), which utilizes antibody-based immunological capture [1] [15] [10]. This guide provides an objective comparison of these techniques, focusing on their core principles, experimental performance, and suitability for different research scenarios.
Core Principles and Methodologies
The Principle of Percoll Gradient Centrifugation
Percoll gradient centrifugation is an isopycnic separation technique that partitions cells based on their intrinsic buoyant density. Percoll itself is a colloidal suspension of silica particles coated with polyvinylpyrrolidone (PVP) to ensure low toxicity and non-penetrability into cells [16] [17] [18].
- Separation Mechanism: When subjected to centrifugal force, Percoll forms a continuous density gradient. Cells within a mixed suspension migrate through this gradient until they reach a position where their own density matches that of the surrounding Percoll medium, a point known as their isopycnic location [19]. This process effectively separates cell types with different densities into discrete bands [1] [11].
- Key Considerations: The success of the separation hinges on several factors. The osmolality of the Percoll solution must be adjusted with saline or culture medium to be isotonic and prevent cell shrinkage or swelling. The choice of centrifuge rotor (fixed-angle is often preferred), g-force, and centrifugation time also critically influence the shape and resolution of the gradient [17].
The Principle of Magnetic Bead Isolation
Magnetic-activated cell sorting (MACS) relies on immunological recognition rather than physical properties. Antibodies conjugated to magnetic beads bind to specific cell-surface antigens unique to the target cell population, such as CD11b for microglia or ACSA-2 for astrocytes [1] [10]. When the cell suspension is placed in a magnetic field, labeled cells are retained within a column while unlabeled cells pass through, enabling positive selection or depletion [1] [15].
The following diagram illustrates the fundamental workflows and separation logic for these two core principles.
Performance Comparison: Purity, Yield, and Viability
Direct methodological comparisons reveal critical differences in the performance of Percoll gradient centrifugation and magnetic bead isolation. The data below summarize key metrics from experimental studies.
Table 1: Quantitative Comparison of Microglia Isolation Techniques
| Performance Metric | Percoll Gradient Centrifugation | Magnetic Bead Isolation (MACS) | Supporting Experimental Data |
|---|---|---|---|
| Purity | High, but can vary with protocol optimization. Effective myelin debris removal [15]. | Very High. Yields purer populations suitable for deep sequencing [10]. | MACS-isolated microglia show minimal contamination, though a slight myeloid cell presence is possible [10]. |
| Yield | Can suffer from excessive cell damage/density-based loss [11]. | High efficiency reported [10]. Tandem protocols allow sequential isolation of multiple cell types from one brain [1]. | A modified Percoll protocol yielded ~1.5x more cells than other methods from adult mice [11]. |
| Viability | >85% viability reported when combined with enzymatic digestion [15]. | >85% viability reported [10]. | Both methods consistently achieve high cell viability post-isolation [15] [10]. |
| Speed | Protocol duration can be lengthy [11]. | Faster processing for single or multiple samples [10]. | A complete MACS protocol (including density gradient step) can be completed within 2 days [15]. |
| Cost & Equipment | Lower reagent cost, avoids expensive antibodies [1]. Requires standard lab centrifuge. | Higher cost due to antibodies/beads [1]. Requires magnetic separator. | Percoll method circumvents use of expensive fluorescent antibodies or immunomagnetic beads [1]. |
Detailed Experimental Protocols
Detailed Protocol: Percoll Gradient Centrifugation for Microglia
The following is a consolidated protocol for isolating microglia from rodent brain tissue using Percoll density gradient centrifugation, adapted from established methodologies [15] [11].
- Tissue Dissociation: Euthanize the animal and rapidly remove the brain. Dissect the desired brain region, remove the meninges carefully, and chop the tissue into small pieces (~1 mm²) using a sterile scalpel. Digest the tissue with a papain/DNase I solution for 30 minutes at 37°C. Triturate the digested tissue to create a single-cell suspension and filter it through a 100 µm cell strainer [15] [11].
- Mycelial Debris Removal: Centrifuge the single-cell suspension at 500 x g for 5 minutes at 4°C. Resuspend the cell pellet in 8 mL of 30% isotonic Percoll (SIP) in DMEM. Carefully underlay this suspension with 70% SIP using a transfer pipette to create a distinct interface. Centrifuge the tubes at 650 x g for 25 minutes at room temperature with the brake OFF [15].
- Cell Collection: After centrifugation, aspirate the top layer containing myelin and cellular debris. Using a pipette, carefully collect the mononuclear cells from the cloudy interface between the 30% and 70% Percoll layers. Transfer this fraction to a new tube, dilute it with Hank's Balanced Salt Solution (HBSS) to reduce Percoll density, and centrifuge at 500 x g for 5 minutes to pellet the cells [15].
- Cell Culture: Aspirate the supernatant, resuspend the cell pellet in growth medium supplemented with fetal bovine serum and antibiotics, and perform a cell count. Seed the cells in culture flasks. The medium can be supplemented with macrophage colony-stimulating factor (M-CSF) to support microglial growth [11].
Detailed Protocol: Magnetic Bead Isolation for Microglia
This protocol outlines the immunomagnetic separation of microglia, which can be performed after initial tissue dissociation or following a Percoll pre-purification step [15] [10].
- Cell Preparation: Generate a single-cell suspension from brain tissue via enzymatic digestion and mechanical dissociation, as described in steps 1-3 of the Percoll protocol [15].
- Antibody Incubation: Centrifuge the collected mononuclear cells at 300 x g for 10 minutes. Resuspend the cell pellet in a buffer (e.g., PBS with EDTA and BSA). Add CD11b (or other microglia-specific marker) antibody-conjugated magnetic beads to the cell suspension and incubate for 15-30 minutes at 4°C [15].
- Magnetic Separation: Place the cell-bead complex in a magnetic separator for several minutes. During this time, CD11b+ microglia will be retained on the column walls. Carefully wash the column several times with buffer to remove any non-specifically bound, unlabeled cells.
- Elution: Remove the column from the magnetic field. Add an appropriate elution buffer or culture medium and flush out the positively selected, purified microglial cells using a plunger [1] [15].
The Scientist's Toolkit: Essential Research Reagents
Successful cell isolation depends on the use of specific, high-quality reagents. The following table lists key materials and their functions for these protocols.
Table 2: Essential Reagents for Microglia Isolation Protocols
| Reagent / Material | Function | Application Notes |
|---|---|---|
| Percoll | Colloidal silica solution forming inert, non-toxic density gradients for cell separation [16] [17]. | Must be diluted to isotonicity with saline (e.g., 10x PBS) or culture medium before use [17]. |
| Papain / DNase I | Enzymatic cocktail for tissue digestion; papain breaks down intercellular proteins, DNase I digests genomic DNA to reduce viscosity [15]. | Preferable to trypsin for minimizing damage to cell surface epitopes, which is critical for subsequent immunomagnetic sorting [11]. |
| CD11b (ITGAM) Microbeads | Antibody-conjugated magnetic beads for positive selection of microglia via specific surface antigen binding [1] [15]. | Target for microglia isolation. The negative fraction can be used for sequential isolation of other brain cells like astrocytes [1]. |
| ACSA-2 Microbeads | Antibody-conjugated magnetic beads for positive selection of astrocytes via Astrocyte Cell Surface Antigen-2 [1] [10]. | Used for tandem isolation of astrocytes after microglia removal. More suitable for purifying astrocytes from newborn brains [10]. |
| M-CSF / GM-CSF | Growth factors (Macrophage and Granulocyte-Macrophage Colony-Stimulating Factor) added to culture medium [11]. | Support the survival and proliferation of primary microglia in culture post-isolation [11]. |
Integrated Workflow for Optimal Purity
For the highest microglial purity, many researchers combine the physical separation of Percoll gradients with the immunological specificity of MACS. The following diagram outlines this integrated approach, which effectively reduces myelin debris and minimizes non-specific cell binding during the magnetic separation step [15].
Both Percoll gradient centrifugation and magnetic bead isolation are robust methods for microglia isolation, each with distinct advantages. The choice between them is not mutually exclusive and should be guided by the specific research objectives, technical constraints, and desired outcomes.
- Percoll Gradient Centrifugation is a cost-effective, antibody-independent method that excels at removing myelin debris and is highly adaptable. It is ideal for studies where budget is a primary concern or where the goal is to isolate multiple cell types from the same tissue sample [1].
- Magnetic Bead Isolation (MACS) offers superior speed and consistently high purity, making it the preferred choice for high-throughput studies or applications like transcriptomics that demand extremely pure cell populations [10]. Its reliance on specific surface markers is both a strength and a limitation.
- Integrated Approach: For the most demanding applications, a combined protocol utilizing Percoll gradient pre-enrichment followed by MACS purification leverages the strengths of both techniques, yielding microglia of the highest purity and viability while mitigating the limitations of either method used alone [15].
Microglia, the resident immune cells of the central nervous system, play vital roles in brain homeostasis, synaptic pruning, and neuroinflammation [20] [21]. The isolation of pure, functional microglia is fundamental to advancing our understanding of brain physiology and pathology. However, researchers face significant challenges in obtaining high-quality microglial populations, primarily concerning cell viability, prevention of unintended activation, and effective removal of myelin contamination [1] [11]. These challenges are particularly pronounced when working with adult or aging brain tissue, where yields are naturally lower and isolation procedures more demanding [22] [11]. This guide objectively compares the two predominant isolation methodologies—immunomagnetic bead separation and Percoll density gradient centrifugation—within the context of these central challenges, providing researchers with experimental data to inform their protocol selection.
Fundamental Challenges in Microglia Isolation
The path to obtaining representative microglial cells is fraught with technical hurdles that can profoundly impact experimental outcomes.
Preserving Viability and Yield: Isolation procedures, particularly enzymatic digestion and mechanical dissociation, impose significant stress on cells. Adult mouse brain isolation typically yields only 300,000–500,000 microglial cells per brain, underscoring the precious nature of each cell [22]. The method of myelin removal critically affects viability; studies show Percoll gradients yield superior viability compared to sucrose or commercial myelin removal beads [12].
Preventing Unwanted Activation: Microglia are exquisitely sensitive to their environment. The very process of isolation can trigger "culture shock," altering their transcriptional profile and inducing an activated state that does not reflect their in vivo phenotype [21] [11]. This is a major concern for studies aiming to mimic specific physiological or pathological states.
Removing Myelin Contamination: Myelin, the lipid-rich material surrounding axons, is a pervasive contaminant in brain cell suspensions. It can interfere with downstream applications like flow cytometry, RNA sequencing, and cell culture by obstructing equipment and signaling pathways [11] [12]. Effective myelin removal is therefore a non-negotiable step in most protocols.
Comparative Methodologies: Magnetic Beads vs. Percoll Gradients
Immunomagnetic Bead Separation (CD11b+ Isolation)
This method utilizes antibody-conjugated magnetic beads targeting specific cell surface markers, most commonly CD11b, to positively select microglia from a mixed cell suspension [23] [12].
Table 1: Key Experimental Reagents for Magnetic Bead Isolation
| Research Reagent | Function in Protocol |
|---|---|
| CD11b Microbeads | Primary antibody-conjugated magnetic beads for positive selection of microglia [12]. |
| Neural Tissue Dissociation Kit | Enzyme blend for gentle tissue digestion into single-cell suspension [21] [12]. |
| Myelin Removal Beads | Antibody-based beads for negative selection and depletion of myelin debris [12]. |
| MS/LS Columns | Placed in a magnetic field, these columns retain labeled cells during washing [12]. |
| IMAG Buffer (PBS/BSA/EDTA) | Buffer to maintain cell viability and prevent clumping during separation [12]. |
Detailed Protocol:
- Tissue Dissociation: Perfused brain tissue is enzymatically digested using a commercial neural dissociation kit (e.g., Miltenyi Biotec) to create a single-cell suspension [21] [12].
- Myelin Removal: The cell suspension is treated with myelin removal beads and passed through a magnetic column to deplete myelin debris [12].
- Microglial Selection: The myelin-depleted suspension is incubated with CD11b microbeads. The labeled cell suspension is then applied to a magnetic column. CD11b+ cells (microglia) are retained, while unbound cells pass through.
- Elution: Pure microglia are eluted from the column after removal from the magnetic field [23] [12].
Percoll Density Gradient Centrifugation
This is a density-based physical separation method that leverages the fact that different cell types have distinct buoyant densities. Microglia are harvested from the interface between specific Percoll concentrations [22] [1].
Detailed Protocol:
- Tissue Dissociation: The perfused brain is minced and digested in a dissociation medium containing enzymes like dispase II, papain, and DNase I, followed by mechanical trituration [22].
- Gradient Preparation: A discontinuous density gradient is prepared in a centrifuge tube, typically with layers of 70%, 37%, and 30% isotonic Percoll (SIP) [22].
- Centrifugation: The single-cell suspension is layered on top of the gradient and centrifuged (e.g., 300×g for 40 minutes at 18°C with the brake disengaged). Myelin and debris migrate to the top, while microglia collect at the 70%-37% Percoll interphase [22].
- Collection: The microglia-containing interphase is carefully aspirated, diluted in buffer, and centrifuged to wash away residual Percoll [22].
Head-to-Head Comparative Data
The choice between magnetic bead and Percoll gradient methods involves trade-offs between purity, yield, activation state, and technical practicality.
Table 2: Method Comparison Based on Experimental Data
| Parameter | Immunomagnetic Beads | Percoll Gradient |
|---|---|---|
| Reported Purity | >90% – highly purified CD11b+ population [12] | >90% – high purity achievable [21] |
| Reported Yield | Lower yield from adult tissue; suitable for molecular analyses [21] [12] | Higher yield from adult mouse brain (300,000–500,000 cells/brain) [22] |
| Viability Post-Isolation | High viability reported; Percoll myelin removal gave the highest viability [12] | High viability achievable; dependent on gentle mechanical dissociation [22] [12] |
| Baseline Activation | Phenotype accurately reflects in vivo state; isolation itself does not activate cells [12] | Risk of activation during longer dissociation and centrifugation steps [11] |
| Key Advantage | Specificity for microglial marker; best for preserving native state and for low-cell-number inputs [12] | High cell yield; no requirement for specific surface antibodies; cost-effective for large-scale isolation [22] [1] |
| Primary Limitation | Lower yield from adult tissue; higher cost of reagents and columns [21] [12] | Potential for mechanical activation; requires optimization of gradient densities [11] |
Impact on Microglial Signaling and Functional Pathways
The isolation methodology can influence the observed microglial functions, including critical processes like chemotaxis, phagocytosis, and cytokine signaling [20].
For instance, phagocytosis, a critical microglial function assessed by the uptake of fluorescent beads or pathogens, appears to be more robust in microglia isolated via certain methods. Primary human microglia and induced pluripotent stem cell (iPSC)-derived microglia show significantly higher phagocytic capacity compared to immortalized cell lines [5]. Furthermore, the secretory profile in response to stimuli like HMGB1 or LPS varies significantly. One study noted that microglia cultured from neonates showed a larger number of differentially expressed genes in response to HMGB1 than those from adult mice [21]. Notably, rodent microglia can be stimulated to secrete large quantities of nitric oxide, a function that is either absent or significantly blunted in human microglia, highlighting a key species difference [3].
The Scientist's Toolkit: Essential Reagent Solutions
Successful isolation hinges on the use of specific, high-quality reagents.
Table 3: Essential Research Reagent Solutions for Microglia Isolation
| Reagent / Kit | Critical Function | Application in Protocols |
|---|---|---|
| Neural Tissue Dissociation Kit | Optimized enzyme blend for gentle and effective brain tissue digestion into single cells. | Universal first step in both magnetic bead and Percoll protocols [21] [12]. |
| CD11b Microbeads | Antibody-conjugated magnetic beads for positive selection of microglia via the CD11b surface marker. | Core reagent for immunomagnetic bead separation [23] [12]. |
| Percoll | Silica nanoparticle solution used to form density gradients for cell separation based on buoyant density. | Core reagent for density gradient centrifugation [22] [12]. |
| DNase I | Enzyme that degrades DNA released by damaged cells, preventing cell clumping and sticky viscosity. | Added during tissue dissociation in most protocols [22]. |
| GM-CSF / M-CSF | Growth factors (Granulocyte/Macrophage Colony-Stimulating Factor) added to culture media to support microglial survival and proliferation in vitro. | Used in post-isolation culture, particularly for adult microglia [21] [11]. |
The decision between immunomagnetic bead isolation and Percoll gradient centrifugation is not a matter of identifying a universally superior technique, but rather of selecting the right tool for the specific research question. The experimental data consistently show that both methods can achieve high purity (>90%). The critical differentiators are yield, preservation of native state, and cost.
For studies requiring the highest fidelity to the in vivo phenotype, such as transcriptomic analyses or sensitive response assays, immunomagnetic separation is generally preferable due to its minimal activation signature. Conversely, for experiments demanding large cell numbers for functional screens or protein-based assays where yield is paramount, the Percoll gradient method offers a robust and cost-effective solution. Ultimately, researchers must weigh the trade-offs between these fundamental parameters against their specific experimental goals to ensure their isolation methodology faithfully supports their scientific inquiry.
Step-by-Step Protocols: From Brain Tissue to Purified Microglia Cultures
Standardized Protocol for Magnetic Bead Isolation Using CD11b Antibodies
Immunomagnetic cell separation using CD11b antibodies provides a highly specific method for isolating microglia from the central nervous system. This technique leverages CD11b (Integrin αM), a surface marker highly expressed on microglia and other myeloid cells, to positively select target cells from dissociated brain tissue. This guide objectively compares CD11b magnetic bead isolation with the traditional Percoll gradient method, examining performance metrics including cell purity, viability, yield, and phenotypic preservation to inform protocol selection for neuroscience research.
The isolation of pure, functionally intact microglia is fundamental to studying neuroinflammation, neurodegenerative diseases, and CNS homeostasis. The choice of isolation methodology significantly impacts experimental outcomes, with CD11b magnetic bead isolation and Percoll gradient centrifugation representing two predominant approaches [1] [24]. Magnetic bead isolation is an immunoaffinity-based technique that uses antibodies against the CD11b surface antigen conjugated to magnetic particles to selectively capture microglia from a single-cell suspension [25] [26]. In contrast, Percoll isolation is a density-based separation that relies on physical differences in cell buoyancy to enrich microglia from other neural cells and myelin debris [1] [24]. This guide provides a standardized protocol for CD11b magnetic bead isolation and presents a direct, data-driven comparison with the Percoll method, equipping researchers with the information needed to select the optimal technique for their specific research context.
CD11b Magnetic Bead Isolation: Principle and Workflow
The CD11b Antigen and Isolation Principle
CD11b (also known as Integrin αM, Mac-1, or CR3) is a subunit of the integrin receptor CD11b/CD18, which is highly expressed on the surface of innate immune cells including microglia, macrophages, neutrophils, and monocytes [25] [26]. Immunomagnetic separation exploits this specific surface expression. The process involves incubating a single-cell brain suspension with magnetic beads (typically 2.7 μm in diameter) conjugated with anti-CD11b antibodies [25]. When placed in a magnetic field, CD11b+ cells bound to the beads are retained, while negative cells are washed away. The positive fraction can then be eluted, yielding a highly purified microglial population [24] [26].
Standardized Step-by-Step Workflow
The following diagram illustrates the core workflow for CD11b magnetic bead isolation:
Detailed Protocol Steps [24] [26]:
- Tissue Preparation and Dissociation: Perfuse mice with ice-cold PBS. Dissect brain regions of interest and subject to enzymatic digestion using a neural tissue dissociation kit (e.g., from Miltenyi Biotec) for 35 minutes at 37°C. Pass the resulting suspension through a 40 μm cell strainer to remove tissue debris.
- Myelin Removal (Critical Pre-step): Pellet dissociated cells and resuspend in 30% Percoll solution. Centrifuge at 700 × g for 10 minutes. Carefully remove the myelin-containing supernatant and wash the cell pellet with HBSS. This step is crucial for reducing contamination and improving bead-binding efficiency [24].
- Magnetic Labeling: Resuspend the cell pellet in IMAG buffer (PBS with 0.5% BSA and 2 mM EDTA). Incubate with anti-CD11b magnetic beads (e.g., 50 μL per 10^7 total cells) for 30 minutes at 6-12°C with thorough mixing [26].
- Magnetic Separation: Place the labeled cell suspension in a magnetic separator (e.g., BD IMag Magnet or MS Columns). Incubate for 6-8 minutes at room temperature. Carefully aspirate the supernatant containing the CD11b-negative fraction.
- Washing and Elution: Remove the tube from the magnet and resuspend the retained positive fraction in buffer. Return the tube to the magnet for a second separation (2-4 minutes) and discard the wash supernatant. Repeat this wash step once. Finally, resuspend the purified CD11b+ cells in an appropriate culture or assay buffer.
Direct Comparison: CD11b Magnetic Beads vs. Percoll Gradient
Performance Metrics and Experimental Data
The following table summarizes key performance characteristics of both methods, synthesized from comparative studies:
| Performance Metric | CD11b Magnetic Bead Method | Percoll Gradient Method |
|---|---|---|
| Cell Purity | Very High (>90%) [24] [10]. Minimal astrocyte/neuronal contamination. | Moderate. Can have myeloid cell contamination [10]. |
| Cell Viability | >85% when combined with Percoll for myelin removal [24] [11]. | High, but can be lower due to prolonged centrifugation causing cell damage [11]. |
| Cell Yield | Good, but dependent on age and tissue source [1]. | Variable; can be lower due to cell loss during density steps [11]. |
| Phenotype Preservation | Excellent. Accurately reflects in vivo state, including quiescent and activated profiles [24]. | Good, but enzymatic/mechanical stress may induce subtle activation [11]. |
| Processing Speed | Relatively Fast (~2-3 hours). Magnetic separation takes minutes [10]. | Slower. Requires long centrifugation steps (up to 30-45 mins) [11]. |
| Technical Ease | Requires antibody titration and optimization. Amenable to automation [27]. | Technically straightforward, but requires careful handling of viscosity [1]. |
| Cost Consideration | Higher cost due to commercial kits and specific antibodies [1]. | Lower reagent cost, but requires specialized equipment for consistent results [1]. |
| Downstream Applications | Ideal for functional assays, sequencing, flow cytometry, and culture [24]. | Suitable for basic culture, RNA/protein analysis, but may have more contamination [1]. |
Experimental Data Supporting the Comparison
- Purity and Contamination: A direct methodological comparison found that while both methods yield high viability (>85%), MACS-sorted microglia can comprise "slight myeloid cells contamination but with a little bit higher efficiency than that sorted by FACS" [10]. The same study noted that MACS processing was faster than FACS.
- Viability and Yield: A critical study systematically evaluating myelin removal methods for subsequent CD11b bead isolation found that "the viability and yield of isolated cells were significantly affected by the myelin removal method." It concluded that "the highest viability and number of CD11b+ cells was obtained with Percoll" over sucrose or anti-myelin beads, highlighting that a hybrid approach can be optimal [24].
- Phenotypic Accuracy: Research demonstrates that CD11b immunomagnetic separation "does not activate the isolated cells" and that "microglia isolated from LPS-treated mice displayed a pro-inflammatory phenotype... whereas microglia isolated from control mice did not," confirming the method preserves both quiescent and activated states [24].
The Scientist's Toolkit: Essential Reagents and Equipment
Successful implementation of the CD11b magnetic bead protocol requires the following key materials.
| Item Category | Specific Product/Reagent Examples | Function in Protocol |
|---|---|---|
| Magnetic Bead Kits | EasySep Mouse CD11b Positive Selection Kit II [28]; BD IMag Anti-CD11b Magnetic Particles - DM [26]; Human CD11b Magnetic Beads [25] | Core reagent for immunomagnetic capture of CD11b+ cells. |
| Cell Separation Magnet | BD IMag Cell Separation Magnet [26]; EasySep Magnet [28] | Device to generate magnetic field for separating bead-bound cells. |
| Dissociation Kit | Neural Tissue Dissociation Kit (Miltenyi Biotec) [24] | Enzymatic blend for generating single-cell suspension from brain tissue. |
| Myelin Removal Reagent | Percoll (GE Healthcare) [24] | Density gradient medium for removing myelin debris post-dissociation. |
| Cell Strainer | 70 μm nylon cell strainer [26] | Removal of cell clumps and tissue debris before separation. |
| Separation Buffer | IMAG Buffer (PBS, 0.5% BSA, 2 mM EDTA) [24] [26] | Buffer for cell washing, labeling, and separation to maintain viability. |
How to Choose Your Method
The decision between CD11b magnetic bead and Percoll gradient isolation should be guided by your specific experimental goals and constraints. The following decision tree outlines a logical selection process:
- Choose CD11b Magnetic Bead Isolation when: Your research requires maximum cell purity and a preserved phenotype for downstream functional assays, single-cell sequencing, or flow cytometry. This is the preferred method for studying specific microglial activation states, transcriptomics, and when working with complex disease models where precise cellular identity is critical [24] [10].
- Choose Percoll Gradient Isolation when: The priority is a cost-effective and technically straightforward method for bulk protein or RNA analysis where some cellular heterogeneity is acceptable. It is also suitable for labs initiating microglia isolation protocols or with limited budgets for specialized immunomagnetic reagents [1] [11].
- Consider a Hybrid Approach: For optimal results, many protocols incorporate a Percoll gradient step for initial myelin removal followed by CD11b magnetic bead separation. This combination leverages the strengths of both methods, effectively reducing debris and enriching for a high-viability, high-purity microglial population [24].
Both CD11b magnetic bead isolation and Percoll gradient centrifugation are validated methods for purifying microglia. The immunomagnetic approach offers superior specificity, purity, and phenotypic fidelity, making it ideal for advanced, mechanistic studies. The density-based Percoll method provides an accessible and economical alternative for foundational research. The standardized protocol and comparative data presented herein empower researchers to make an evidence-based selection, ultimately enhancing the reliability and reproducibility of microglial research in neuroscience and drug development.
Optimized Protocol for Percoll Gradient Separation and Myelin Removal
The isolation of pure, functionally intact microglia is a cornerstone of neuroimmunology research. The choice of isolation methodology significantly impacts cell yield, purity, and phenotypic preservation, thereby influencing subsequent experimental outcomes. This guide provides a systematic, data-driven comparison between the established density-based separation using Percoll gradients and the increasingly popular immunomagnetic bead-based isolation. We objectively evaluate these techniques based on quantitative metrics including microglial yield, purity, viability, and the preservation of native phenotypes, providing researchers with the evidence necessary to select the optimal protocol for their specific applications.
Microglia, the resident macrophages of the central nervous system, play pivotal roles in brain development, homeostasis, and the neuroinflammation associated with virtually all CNS disorders [12]. The study of these cells in their native state requires isolation methods that are not only efficient but also minimize cellular activation and preserve physiological phenotypes. The brain's unique environment, characterized by high lipid content and complex cellular interactions, presents specific challenges for cell isolation, particularly the need to remove vast amounts of myelin debris which can interfere with downstream analyses [29].
Two principal methodologies have emerged for microglial isolation: Percoll gradient centrifugation, a density-based physical separation method, and immunomagnetic bead sorting (MACS), an antibody-based affinity technique. The ongoing debate in the field centers on which method offers superior performance in terms of purity, yield, and functional preservation. This guide synthesizes current experimental evidence to directly compare these techniques, providing detailed protocols and quantitative data to inform protocol selection for research and drug development.
Technical Comparison: Percoll Gradient vs. Magnetic Bead Isolation
The following comparison is based on aggregated data from multiple published studies that have directly or indirectly quantified the performance of these two isolation methods.
Table 1: Quantitative Comparison of Microglia Isolation Methods
| Performance Metric | Percoll Gradient Method | Magnetic Bead (MACS) Method |
|---|---|---|
| Cell Viability | High (Reportedly the highest among methods tested) [12] | High, but can be lower depending on bead binding and elution [11] |
| Purity (CD11b+ Cells) | High [30] | Very High (Highly purified without astrocyte/neuronal contamination) [12] |
| Microglial Yield | High, but some loss can occur during gradient steps [30] | High, efficient recovery of CD11b+ cells [12] |
| Phenotype Preservation | Preserves in vivo phenotype, suitable for activated microglia [12] | Preserves in vivo phenotype, reflects both quiescent and activated states [12] |
| Cost & Technical Demand | Lower reagent cost, requires optimization of gradient concentration [29] [31] | Higher reagent cost, streamlined and reproducible protocol [32] |
| Throughput & Speed | Moderate, involves lengthy centrifugation [11] | Fast, rapid separation via magnetic field [32] |
| Key Advantage | Effective myelin removal and cell separation based on physical properties [29] | High specificity for CD11b+ cells, minimal non-microglial contamination [12] |
| Main Limitation | Can be harsh on cells, potentially affecting viability and function [11] | Antibody binding could theoretically affect some downstream applications |
Workflow Visualization
The following diagram illustrates the key decision points and procedural steps involved in the two primary microglia isolation workflows, from tissue dissociation to the final isolated cell population.
Detailed Experimental Protocols
Optimized Percoll Gradient Protocol
Principle: This method separates cells based on their differential buoyant densities. Microglia, being less dense than myelin but more dense than other neural cells, can be partitioned into a distinct layer.
Step-by-Step Procedure:
Tissue Dissociation:
- Transcardially perfuse mice with ice-cold PBS to remove circulating blood cells [30].
- Dissect the brain region of interest and mechanically dissociate it into small pieces in HBSS.
- Enzymatic digestion is critical. Studies have compared various enzymes. Accutase has been shown to result in one of the highest microglial yields with low variance [30]. Alternatively, papain (1 mg/mL) or a combination of papain and dispase II has been found effective for a balanced isolation of multiple brain cell types, especially in adult mice [31]. Incubate for 30 minutes at 37°C.
Percoll Solution Preparation:
- First, prepare Isotonic Percoll (SIP): Mix 90% Percoll with 10% 10X PBS. Ensure all components are at room temperature before mixing [32].
- The optimal concentration for myelin removal is critical. Research indicates that centrifugation at ≥24% SIP is required to effectively separate myelin debris from cells, with 26% SIP leaving no observable myelin debris [29]. A 30% Percoll solution (prepared by mixing 70% SIP with 30% 1X PBS) is also commonly and effectively used [12].
Density Gradient Centrifugation:
- Resuspend the single-cell suspension in the prepared Percoll solution (e.g., 24-30% SIP).
- Centrifuge at 700 × g for 10 minutes at room temperature, with the brake disabled to prevent disturbing the gradient [12].
- After centrifugation, myelin will be in the supernatant. Carefully aspirate the supernatant without disturbing the cell pellet at the bottom of the tube.
Cell Collection and Washing:
- Resuspend the cell pellet in a buffered solution like HBSS or Post-Percoll Solution (HBSS with calcium/magnesium, 5% FBS, 10 μM HEPES) [32].
- Centrifuge again to wash away residual Percoll. The pellet now contains an enriched population of microglia, ready for flow cytometry analysis or culture.
Immunomagnetic Bead (MACS) Protocol
Principle: This method uses magnetic microbeads conjugated to an anti-CD11b antibody to specifically label microglia, which are then isolated in a magnetic field.
Step-by-Step Procedure:
Tissue Dissociation and Myelin Removal:
- The initial steps of perfusion, dissection, and enzymatic digestion (e.g., using a neural tissue dissociation kit) are similar to the Percoll protocol [12].
- While MACS can be performed directly on the dissociate, a prior myelin removal step is often recommended. This can be achieved using a commercial Debris Removal Solution or a preliminary Percoll or sucrose gradient to improve the efficiency of the magnetic separation [32] [12].
Magnetic Labeling:
- Pass the single-cell suspension through a 40 μm cell strainer to remove tissue debris.
- Resuspend the cell pellet in cold MACS Buffer (PBS, pH 7.4, supplemented with 0.5% BSA and 2 mM EDTA).
- Incubate the cell suspension with PE-conjugated anti-CD11b antibodies for 10 minutes in the cold, followed by incubation with anti-PE magnetic microbeads for 15 minutes [12]. The amounts should follow the manufacturer's guidelines.
Magnetic Separation:
- Place the labeled cell suspension onto a pre-washed LS or MS MACS Column positioned in a magnetic separator.
- The CD11b-negative cells will pass through the column and can be collected as the effluent fraction.
- After washing the column with buffer, remove it from the magnetic field. Elute the positively selected CD11b+ microglia by applying buffer and firmly pushing the plunger.
Post-Isolation Handling:
The Scientist's Toolkit: Essential Research Reagents
The following reagents and kits are fundamental for the successful execution of the microglia isolation protocols described above.
Table 2: Key Reagents for Microglia Isolation Protocols
| Reagent / Kit | Function / Application | Key Considerations |
|---|---|---|
| Percoll | Silica-based density gradient medium for separation of cells and removal of myelin debris. | Must be rendered isotonic by mixing with 10X PBS before use. Concentration (24-30%) is critical for efficiency [29] [32]. |
| Anti-CD11b Microbeads (Miltenyi) | Magnetic beads for positive selection of microglia via immunomagnetic separation (MACS). | The backbone of the MACS protocol. Recognizes the integrin alpha M (ITGAM) surface protein [32] [12]. |
| Accutase | Enzymatic blend for tissue dissociation. | Shown to provide high microglial yield with low experimental variance [30]. |
| Papain | Protease for enzymatic digestion of brain tissue. | Effective for combined isolation of microglia, astrocytes, and infiltrating leukocytes [31]. |
| Collagenase A | Enzyme for chemical dissociation of brain tissue. | Commonly used in historical protocols; performance may vary compared to newer enzymes [32]. |
| DNase I | Enzyme that digests DNA released by damaged cells. | Reduces cell clumping and is typically added to enzymatic digestion mixes to improve cell yield and viability [32]. |
| MACS Buffer | Buffer for magnetic cell separation. | Protects cell viability and reduces non-specific binding during the MACS procedure [32]. |
| Debris Removal Solution | Commercial solution for myelin removal. | An alternative to Percoll for cleaning samples before MACS separation [32]. |
Discussion and Concluding Recommendations
The choice between Percoll gradient and magnetic bead isolation is not a matter of one being universally superior, but rather which is best suited for the specific research context.
For studies requiring the highest purity with minimal astrocytic or neuronal contamination, the immunomagnetic bead (MACS) method is recommended. Its high specificity for CD11b+ cells ensures a pure microglial population and is excellent for molecular analyses like qRT-PCR and RNA sequencing [12]. The protocol is highly reproducible and less prone to user variation once established.
For research where high cell yield and the simultaneous isolation of other glial cells are priorities, the Percoll gradient method is a robust choice. It effectively removes myelin and can be more cost-effective for processing large numbers of samples [30]. It remains the preferred method for many and avoids potential concerns of antibody binding affecting cell function.
A critical consideration is that these methods are not mutually exclusive. Many optimized protocols now use a hybrid approach, employing a gentle Percoll or sucrose gradient for initial myelin debris removal, followed by MACS separation for final purification of microglia [12]. This combination can leverage the strengths of both techniques to achieve superior results.
Ultimately, researchers must weigh the parameters of purity, yield, viability, cost, and technical feasibility against their experimental goals. The data and protocols provided herein offer a foundation for making an evidence-based decision to ensure the reliability and translational relevance of microglial research.
The isolation of pure, functional primary cells from brain tissue is a cornerstone of neuroscience research, enabling precise studies of cellular behavior, signaling pathways, and disease mechanisms. The central challenge lies in efficiently isolating multiple, high-purity cell types from a single, often limited, biological sample. Traditional methods that process separate aliquots for each cell type are inefficient, require more animal subjects, and introduce inter-sample variability. Within this context, tandem isolation protocols have emerged as a powerful solution, allowing for the sequential separation of neurons, astrocytes, and microglia from a single tissue preparation.
This guide objectively compares the performance of the tandem magnetic bead isolation method against a popular alternative—the Percoll density gradient—focusing on microglia purity, yield, and functional integrity. The comparison is framed within a broader thesis on optimizing cell isolation for neurodegenerative disease research, providing researchers and drug development professionals with the data needed to select the most appropriate method for their experimental goals.
Core Methodologies and Comparison Framework
Tandem Magnetic-Activated Cell Sorting (MACS)
The tandem MACS protocol is an immunomagnetic method that uses sequential positive and negative selection steps to isolate multiple cell types from a single-cell suspension. A well-established protocol involves isolating microglia, astrocytes, and neurons in sequence from the same mouse brain tissue [1].
The process begins with a single-cell suspension obtained from dissected brain tissue through mechanical disruption and enzymatic digestion (e.g., with trypsin). This suspension is first incubated with CD11b (ITGAM) antibody-conjugated magnetic beads to capture microglia, which are retained in a magnetic column while the negative fraction passes through [1] [23]. The flow-through is then incubated with ACSA-2 (Astrocyte Cell Surface Antigen-2) antibody-conjugated beads to positively select astrocytes [1]. Finally, the remaining cell suspension (negative for both CD11b and ACSA-2) is incubated with a biotin-antibody cocktail targeting non-neuronal cells and depleted using streptavidin beads, leaving behind a purified neuronal population by negative selection [1]. This sequential process maximizes the use of a single sample.
Percoll Density Gradient Centrifugation
Percoll gradient centrifugation is a density-based physical separation method that avoids the use of antibodies. In this technique, a pre-formed density gradient is created by centrifuging a solution of Percoll, a colloidal silica coated with polyvinylpyrrolidone [1] [11]. The single-cell suspension from the brain is layered on top of the gradient and centrifuged. Cells migrate to and band at their specific buoyant densities, effectively separating different cell types [11]. For example, one protocol designed to circumvent enzymatic digestion isolates primary microglia and astrocytes from rodent CNS based on their inherent density differences [1]. While effective at removing myelin and cellular debris, the procedure involves long centrifugation times and can subject cells to mechanical stress during the dissociation process prior to spinning [11].
Objective Performance Comparison: Key Experimental Data
Direct comparative studies on these methods are limited, but data from individual protocol papers and a methodological comparison study allow for a performance summary. The following table synthesizes key quantitative metrics for microglia isolation, which is often the primary focus of purity and yield analyses.
Table 1: Performance Comparison of Microglia Isolation Techniques
| Performance Metric | Tandem MACS (CD11b+) | Percoll Gradient | Notes and Context |
|---|---|---|---|
| Purity (CD11b+) | ~95% [23] | Acceptable, but potentially lower vs. MACS [11] | MACS purity is well-documented. Percoll purity can be sufficient for many applications. |
| Cell Yield | High, but age-dependent [1] | Variable; can suffer from low yield and cell damage [11] | Yield for both methods is influenced by protocol specifics and animal age. |
| Cell Viability | Generally high [34] | Can be compromised by mechanical stress [11] | The gentle magnetic labeling in MACS is less disruptive. |
| Relative Cost | Higher (antibodies, magnetic columns) [1] | Lower (common laboratory reagents) [11] | Percoll is a cost-effective alternative, though specialized kits exist. |
| Technical Demand | Moderate (requires specific antibodies) [34] | Accessible and cost-effective [34] | Percoll requires a centrifuge but is otherwise simple. |
| Risk of Activation | Potential for activation from antibody binding [34] | Risk from prolonged centrifugation and dissociation [11] | Both methods require careful execution to minimize activation. |
| Multi-cell Type Output | Yes (Sequential isolation of microglia, astrocytes, neurons) [1] | Limited (Typically isolates microglia and astrocytes only) [1] | Tandem MACS is uniquely efficient for obtaining three distinct populations. |
A 2023 study that compared three microglia isolation protocols, including a modified protocol (PROTOCOL 1) based on density gradients and adherence, provides insightful performance data. While not a direct MACS-vs-Percoll comparison, it highlights the variability in outcomes achievable with different methodological approaches. The study reported significant differences in cell yield and noted that different preparation protocols can result in slightly different microglial phenotypes, which can influence experimental results [11].
Detailed Experimental Protocols
Detailed Protocol: Tandem MACS for Microglia, Astrocytes, and Neurons
This protocol is adapted for a single brain from a 9-day-old mouse [1]. Researchers must optimize conditions for tissue from other ages or species.
Step 1: Tissue Dissociation.
- Dissect the brain region of interest and carefully remove the meninges.
- Mechanically dissociate the tissue in a suitable buffer.
- Use enzymatic digestion with trypsin (e.g., 15-20 min at 37°C) to create a single-cell suspension.
- Inactivate the protease, filter the homogenate through a 70-μm cell strainer, and centrifuge to pellet cells. Resuspend the pellet in a cold, buffered solution containing Fc receptor blockers to reduce non-specific antibody binding.
Step 2: Microglia Isolation (CD11b+ Selection).
- Incubate the total cell suspension with anti-CD11b microbeads for 15-20 minutes at 4-8°C.
- Pass the cell-bead mixture through a pre-washed magnetic column placed in a separator. The labeled microglia are retained in the column.
- Collect the flow-through fraction (CD11b- cells) for subsequent isolations.
- Remove the column from the magnet and flush out the positively selected microglia using a syringe plunger and an appropriate buffer.
Step 3: Astrocyte Isolation (ACSA-2+ Selection).
- Take the CD11b- flow-through fraction and centrifuge it.
- Resuspend the cell pellet in buffer and incubate with anti-ACSA-2 microbeads.
- Pass the mixture through a new magnetic column. The ACSA-2+ astrocytes are retained.
- Collect the flow-through fraction (CD11b-/ACSA-2- cells) for the final step.
- Elute the purified astrocytes from the column.
Step 4: Neuronal Isolation (Negative Selection).
- Take the CD11b-/ACSA-2- cell fraction and centrifuge it.
- Incubate the cell pellet with a biotinylated antibody cocktail against non-neuronal cells (e.g., targeting remaining microglia, astrocytes, and oligodendrocyte precursors), followed by incubation with anti-biotin microbeads.
- Pass the mixture through a magnetic column. The labeled non-neuronal cells are retained, and the flow-through contains the purified neurons.
Post-Isolation: Count cells and assess viability using trypan blue exclusion. Plate cells in pre-coated culture vessels with optimized medium formulations. Phenotypic characterization of each batch using markers like IBA-1 (microglia), GFAP (astrocytes), and MAP-2 (neurons) is recommended to ensure purity and monitor for activation [1].
Detailed Protocol: Percoll Gradient for Microglia and Astrocytes
This protocol, based on Agalave et al., is designed to isolate microglia and astrocytes without enzymatic digestion or expensive antibodies [1].
Step 1: Tissue Homogenization.
- Dissect the brain and remove meninges. Mechanically dissociate the tissue using a gentleMACS Dissociator or by trituration through pipette tips in a cold, isotonic solution. Avoid enzymatic digestion.
Step 2: Gradient Preparation and Centrifugation.
- Prepare a pre-formed, discontinuous Percoll gradient. For example, layer different concentrations of Percoll (e.g., 30%, 50%, 70%) in a centrifuge tube.
- Carefully layer the single-cell suspension on top of the gradient.
- Centrifuge at a fixed speed and temperature (e.g., 500 × g for 20-30 minutes at 18°C) with the brake disengaged to prevent gradient disturbance.
Step 3: Cell Collection.
- After centrifugation, distinct cell bands will be visible at the interfaces of the different densities.
- Microglia typically band at a lower density (e.g., at the 50%/70% interface), while astrocytes and other neural cells band at higher densities.
- Carefully aspirate the desired cell bands using a Pasteur pipette.
Step 4: Washing and Culture.
- Transfer the collected cell fractions to a new tube containing a large volume of buffer to dilute the Percoll.
- Centrifuge the cells to form a pellet and wash at least twice to remove all traces of Percoll.
- Resuspend the purified microglia and astrocytes in their respective culture media for downstream applications.
Diagram 1: Tandem Isolation Workflow Comparison. This diagram illustrates the sequential, antibody-dependent steps of the Tandem MACS protocol versus the density-based, simultaneous separation of the Percoll gradient method.
The Scientist's Toolkit: Essential Research Reagents
Successful cell isolation relies on specific, high-quality reagents. The following table details essential materials for executing the tandem MACS protocol.
Table 2: Essential Reagents for Tandem MACS Isolation
| Item | Function / Application | Specific Examples / Notes |
|---|---|---|
| Anti-CD11b Microbeads | Positive selection of microglial cells. | Also known as ITGAM. Binds to microglia and other myeloid cells [1] [23]. |
| Anti-ACSA-2 Microbeads | Positive selection of astrocytic cells. | Targets Astrocyte Cell Surface Antigen-2 from the negative fraction after microglia removal [1]. |
| Non-Neuronal Biotin-Ab Cocktail | Negative selection of neuronal cells. | Depletes remaining non-neuronal cells (e.g., oligodendrocytes, endothelial cells) to yield a pure neuronal population [1]. |
| Magnetic Separator & Columns | Physical separation of magnetically labeled cells. | Various systems (e.g., MACS Separators from Miltenyi Biotec) are available, scaled to sample size. |
| Cell Separation Buffer | Suspension medium for isolation steps. | Typically a cold, buffered salt solution (e.g., PBS) with additives like EDTA and bovine serum albumin to prevent clumping. |
| Enzymatic Digestion Mix | Tissue dissociation to create single-cell suspension. | Often includes trypsin or papain, and DNase I to digest intercellular proteins and reduce clumping [1]. |
| Phenotypic Validation Antibodies | Post-isolation confirmation of cell purity and identity. | IBA-1 (for microglia) [1], GFAP (for astrocytes) [1], MAP-2 (for neurons) [1]. |
The choice between tandem MACS and Percoll gradient isolation is not a matter of one being universally superior, but rather which is optimal for a specific research question and resource context.
Tandem MACS is the definitive method when the experimental goal is to obtain three highly pure cell populations (microglia, astrocytes, and neurons) from a single sample. Its principal advantages are high purity and the efficient multiplexing of cell types, which minimizes inter-sample variability and animal use. These benefits come at a higher financial cost and require the availability of specific, high-affinity antibodies. The potential for antibody-induced cell activation must also be considered in functional assays [1] [34].
Percoll Gradient Centrifugation offers a cost-effective and antibody-free alternative, making it ideal for studies where antibody binding might interfere with downstream analysis or when budget constraints are a primary concern. It is well-suited for simultaneously isolating microglia and astrocytes. Its limitations include a generally lower yield and purity compared to MACS, an inability to easily isolate neurons, and a greater risk of cell damage or activation due to mechanical stress during processing [1] [11].
For research focused on high-content multi-omics, functional studies requiring minimal cell activation, or the isolation of neurons, tandem MACS presents a compelling case. Conversely, for rapid, cost-effective isolation of microglia and astrocytes for initial screening or when working with limited antibody options, Percoll gradients remain a valuable and accessible tool. As single-cell technologies and the need to model complex cell-cell interactions advance, the ability to cleanly isolate multiple native cell types from one sample will only grow in importance, solidifying the role of tandem isolation strategies in the neuroscience toolkit.
The choice between immunomagnetic separation (e.g., CD11b magnetic beads) and density gradient centrifugation (e.g., Percoll) for microglia isolation directly influences the subsequent cellular phenotype and health, making the ensuing culture conditions a critical determinant of experimental success [1] [35]. Immunomagnetic separation leverages antibody-antigen interactions to specifically target and isolate microglia, typically using antibodies against surface markers like CD11b (ITGAM) [36] [12]. In contrast, Percoll gradient isolation is a density-based method that separates cells physically, circumventing the use of antibodies and potential enzymatic digestion that can affect cell viability and surface antigen integrity [1] [37]. A primary concern with any isolation method, particularly those involving enzymatic and mechanical dissociation, is the induction of ex vivo activation artifacts, which can alter the microglia's transcriptomic and translatomic profiles away from their true in vivo state [35]. Therefore, the initial isolation event sets the stage, and the subsequent culture environment—media, supplements, and substrate—is pivotal for maintaining microglial homeostasis or for selectively guiding their activation for disease modeling.
Comparative Analysis of Post-Isolation Culture Conditions
The table below summarizes key culture parameters for microglia isolated via different methodologies, highlighting how the isolation technique often dictates the optimal initial culture environment.
Table 1: Culture Conditions for Microglia Post-Isolation
| Culture Parameter | Commonly Used Formulations & Coatings | Isolation Method Context & Considerations |
|---|---|---|
| Basal Media | DMEM high glucose [38], DMEM/F-12 with GlutaMAX [11] | Used for microglia from magnetic bead (CD11b+) isolation [38] and Percoll-based protocols [11]. |
| Serum Supplement | 10% Fetal Bovine Serum (FBS) [36] [11] [38] | A standard supplement for primary microglial culture following both magnetic bead and Percoll isolation. |
| Growth Factors & Conditioned Medium | M-CSF (100 ng/mL) and GM-CSF (100 ng/mL) [11]; 50% conditioned medium from mixed brain cells [11] | Critical for survival and proliferation of microglia isolated from adult mice via modified protocols, often after Percoll gradients. Colony-stimulating factors support microglial health post-isolation [38]. |
| Substrate Coating | Poly-D-Lysine [36] [38] | Used to promote adherence for microglia cultured after both magnetic bead [38] and refined CD11b isolation [36]. Essential for plating cells after Percoll-based flow cytometry protocols [37]. |
| Specialized Supplements | 5 μg/mL Insulin [38] | Included in media for certain specialized microglial cultures derived from magnetic bead isolation. |
Detailed Experimental Protocols for Culture Setup
Protocol for Culturing CD11b+ Immunomagnetically Isolated Microglia
This protocol is adapted from methodologies used to culture microglia isolated with magnetic beads targeting the CD11b surface antigen [36] [38].
- Pre-coating Culture Vessels: Prior to cell plating, coat culture flasks, plates, or dishes with Poly-D-Lysine to enhance cellular adhesion. Incubate the vessels with the coating solution for the recommended time, then aspirate and allow them to air dry completely under sterile conditions [36] [38].
- Preparation of Complete Culture Medium: Prepare a growth medium composed of DMEM high glucose, supplemented with 10% Fetal Bovine Serum (FBS), 1% penicillin/streptomycin, and 0.1% GlutaMAX [38]. For certain applications, further supplementation with 5 μg/mL insulin can be beneficial [38].
- Plating and Initial Culture: Resuspend the freshly isolated CD11b+ microglial cell pellet in the complete culture medium. Plate the cells at an appropriate density (e.g., (2.5 \times 10^5) cells/mL) onto the poly-D-lysine-coated vessels [38].
- Incubation and Medium Refreshment: Place the cultured cells in a humidified incubator at 37°C with 5% CO2. The growth medium may be replaced with fresh medium after a period of several hours (e.g., ≥6 hours) to remove non-adherent cells and debris, or changed according to specific experimental requirements [36] [11].
Protocol for Culturing Microglia Isolated via Percoll Gradient
This protocol is suited for microglia isolated through density gradient methods, which are often from adult or aging mice [11].
- Specialized Medium Formulation: Prepare a 1:1 mixture of DMEM/F-12 with GlutaMAX and 50% conditioned medium collected from mixed glial cultures established from the brain. This conditioned medium provides essential trophic factors supportive of microglial health [11].
- Serum and Antibiotic Supplementation: To the medium mixture, add 10% FBS and 1% penicillin/streptomycin [11].
- Growth Factor Addition for Adult Microglia: For microglia isolated from adult mice, which can be more challenging to maintain, supplement the medium with Macrophage Colony-Stimulating Factor (M-CSF; 100 ng/mL) and Granulocyte-Macrophage Colony-Stimulating Factor (GM-CSF; 100 ng/mL) 24 hours after initial plating. These factors support survival and proliferation [11].
- Plating and Maintenance Culture: Plate the Percoll-purified microglia in the prepared medium. Maintain the cultures for at least 7 days to allow cells to recover their sub-reactive morphology, with medium changes performed every 3 days [11].
The following workflow diagram illustrates the key decision points and steps in these two primary culture pathways.
The Scientist's Toolkit: Essential Reagents for Microglial Culture
Successful microglial culture requires specific reagents, each serving a distinct function in maintaining cell viability, phenotype, and adherence.
Table 2: Key Research Reagent Solutions for Microglia Culture
| Reagent / Material | Primary Function in Culture |
|---|---|
| Poly-D-Lysine | A synthetic polymer that coats the surface of culture vessels, enhancing the adhesion of primary cells like microglia by interacting with the cell membrane [36] [38]. |
| Dulbecco's Modified Eagle Medium (DMEM) | A widely used basal nutrient mixture providing essential amino acids, vitamins, and energy sources for sustaining microglial cell growth and metabolism [38]. |
| Fetal Bovine Serum (FBS) | Provides a complex mixture of proteins, growth factors, and hormones that are crucial for cell survival, proliferation, and attachment [36] [11] [38]. |
| Macrophage Colony-Stimulating Factor (M-CSF) | A growth factor that supports the survival, proliferation, and differentiation of cells in the macrophage lineage, including primary microglia [11]. |
| Granulocyte-Macrophage Colony-Stimulating Factor (GM-CSF) | A cytokine that promotes the proliferation and maintenance of microglial cells, particularly important for cultures established from adult brain tissue [11]. |
| Conditioned Medium from Mixed Glial Culture | A medium collected from heterogeneous brain cell cultures, containing a natural cocktail of secreted factors that support microglial health and mimic aspects of the in vivo microenvironment [11]. |
The journey of primary microglia from in vivo state to in vitro model is fraught with challenges, including the risk of ex vivo activation and phenotypic drift [35]. The choice between magnetic bead and Percoll isolation is the first critical step that influences the required culture conditions. Immunomagnetic separation offers high specificity and is well-paired with standard DMEM and poly-D-lysine coatings [36] [38]. In contrast, Percoll isolation, often employed for more sensitive adult microglia, frequently necessitates a more supportive environment including conditioned medium and exogenous growth factors like M-CSF and GM-CSF to ensure cell survival and maintain a representative phenotype [11]. Ultimately, there is no one-size-fits-all solution. Researchers must align their post-isolation culture strategy with their chosen isolation method and experimental goals, whether that involves maintaining a homeostatic state or probing activated responses, to ensure the validity and relevance of their findings in microglial biology.
Maximizing Success: A Guide to Troubleshooting and Optimizing Your Isolation
The isolation of primary brain cells, particularly microglia, is a cornerstone of neuroscience research, enabling the study of cellular behavior, signaling pathways, and mechanisms underlying central nervous system (CNS) disorders [1]. The pursuit of high-purity, high-yield microglial cultures is paramount for generating reliable and translatable data, especially in drug development. The choice of isolation technique critically impacts the success of downstream applications. This guide provides an objective comparison of two predominant methods—magnetic bead isolation and Percoll gradient centrifugation—focusing on their performance in isolating microglia, with a specific emphasis on how animal age and tissue source affect the critical outcomes of yield and purity. Understanding these variables allows researchers to select and optimize protocols that best suit their experimental goals, whether for high-throughput screening, functional assays, or omics studies [35] [5].
Methodological Comparison: Magnetic Bead Isolation vs. Percoll Gradient
The selection of an isolation method involves trade-offs between purity, yield, cost, technical demand, and the potential for inducing ex vivo artifacts. The table below provides a direct comparison of magnetic-activated cell sorting (MACS) and Percoll gradient centrifugation based on key performance metrics.
Table 1: Comparative Analysis of Microglia Isolation Techniques
| Feature | Magnetic Bead Isolation (MACS) | Percoll Gradient Centrifugation |
|---|---|---|
| Separation Principle | Immunoaffinity: Antibody-conjugated magnetic beads bind specific cell surface markers (e.g., CD11b) [1] [39]. | Density: Separates cells based on physical buoyancy density [1] [11]. |
| Typical Purity | High (>90% for CD11b+ cells); can be slightly contaminated with other myeloid cells [10]. | High; effective at removing myelin and cellular debris, leading to pure microglial fractions [11]. |
| Typical Yield | High; efficient positive or negative selection minimizes cell loss [10]. | Variable; can be lower due to cell loss during multiple centrifugation and washing steps [11]. |
| Cost | Higher (cost of antibodies and magnetic columns) [1]. | Lower (uses common laboratory reagents) [11]. |
| Technical Skill | Moderate; requires specific protocol steps but is relatively straightforward [11]. | Moderate; requires careful preparation of gradients to avoid cell damage [11]. |
| Speed | Fast; processing is quicker than FACS and suitable for multiple samples [10] [35]. | Slower; involves long centrifugation times which can risk cell damage [11]. |
| Ex Vivo Activation | Introduces an activation signature, primarily during tissue dissociation rather than the sorting itself [35]. | Mechanical and enzymatic dissociation can also induce activation; requires optimization to minimize [35]. |
| Key Advantage | High specificity and purity, ideal for downstream molecular applications like sequencing [10] [35]. | Lower cost, avoids antibody binding, and does not require specialized equipment like a magnet [1] [11]. |
| Key Disadvantage | Antibody binding may theoretically affect cell function, though major activation occurs during dissociation [35]. | Lower and more variable cell yield; lengthy procedure [11]. |
Diagram 1: Microglia Isolation Workflow Comparison
The Critical Role of Animal Age and Tissue Source
The age and source of the animal tissue are not mere experimental details but are deterministic factors that directly influence the yield, purity, and biological relevance of the isolated microglia.
Impact on Yield and Viability
Isolating microglia from adult or aged mice presents significant challenges compared to neonates. The yield from an aged mouse brain is considerably lower, typically around ~1 x 10^6 cells per brain (from two cortices), which is sufficient for plating in only two wells of a 12-well plate [4]. In contrast, protocols for neonatal mice consistently report higher yields. This decline is attributed to increased myelin content in the adult brain, which can interfere with isolation, and the inherent sensitivity of aged cells to the stress of the dissociation process [11]. Furthermore, microglia from aged animals often exhibit a "primed" phenotype, characterized by higher baseline expression of activation markers like CD45 and MHC II, making them more susceptible to ex vivo activation during isolation [4].
Impact on Phenotype and Functional Relevance
The use of microglia from the appropriate age group is critical for modeling age-related diseases. Aged microglia are susceptible to "inflammaging," a state of elevated baseline inflammation. They show differential gene transcription, increased phagocytic ability, and prolonged activation compared to microglia from younger adults [4]. Consequently, primary microglia isolated from neonatal mice are not fully mature and behave differently from adult microglia, making them unsuitable for studying neurodegenerative diseases where aging is a primary risk factor [11] [5]. For research on conditions like Alzheimer's disease, which primarily affects individuals over 65, the use of microglia from mice aged 18 months or older is considered most appropriate [4].
Table 2: Impact of Animal Age on Microglial Isolation and Characteristics
| Characteristic | Neonatal (Postnatal Day 1-10) | Adult (2-6 Months) | Aged (18+ Months) |
|---|---|---|---|
| Typical Yield | High [11] | Moderate | Low (~1x10^6 per brain) [4] |
| Ease of Isolation | Easy; less myelin, higher cell viability [11]. | More challenging; requires optimized protocols. | Most challenging; low proliferation capacity, high sensitivity [4]. |
| Baseline Activation | Low | Homeostatic | Elevated ("Inflammaging") [4] |
| Phenotypic State | Developmentally immature [11] [5]. | Fully mature, homeostatic. | Primed, dysregulated, involved in neurodegeneration [4]. |
| Best Suited For | Developmental studies, high-yield needs. | General adult brain physiology and disease. | Modeling age-related neurodegenerative diseases [4] [11]. |
Optimizing Protocols for Enhanced Performance
Detailed Experimental Protocols
To achieve consistent results, adherence to a well-optimized protocol is essential. Below are detailed methodologies for the two compared isolation techniques, incorporating key optimizations.
Immunomagnetic Bead Isolation (MACS) for Microglia
This protocol can be used to sequentially isolate microglia, astrocytes, and neurons from the same mouse brain, maximizing the utility of a single tissue sample [1] [39].
- Tissue Dissociation: Euthanize the mouse and rapidly remove the brain. Dissect the desired region (e.g., cortex, hippocampus) and remove the meninges thoroughly. Mechanically mince the tissue into small fragments using a sterile scalpel.
- Enzymatic Digestion: Incubate the tissue fragments in an enzyme solution containing papain (2.5 U/mL) and DNase (10 U/mL) in Hibernate A medium for 15 minutes at 37°C with continuous rotation [5]. Triturate the tissue 10-20 times with a fire-polished pipette to create a single-cell suspension.
- Cell Suspension Preparation: Pass the suspension through a 70 μm cell strainer. Centrifuge the filtrate at 160-500 x g for 5 minutes. Resuspend the cell pellet in a buffer such as PBS containing 0.5% BSA and 2 mM EDTA.
- Magnetic Labeling and Separation: Incubate the cell suspension with CD11b (for microglia) or ACSA-2 (for astrocytes) antibody-conjugated magnetic beads for 15 minutes at 4°C [1] [39]. Place the sample in a magnetic field separator. The CD11b+ microglia will be retained in the column, while the negative fraction (containing astrocytes and neurons) passes through.
- Washing and Elution: After extensive washing, remove the column from the magnetic field and flush out the positively selected microglia. The negative fraction can be subsequently used for the isolation of other cell types, such as astrocytes using ACSA-2 beads [1].
Density Gradient Isolation Using Percoll
This is a cost-effective, equipment-light method that avoids the use of antibodies [1] [11].
- Initial Steps: Perform steps 1-3 from the MACS protocol to obtain a single-cell suspension.
- Gradient Preparation: Create a discontinuous density gradient. A common and effective setup involves carefully layering the cell suspension (or resuspended cells in a low-density solution) over a higher density Percoll solution (e.g., 70%) [11]. Alternatively, a single 30% Percoll gradient can be used for a more balanced recovery of multiple neural cell types, including microglia and astrocytes [31].
- Centrifugation: Centrifuge the gradient at high speed (e.g., 1400 x g) for 15-30 minutes at 4°C with the brake disengaged to prevent disturbing the gradient [11] [31].
- Cell Collection: After centrifugation, microglia will form a distinct band at the interface between the two densities. Carefully collect this band using a Pasteur pipette.
- Washing: Transfer the collected cell fraction to a new tube, add at least 3-5 volumes of PBS or culture medium, and centrifuge at 500 x g for 5-10 minutes to remove the Percoll. Resuspend the purified microglial pellet in the desired medium.
Minimizing Ex Vivo Confounds
A significant concern in microglial isolation is the introduction of ex vivo activation during the dissociation process, which can confound transcriptomic and functional analyses [35]. Research shows that this activation arises primarily during the enzymatic and mechanical tissue dissociation, not during the subsequent sorting step (whether MACS or FACS) [35]. Two key strategies to minimize this are:
- Inhibition of Transcription/Translation: Supplementing the dissociation and washing buffers with inhibitors of transcription (e.g., Actinomycin D) and translation (e.g., Anisomycin) can effectively prevent the induction of an ex vivo activation signature [35].
- Cold Non-Enzymatic Dissociation: As an alternative, performing tissue dissociation using non-enzymatic, mechanical methods entirely at 4°C has been shown to successfully prevent ex vivo activation artifacts [35].
The Scientist's Toolkit: Essential Research Reagents
Successful isolation and culture of microglia depend on a set of key reagents. The following table lists essential materials and their functions based on the protocols analyzed.
Table 3: Key Reagents for Microglia Isolation and Culture
| Reagent / Material | Function / Purpose | Example Protocols |
|---|---|---|
| Papain | Proteolytic enzyme for gentle tissue dissociation and cell liberation [5] [31]. | Used in combination with DNase for human and mouse brain dissociation [5] [31]. |
| CD11b MicroBeads | Antibody-conjugated magnetic beads for specific immunocapture of microglia via the CD11b surface marker [1] [39]. | Used in MACS protocols for positive selection of microglia from a mixed brain cell suspension [1] [39]. |
| Percoll | Colloidal silica solution used to form density gradients for the physical separation of cells based on size and density [1] [11]. | Used in 30%/70% or single 30% gradients to isolate microglia and remove myelin debris [11] [31]. |
| Poly-d-lysine (PDL) | Synthetic polymer used to coat culture surfaces, promoting adhesion of primary cells like microglia and neurons [4]. | Used to coat plates or flasks before seeding isolated adult mouse microglia [4]. |
| M-CSF / GM-CSF | Macrophage and Granulocyte-Macrophage Colony-Stimulating Factors; cytokines added to culture media to support survival and proliferation of primary microglia [11]. | Added to the medium for culturing microglia isolated from adult mice [11]. |
Diagram 2: Factors Determining Isolation Success
The optimal isolation of microglia requires a carefully balanced strategy that aligns the choice of technique (MACS vs. Percoll) with the biological context of the study, particularly the age of the animal and the required balance between yield and purity. Magnetic bead isolation offers superior speed and purity for sequencing and other molecular applications, while Percoll gradients provide a cost-effective alternative that avoids antibody binding. Critically, the use of age-appropriate tissue sources is non-negotiable for modeling adult neurodegenerative diseases. By integrating the optimized protocols and artifact-minimization strategies outlined in this guide—such as the use of transcriptional inhibitors or cold non-enzymatic dissociation—researchers can significantly enhance the quality, reliability, and physiological relevance of their primary microglial cultures, thereby strengthening the translational potential of their findings in drug development and basic neuroscience.
The isolation of pure and viable microglia is a cornerstone of neuroscience research, enabling the study of neuroinflammation, neurodegenerative diseases, and cellular mechanisms. The choice between magnetic bead isolation and Percoll gradient methods often involves a critical trade-off between cell purity and cell viability. A primary source of low cell viability across these protocols is the stress induced by enzymatic digestion and mechanical dissociation during the initial creation of a single-cell suspension from brain tissue. This guide objectively compares the performance of different isolation methodologies, focusing on how they mitigate or exacerbate these stressors, to inform researchers and drug development professionals.
Key Stressors in Microglia Isolation
The Impact of Enzymatic Digestion
Enzymatic digestion, typically using papain or trypsin, is widely used to dissociate brain tissue. However, this process subjects cells to significant stress.
- Transcriptomic Alterations: Procedures using enzymatic dissociation at 37°C introduce drastic, confounding changes in gene expression profiles, potentially masking the true in vivo state of the microglia [40].
- Induced Activation: The enzymatic and mechanical dissociation process itself can cause ex vivo microglial activation, an artifact that may interfere with studying genuine disease-related activation states [35].
The Role of Mechanical Stress
Mechanical disruption—through chopping, trituration, or homogenization—is necessary to break down tissue structure but can physically damage cells.
- Cell Damage and Death: Harsh mechanical processing directly compromises cell membrane integrity, reducing viability and yield [35]. This is a particular concern when isolating fragile cells or cells from aged models [11].
- Myelin Contamination: Mechanical disruption can release large amounts of myelin, which interferes with downstream applications and can require additional, potentially stressful, cleaning steps [11].
Comparative Analysis of Isolation Method Performance
The following table summarizes quantitative data on the performance of different isolation strategies in relation to these stressors.
Table 1: Performance Comparison of Microglia Isolation Methods
| Isolation Method | Reported Purity | Impact on Viability & Transcriptome | Key Advantages | Key Disadvantages |
|---|---|---|---|---|
| Enzymatic Dissociation (Standard) [5] [21] | >90% (with CD11b+ selection) | Induces significant ex vivo activation and transcriptomic changes [35] [40]. | High efficiency and purity; well-established protocol. | Potential alteration of cell surface markers; lower transcriptomic fidelity. |
| Magnetic-Activated Cell Sorting (MACS) [1] [10] | High (Microglia: >90%) | Activation occurs during tissue dissociation prior to sorting [35]. Yields high viability (>85%) [10]. | Fast processing; suitable for multiple samples; high cell yield [10] [35]. | Potential for slight myeloid cell contamination [10]. |
| Fluorescence-Activated Cell Sorting (FACS) [10] [35] | Potentially higher than MACS [10] | Yields high viability (>85%) and the purest population for sequencing [10]. Activation from dissociation, not sorting [35]. | Highest purity; minimal contamination. | Slower than MACS; requires specialized, expensive equipment [10]. |
| Percoll Gradient [1] [11] | High | Avoids enzymatic stress when using non-enzymatic protocols [1]. Effective myelin debris removal [11]. | Lower cost; avoids expensive antibodies [1]. | Long centrifugation can cause cell damage and low yield [11]. |
| Mechanical Homogenization (at 4°C) [40] | High (suitable for sequencing) | Preserves transcriptomic integrity by avoiding heated enzymatic digestion [40]. | Minimal ex vivo activation; high transcriptomic fidelity. | May require subsequent FACS or Percoll step for purification [40]. |
Table 2: Experimental Data on Strategies to Minimize Isolation Stress
| Experimental Approach | Key Experimental Findings | Implication for Cell Viability & Function |
|---|---|---|
| Inhibitor Cocktail [35] | Supplementing dissociation media with transcriptional/translational inhibitors prevented most ex vivo activation signatures. | Preserves native transcriptional state, improving data accuracy for genomic studies. |
| Non-Enzymatic, Cold Dissociation [35] [40] | Mechanical homogenization at 4°C successfully prevented the ex vivo activational signature. | Maintains a microglial profile closer to the in vivo state, maximizing transcriptomic integrity. |
| Protocol for Aged Mice [11] | A modified protocol focusing on gentle mechanical and enzymatic techniques provided an optimal yield of functional microglia. | Critical for studying age-related diseases, as microglia from aged brains are particularly susceptible to isolation stress. |
Detailed Experimental Protocols
This protocol is designed to avoid the transcriptional artifacts introduced by enzymatic digestion.
- Tissue Collection: Euthanize mouse and rapidly remove the brain.
- Homogenization: Place the brain tissue in a Dounce homogenizer with cold buffer. Perform gentle mechanical homogenization entirely at 4°C.
- Cell Suspension Filtering: Filter the resulting homogenate through a 70 µm cell strainer to remove clumps.
- Microglia Isolation: Isolate microglia from the single-cell suspension using a Percoll density gradient followed by flow cytometry sorting based on cell-surface markers (e.g., CD11b).
A common protocol for obtaining high microglial yield, with notes on minimizing artifacts.
- Tissue Collection and Dicing: Perfuse mouse with ice-cold saline, remove the brain, and mechanically dice it into small fragments.
- Enzymatic Digestion: Incubate tissue fragments in an enzyme solution (e.g., papain 2.5 U/mL and DNase 10 U/mL) for 15 minutes at 37°C with continuous rotation.
- Mechanical Trituration: Triturate the tissue-enzyme mixture ~20 times with a stripette to further dissociate cells.
- Reaction Termination and Filtering: Inactivate the protease, and pass the homogenate through a 70 µm cell strainer.
- Microglia Isolation: Centrifuge the cell suspension and proceed with your chosen primary isolation method (MACS or Percoll).
To minimize artifacts in this protocol, researchers can incorporate the inhibitor cocktail suggested by [35] during the dissociation steps.
Visualizing the Experimental Workflows
The diagram below contrasts the two primary experimental workflows for microglia isolation, highlighting key steps where stress occurs and mitigation strategies can be applied.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents for Microglia Isolation and Culture
| Reagent / Kit | Function in Protocol | Specific Example |
|---|---|---|
| Papain & DNase [5] | Enzymatic digestion of intercellular proteins and DNA to dissociate tissue. | 2.5 U/mL papain with 10 U/mL DNase in Hibernate A media [5]. |
| Neural Tissue Dissociation Kit [21] | Commercial kit providing optimized enzymes for brain tissue dissociation. | Used according to manufacturer's instructions (Miltenyi Biotec) [21]. |
| Percoll [1] [11] | Density gradient medium for separating microglia based on buoyancy; removes myelin and debris. | Used in a standardized density gradient protocol [1] [11]. |
| CD11b Microbeads [1] [21] | Magnetic beads conjugated to anti-CD11b antibodies for positive selection of microglia via MACS. | Incubation of cell suspension with CD11b microbeads [1]. |
| M-CSF & GM-CSF [11] [21] | Growth factors added to culture media to support survival and proliferation of primary microglia. | 100 ng/mL of each factor for adult microglia cultures [11] [21]. |
| Transcriptional/Translational Inhibitors [35] | Cocktail added during dissociation to prevent artifactual activation and preserve native gene expression. | e.g., Actinomycin D (transcription) and Anisomycin (translation) [35]. |
The choice between magnetic bead isolation and Percoll gradients is intrinsically linked to the initial steps of tissue dissociation. Enzymatic digestion, while efficient for high yield, is a major contributor to low viability and transcriptomic artifacts. Mechanical stress during dissociation further compounds this problem. For studies where preserving the native transcriptional state of microglia is paramount, such as in single-cell RNA sequencing or functional phenotyping, mechanical homogenization at 4°C coupled with a Percoll gradient or FACS offers a superior path. Conversely, for applications requiring high cell numbers where some activation is acceptable, optimized enzymatic protocols with MACS sorting remain a viable and efficient option. Ultimately, aligning the isolation strategy with the experimental question—and rigorously applying stress-mitigation techniques—is essential for generating reliable and translatable data in microglia research.
The study of microglia, the resident immune cells of the central nervous system, is crucial for understanding brain homeostasis, development, and neurodegenerative diseases. A significant challenge in this field is the prevention of unwanted microglial activation during isolation and culture, as their ex vivo activation can confound experimental results and lead to misleading conclusions. This activation is profoundly influenced by culture duration and the use of specific growth factors. This guide objectively compares two common microglia isolation techniques—immunomagnetic sorting (MACS) and Percoll gradient centrifugation—within the broader research context of maximizing microglial purity and minimizing activation artifacts. The performance of these methods is evaluated based on experimental data including cell yield, purity, viability, and the expression of activation markers.
Experimental Protocols for Microglia Isolation
Immunomagnetic Cell Sorting (MACS) Protocol
The MACS protocol utilizes magnetic beads conjugated with antibodies against specific microglial surface markers, such as CD11b, for positive selection [6].
- Timing: Approximately 5 days for the entire culture and isolation process [6].
- Key Steps:
- Cell Suspension Preparation: A single-cell suspension is prepared from brain tissue via enzymatic and mechanical dissociation.
- Magnetic Labeling: The cell suspension is resuspended in MACS buffer and incubated with anti-CD11b magnetic beads at 4°C for 15 minutes [6].
- Magnetic Separation: The labeled cell suspension is applied to a MS column placed in a magnetic field. Unlabeled cells pass through, while microglia are retained.
- Elution: The column is removed from the magnetic field, and the positively selected CD11b⁺ microglia are flushed out [6].
- Culture: Isolated microglia are pelleted and resuspended in specialized astrocyte-conditioned growth medium (AGM) or similar, often supplemented with macrophage colony-stimulating factor (M-CSF) to support survival [4] [11].
Percoll Gradient Centrifugation Protocol
This is a density-based method that separates microglia from other neural cells without relying on antibody binding [1] [11].
- Timing: The isolation process itself is generally faster than MACS, though tissue dissociation time is similar.
- Key Steps:
- Tissue Dissociation: Brain tissue is dissociated into a single-cell suspension.
- Gradient Preparation: A discontinuous density gradient is prepared using Percoll solutions.
- Centrifugation: The cell suspension is layered onto the gradient and centrifuged. Microglia, which have a specific density, migrate to the interface between specific Percoll concentrations while myelin and other debris are removed.
- Cell Collection: The microglial layer is carefully collected from the gradient.
- Culture: Cells are washed, counted, and cultured in medium supplemented with growth factors such as M-CSF and granulocyte-macrophage colony-stimulating factor (GM-CSF) to promote proliferation and maintain function [11].
Performance Comparison: Magnetic Beads vs. Percoll Gradient
The following tables summarize key performance metrics and functional characteristics of microglia isolated using MACS and Percoll gradient methods, based on published experimental data.
Table 1: Quantitative Comparison of Yield, Purity, and Viability
| Performance Metric | Magnetic Bead (MACS) Isolation | Percoll Gradient Isolation | Supporting Experimental Data |
|---|---|---|---|
| Cell Yield | ~1-3 x 10⁶ cells per adult mouse brain [6] [4] | Higher yield compared to some MACS protocols [11] | A modified protocol achieved significantly higher yield than two other methods [11]. |
| Purity (CD11b⁺) | High (>90% reported in some studies) [10] | High, but may have slight contamination [10] | MACS can have slight myeloid cell contamination; FACS can yield purer populations [10]. |
| Cell Viability | High (>85%) [10] | High (>85%) [10] | Both methods demonstrate high viability post-isolation [10]. |
| Processing Speed | Faster for single or multiple samples [10] [35] | Requires long centrifugation steps [11] | MACS processing time is generally faster than FACS or lengthy gradient centrifugations [10] [35]. |
Table 2: Comparison of Functional and Activation Characteristics
| Characteristic | Magnetic Bead (MACS) Isolation | Percoll Gradient Isolation | Supporting Experimental Data |
|---|---|---|---|
| Unwanted Activation | Moderate; primarily from tissue dissociation, not the sort itself [35] | Moderate; enzymatic digestion can induce activation [11] [35] | Ex vivo activation occurs mainly during initial tissue dissociation, not during the sorting step [35]. |
| Effect of Culture Duration | Morphology changes shortly after purification; experiments should be performed quickly [1] | Cells require ~7 days in culture to recover a sub-reactive morphology [11] | Cells start to change morphology shortly after MACS purification [1]. |
| Effect of Growth Factors | Often uses M-CSF in culture medium [4] | Uses M-CSF and GM-CSF to support growth and function [11] | M-CSF and GM-CSF are added to the medium to support microglial culture [11]. |
| Key Advantages | High purity, standardized kits, fast processing [10] | Avoids antibody binding, cost-effective for equipment [1] [11] | Percoll circumvents expensive antibodies and enzymatic digestion that can affect viability [1]. |
Signaling Pathways and Experimental Workflows
The isolation process and subsequent culture conditions directly influence key intracellular signaling pathways in microglia, such as the SYK-PLCγ2 pathway, which is critical for their response to environmental cues and can be associated with both wanted and unwanted activation [41].
Microglia Signaling Pathway
The workflow below outlines the critical steps for both isolation methods and highlights key points where interventions can minimize ex vivo activation.
Microglia Isolation Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
The following table details essential reagents and their functions for successfully isolating and culturing microglia while minimizing activation.
Table 3: Essential Reagents for Microglia Isolation and Culture
| Reagent / Kit | Function / Application | Specific Example |
|---|---|---|
| Anti-CD11b Microbeads | Magnetic beads for positive selection of microglia via MACS [6]. | Miltenyi Biotec CD11b Microbeads (human and mouse) [6]. |
| Percoll | Density gradient medium for separating cells based on buoyancy; used to isolate microglia and remove myelin debris [1] [11]. | Cytiva Percoll [1]. |
| M-CSF (Macrophage Colony-Stimulating Factor) | Growth factor critical for microglial survival, proliferation, and maintenance in culture [4] [11]. | Recombinant Mouse M-CSF [11]. |
| GM-CSF (Granulocyte-Macrophage Colony-Stimulating Factor) | Growth factor used alongside M-CSF to support microglial growth and function in vitro [11]. | Recombinant Mouse GM-CSF [11]. |
| Astrocyte-Conditioned Medium (AGM) | Specialized medium collected from astrocyte cultures, containing supportive factors for microglial health [6]. | Prepared in-lab from primary astrocyte cultures. |
| Transcriptional/Translational Inhibitors | Cocktails added during tissue dissociation to suppress rapid gene expression changes and prevent ex vivo activation artifacts [35]. | Actinomycin D (transcription inhibitor), Cycloheximide (translation inhibitor) [35]. |
Both magnetic bead isolation and Percoll gradient centrifugation are effective for obtaining viable, high-purity microglia. The choice between them depends on specific research priorities. MACS offers superior speed and standardized purity, making it ideal for high-throughput studies where antibody binding is not a concern. Conversely, the Percoll gradient is a cost-effective alternative that avoids potential activation from antibody binding, though it requires careful handling to prevent mechanical stress.
Critically, this comparison shows that unwanted activation is primarily induced during the initial tissue dissociation phase, regardless of the subsequent isolation method [35]. Therefore, the most effective strategy for preventing artifacts involves optimizing pre-isolation steps. Researchers can successfully preserve native microglial states by employing transcriptional/translational inhibitors, using gentle, low-temperature dissociation protocols, carefully managing culture duration, and supplementing media with essential growth factors like M-CSF. This holistic approach ensures that experimental outcomes more accurately reflect in vivo microglial biology.
Flow cytometry analysis of central nervous system (CNS) tissues presents unique challenges due to the abundance of myelin, a lipid-rich substance that can obscure detection and accurate quantification of target cells, particularly microglia and other immune populations [30] [29]. Effective myelin removal is therefore a critical preparatory step that significantly impacts cell yield, viability, and the reliability of downstream applications. This guide provides an objective comparison of the most common myelin removal methods—Percoll and sucrose density gradients, along with commercial debris removal solutions—within the broader context of microglia isolation research. As the field increasingly focuses on obtaining high-purity microglial populations for sophisticated applications like single-cell sequencing and proteomic analysis, the choice between magnetic bead isolation and Percoll gradient centrifugation becomes particularly relevant [10] [11]. We synthesize experimental data from recent studies to help researchers and drug development professionals select the most appropriate method for their specific research objectives.
Method Comparison at a Glance
The table below summarizes the key characteristics, advantages, and limitations of the primary myelin removal methods used in CNS research.
Table 1: Comprehensive Comparison of Myelin Removal Methods
| Method | Principle of Separation | Reported Advantages | Reported Limitations |
|---|---|---|---|
| Percoll Gradient | Density-based centrifugation using silica particles [30] | - Superior removal of non-immune cells and myelin debris [30] [29]- Results in high microglial viability and yield [12]- Considered the "gold standard" for microglia isolation [11] | - Requires optimization of concentration (typically 24-30%) [29]- Longer centrifugation process [11] |
| Sucrose Gradient | Density-based centrifugation using sucrose solution [30] | - Simpler and cheaper reagent [30]- Provides comparable microglial yield to Percoll in some studies [30] | - Less effective at removing non-immune cells compared to Percoll [30]- May result in lower cell viability [12] |
| Commercial Kits (e.g., Myelin Removal Beads, OptiPrep) | Immunomagnetic binding or ready-made density gradients [42] [12] | - Standardized and convenient protocol [12]- Effective for sensitive tissues like injured spinal cord [42] | - Higher cost per sample [30]- May require specialized columns or buffers [12] |
| No Myelin Removal | Omits the density separation step [43] | - Fastest and simplest protocol [43]- Avoids potential cell loss during gradient steps [43] | - Myelin debris interferes with flow cytometry accuracy and sensitivity [42] [29]- Not suitable for precise immune cell quantification [42] |
Detailed Experimental Protocols
Percoll Density Gradient Protocol
The Percoll method is widely regarded as the most effective for achieving a balance of high microglial yield and purity [30] [12] [11].
- Solution Preparation: Prepare a stock isotonic Percoll (SIP) solution. For myelin removal, concentrations between 24% and 30% SIP are recommended. Studies indicate that 24% SIP is sufficient to remove myelin debris without compromising cell yield, while higher concentrations (30%) are commonly used for microglia isolation [30] [29].
- Sample Preparation: Perform enzymatic digestion of brain tissue using a suitable enzyme (e.g., accutase, trypsin, or papain) to create a single-cell suspension [30] [29].
- Centrifugation: Resuspend the cell pellet in the prepared SIP solution. Centrifuge at 700-726 x g for 10-15 minutes at 20°C [30] [12].
- Cell Collection: After centrifugation, myelin forms a layer on top of the solution. Carefully aspirate this supernatant. The pellet contains the cells, which should be washed with a buffer like HBSS before downstream applications [30] [42].
Sucrose Density Gradient Protocol
The sucrose protocol offers a cost-effective alternative, though it may be less effective in removing non-immune cells [30].
- Solution Preparation: Prepare a 0.9 M sucrose solution in HBSS [30] [12].
- Sample Preparation: Generate a single-cell suspension from enzymatically digested brain tissue.
- Centrifugation: Resuspend the cell pellet in the sucrose solution and centrifuge at 700 x g for 10 minutes [12].
- Cell Collection: Aspirate the supernatant containing myelin and collect the cell pellet for washing and analysis [30].
Commercial Kits and OptiPrep Protocol
Commercial kits, including immunomagnetic myelin removal beads and OptiPrep gradients, provide standardized and effective alternatives [42] [12].
- Immunomagnetic Beads:
- Incubate the single-cell suspension with anti-myelin magnetic beads.
- Pass the sample through a magnetic column. Myelin-bound beads are retained in the column, while purified cells are collected in the effluent [12].
- OptiPrep Gradient:
- Create a discontinuous gradient by layering solutions of decreasing density (e.g., 350, 250, 200, and 150 μL of diluted OptiPrep per 1 mL) in a conical tube.
- Layer the dissociated cell suspension on top and centrifuge at 726 x g for 15 minutes.
- Aspirate the top layer containing myelin debris. Inflammatory cells are primarily found in the pellet [42].
The following diagram illustrates the core decision-making workflow for selecting and implementing these key myelin removal methods.
Quantitative Performance Data
Microglia Yield and Viability
Direct comparisons reveal how the choice of myelin removal method impacts critical outcome measures. A systematic study testing different protocols on adult mouse brains found that while Percoll and sucrose yielded a comparable number of microglia (CD11b+CD45int cells), Percoll was significantly more effective at removing non-immune cells from the sample [30].
Table 2: Experimental Outcomes by Myelin Removal Method
| Method | Microglial Yield | Cell Viability | Purity / Effectiveness |
|---|---|---|---|
| Percoll | Comparable high yield to sucrose [30] | High viability (>85%) [12] [10] | Superior removal of non-immune cells and myelin debris [30] [29] |
| Sucrose | Comparable high yield to Percoll [30] | Lower viability compared to Percoll [12] | Less effective in removing non-immune cells [30] |
| Commercial Beads | Not specified | Not specified | Effective myelin removal, but lower yield of CD11b+ cells vs. Percoll [12] |
| No Removal | N/A | N/A | Myelin debris obscures flow cytometry, drastically reduces sensitivity [42] |
Impact on Downstream Applications
The method of myelin removal can influence the success of subsequent analyses:
- Flow Cytometry: The presence of myelin debris severely compromises flow cytometry analysis by creating events that can be mistaken for cells, leading to inaccurate quantification. One study demonstrated that the percentage of polymorphonuclear leukocytes (PMNs) detected in injured spinal cord samples without myelin removal was only a fraction (0.5%) of what was detected (5.1%) in samples purified with an OptiPrep gradient [42].
- Cell Culture and Phenotyping: Effective myelin removal is crucial for culturing microglia with preserved phenotypes. Protocols using Percoll gradients yield microglia that accurately reflect their in vivo state, allowing for proper observation of activation, as seen in responses to lipopolysaccharide (LPS) challenge [12] [11].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Myelin Removal and Microglia Isolation
| Reagent / Kit | Primary Function | Example Use Case |
|---|---|---|
| Percoll | Density gradient medium for separation of cells from myelin debris [30] [29] | Standard high-purity microglia isolation from adult mouse brain [30] [11] |
| Sucrose | Low-cost density gradient medium [30] | Protocol comparison studies; budget-conscious isolation with acceptable yield [30] [12] |
| Anti-Myelin Magnetic Beads | Immunomagnetic removal of myelin debris [12] | Standardized myelin clearance without centrifugation gradients [12] |
| OptiPrep | Ready-made density gradient medium [42] | Sensitive tissue like injured spinal cord; improves flow cytometry sensitivity [42] |
| Enzymes (Accutase, Trypsin, Papain) | Tissue dissociation to create single-cell suspensions [30] [29] | Essential first step before any myelin removal protocol [30] |
| CD11b Microbeads | Positive selection of microglia via immunomagnetic separation [12] [10] | Obtaining highly pure microglial populations after myelin removal [12] |
The choice of myelin removal method is a critical determinant in the success of CNS cell isolation protocols. Based on the comparative data:
- For research requiring the highest standards of microglial purity, viability, and accurate flow cytometry quantification, Percoll density gradient centrifugation is the recommended and most reliable method. Its superiority in removing non-immune cells and myelin debris makes it the gold standard for rigorous investigations [30] [29].
- Sucrose gradients present a viable, cost-effective alternative for experiments where the primary focus is on microglial yield and ultimate purity is less critical, acknowledging its lower effectiveness in removing non-immune cells [30].
- Commercial kits, including myelin removal beads and OptiPrep, offer standardization and convenience and are particularly valuable for processing sensitive tissues like the injured spinal cord or for laboratories seeking to minimize protocol variability [42] [12].
Ultimately, the optimal choice depends on the specific research question, tissue type, and downstream application. However, the body of evidence strongly supports the use of Percoll gradients in the context of microglia isolation for high-purity research applications, including those comparing magnetic bead isolation with traditional density gradient methods [30] [10] [11].
Head-to-Head Comparison: Purity, Phenotype, and Functional Outcomes
For neuroscientists studying neuroinflammation, neurodegeneration, and central nervous system homeostasis, the isolation of pure microglial populations is a critical technical prerequisite. The choice between immunomagnetic separation (using magnetic beads) and density gradient centrifugation (using Percoll) represents a fundamental methodological crossroads, with direct implications for experimental outcomes and resource allocation [1] [44]. This guide provides an objective, data-driven comparison of these two predominant techniques, focusing on the critical metrics of cell purity and yield that most significantly impact downstream applications. By synthesizing experimental evidence from recent protocols, we aim to equip researchers with the evidence needed to select the most appropriate isolation strategy for their specific research context.
Quantitative Data Comparison: Purity, Yield, and Viability
The following tables consolidate quantitative findings from published protocols to facilitate direct comparison of performance metrics.
Table 1: Comparison of Overall Microglial Purity and Yield
| Isolation Method | Reported Purity (%) | Reported Yield (Cells per Brain) | Key Markers Used for Validation | Reference |
|---|---|---|---|---|
| Immunomagnetic Beads (CD11b+) | >95% [12] | ~3 × 10^6 (Adult mouse) [6] | CD11b, CD45, F4/80 [45] | [45] [6] |
| Percoll Density Gradient | Highly purified population [13] | Higher yield vs. magnetic beads in one study [6] | CX3CR1, Siglec-H [13] | [6] [13] |
| Modified Protocol (Combination) | High purity [11] | ~500,000 (6-month-old mouse) [11] | CD11b (ICC) [11] | [11] |
Table 2: Comparison of Critical Functional Metrics
| Metric | Immunomagnetic Beads | Percoll Gradient |
|---|---|---|
| Cell Viability | Preserved phenotype reported [12] | Highest viability and cell number in a direct comparison [12] |
| Phenotype Preservation | Accurately reflects in vivo state, including for activated microglia [12] | Phenotype comparable to other methods; isolation procedure itself does not activate microglia [12] |
| Downstream Applications | Flow cytometry, cell culture, stimulation/phagocytosis assays, -omics profiling [45] | Suitable for cell culture and functional assays; provides healthy cells for culture [13] |
| Technical Notes | Potential for magnetic bead retention altering downstream experiments [13] | Effective myelin removal is a critical step [45] |
Detailed Experimental Protocols
A clear understanding of the underlying methodologies is crucial for interpreting comparative data.
Immunomagnetic Bead Separation Protocol
The immunomagnetic approach uses antibodies to selectively target and separate microglia. A typical protocol, as described by Springer Nature, involves these key steps [45]:
- Tissue Dissociation: The perfused adult mouse brain is minced and subjected to enzymatic digestion using a cocktail containing collagenase D, TLCK, DNaseI, and dispase in HBSS for 1 hour at 37°C under gentle rotation [45].
- Mechanical Homogenization: The digested tissue is further dissociated on ice using a Dounce homogenizer (20 passes with the large-clearance pestle) [45].
- Myelin Removal: The cell suspension is mixed with 35% isotonic Percoll and centrifuged. The supernatant containing myelin is aspirated, and the pelleted mixed brain cells are collected [45].
- Magnetic Labeling and Separation: The cell pellet is resuspended in separation buffer and incubated with anti-CD11b MicroBeads for 15 minutes at 4°C. The cell-bead suspension is then applied to an LS column placed in a magnetic field. The CD11b-negative fraction flows through, while the CD11b-positive microglia are retained and subsequently eluted [45].
Percoll Density Gradient Protocol
The Percoll method separates cells based on their inherent buoyant density. A novel protocol from PMC provides a robust example [13]:
- Tissue Dissociation and Homogenization: The mouse brain (cerebellum and brain stem often removed) is harvested and dissociated using a mechanical dissociator (e.g., gentleMACS Octo Dissociator with Heaters) with specific enzyme mixes. The resulting homogenate is further processed in a Potter-Elvehjem tissue grinder to increase yield [13].
- Density Gradient Centrifugation: The homogenized cell suspension is filtered through a 70 μM strainer. A two-layer discontinuous Percoll gradient is prepared—typically 2 mL of 70% Percoll at the bottom, carefully overlaid with 10 mL of the cell suspension mixed with a lower concentration of Percoll (e.g., Stock Isotonic Percoll) [13].
- Microglial Collection: The tube is centrifuged (e.g., 800 × g for 45 minutes with no brake). Microglia, which have a higher density than myelin and other neural cells, form a distinct pellet at the bottom of the tube. The supernatant and myelin layer are aspirated, and the pelleted microglia are washed and resuspended for culture or analysis [13].
The Scientist's Toolkit: Essential Research Reagents
Successful microglial isolation relies on a core set of reagents and tools. The table below details these essential components and their functions.
Table 3: Key Reagent Solutions for Microglia Isolation
| Reagent/Kit | Function in Protocol | Specific Example |
|---|---|---|
| Enzyme Dissociation Kit | Digests extracellular matrix and intercellular proteins to create single-cell suspension. | Neural Tissue Dissociation Kit (P) (Miltenyi Biotec) [13] |
| Percoll | Forms density gradient for separation of microglia (higher density) from myelin and debris (lower density). | Isotonic Percoll prepared with 10x HBSS [45] |
| Immunomagnetic Beads | Binds to microglial surface markers (e.g., CD11b) for antibody-based positive selection. | CD11b MicroBeads, human/mouse (Miltenyi Biotec) [45] |
| Separation/MACS Buffer | Provides the appropriate ionic and protein environment for antibody binding and magnetic separation. | PBS, pH 7.2, 0.5% BSA (low endotoxin), 2 mM EDTA [45] |
| Cell Culture Medium | Supports survival, growth, and maintenance of isolated microglia in vitro. | DMEM/F-12 with GlutaMAX, 10% FBS, antibiotics; supplemented with M-CSF/GM-CSF [11] |
The direct comparison of purity and yield data reveals that both immunomagnetic bead separation and Percoll density gradient centrifugation are capable of yielding highly pure microglia for research. The choice is ultimately context-dependent. Immunomagnetic separation offers exceptional purity and straightforward positive selection, making it ideal for transcriptomic studies and assays requiring minimal contamination from other neural cells. In contrast, the Percoll gradient method demonstrates a compelling advantage in cell viability and yield, is less expensive, and avoids potential activation from antibody binding, making it superior for primary cell culture and functional assays where large numbers of healthy cells are paramount. Researchers must therefore weigh the importance of ultimate purity against total yield and viability for their specific downstream applications.
For neuroscientists studying microglia, the resident immune cells of the central nervous system, obtaining a pure cell population that accurately reflects in vivo biology is paramount. The process of isolating these sensitive cells from brain tissue can inadvertently activate them, altering their transcriptional profile and potentially confounding research outcomes [35]. This guide provides an objective comparison of the two predominant isolation techniques—magnetic bead isolation and Percoll gradient centrifugation—focusing on their impact on the baseline gene expression crucial for accurate transcriptional profiling.
Methodological Principles and Protocols
Magnetic-Activated Cell Sorting (MACS)
MACS utilizes antibody-conjugated magnetic beads targeting specific cell surface markers, such as CD11b for microglia, to separate cells under a magnetic field. The sequential "tandem protocol" allows isolation of multiple cell types from the same brain tissue sample: microglia (CD11b+), followed by astrocytes (ACSA-2+), and finally neurons through negative selection [1].
A critical methodological consideration is that enzymatic and mechanical dissociation during cell preparation primarily drives ex vivo activation signatures, not the sorting process itself [35]. Supplementing dissociation buffers with transcriptional and translational inhibitors effectively minimizes these artifactual gene expression changes [35].
Percoll Gradient Centrifugation
Percoll gradient isolation is a density-based method that separates cells according to their buoyancy without requiring specific antibodies. This approach effectively removes myelin debris—a significant advantage when working with adult brain tissue [1] [12].
Recent protocol refinements emphasize mechanical homogenization exclusively at low temperatures (4°C) to completely eliminate heat-induced enzymatic artifacts [40]. This "cold mechanical" method preserves transcriptomic integrity, making it particularly suitable for RNA sequencing studies [40].
The workflow diagram below illustrates the key steps and decision points in microglia isolation for transcriptional profiling:
Performance Comparison: Experimental Data
Direct methodological comparisons reveal how isolation techniques affect key performance metrics including cell purity, yield, viability, and transcriptional integrity.
Table 1: Quantitative Comparison of Microglia Isolation Methods
| Performance Metric | Magnetic Bead (MACS) | Percoll Gradient | Experimental Context |
|---|---|---|---|
| Purity | ~95% CD11b+ cells [12] | High, but may contain slight myeloid contamination [10] | Adult mouse brain, flow cytometry validation |
| Cell Yield | High efficiency [10] | Lower yield due to extensive processing [11] | Per adult mouse brain |
| Cell Viability | >85% [10] | Viability significantly affected by myelin removal method [12] | Trypan blue exclusion/Live-Dead staining |
| Activation State | Preserves in vivo phenotype when optimized [12] | Minimal activation with cold mechanical protocol [40] | Comparison to in vivo state |
| Technical Complexity | Moderate (requires antibody expertise) | Low to Moderate | Equipment and training needs |
| Throughput | Faster processing for single/multiple samples [10] | Lengthy centrifugation steps | Time per sample |
| Cost Considerations | Higher (antibodies, magnetic columns) | Lower (common laboratory reagents) | Per isolation |
Table 2: Transcriptional Artifact Assessment by Isolation Method
| Transcriptional Impact | MACS with Enzymatic Dissociation | Percoll with Cold Mechanical | References |
|---|---|---|---|
| Ex Vivo Activation | Significant without inhibitors [35] | Minimal with 4°C protocol [40] | Inflammatory gene induction |
| Housekeeping Gene Integrity | Potential alteration | Better preservation | [46] |
| Suitable for RNA Sequencing | With transcriptional/translational inhibitors | Excellent suitability | [40] [35] |
| Cell-Type Specific Signals | Well-preserved after isolation | Well-preserved after isolation | [35] |
The Scientist's Toolkit: Essential Research Reagents
Successful microglia isolation requires specific reagents and materials to maintain cell viability and minimize transcriptional artifacts.
Table 3: Essential Research Reagents for Microglia Isolation
| Reagent/Material | Function | Specific Examples |
|---|---|---|
| CD11b Microbeads | Immunomagnetic labeling for MACS | Anti-CD11b conjugated magnetic beads [1] |
| Percoll Solution | Density gradient medium for cell separation | 30-70% isotonic Percoll gradients [1] [12] |
| Transcriptional/Translational Inhibitors | Prevents ex vivo activation during processing | Actinomycin D, cycloheximide [35] |
| Neural Dissociation Kit | Enzymatic tissue digestion | Papain-based neural dissociation kits [12] |
| Myelin Removal Beads | Optional myelin depletion | Anti-myelin magnetic beads [12] |
| Cell Strainers | Removal of tissue aggregates | 40μm nylon mesh filters [12] |
| Viability Stains | Assessment of cell integrity | Trypan blue, Live/Dead fixable stains [12] |
Discussion and Research Implications
The choice between MACS and Percoll isolation methods significantly influences transcriptional profiling outcomes and should align with specific research goals.
MACS isolation provides superior purity and is ideal for studies requiring highly purified microglial populations, particularly when followed by downstream applications like single-cell RNA sequencing [10]. However, researchers must implement inhibitor cocktails during tissue dissociation to mitigate ex vivo activation [35]. The ability to sequentially isolate multiple neural cell types from a single sample makes MACS particularly valuable for comprehensive studies of neural circuits [1].
Percoll gradient centrifugation offers distinct advantages for transcriptomic integrity, especially when paired with cold mechanical dissociation [40]. This method eliminates heat-induced enzymatic artifacts, making it preferable for sensitive gene expression assays. Additionally, its lower cost and technical simplicity make it accessible for laboratories with limited resources [11].
For aging studies, special considerations apply. The proportion of oligodendrocyte precursor cells (OPCs) decreases during aging, while mature oligodendrocytes increase [46]. Furthermore, inhibitory neurons show increased transcriptional variability with age [46], potentially making them more susceptible to isolation-induced stress.
Both magnetic bead isolation and Percoll gradient centrifugation can successfully purify microglia for transcriptional profiling when optimized with appropriate safeguards. MACS provides higher purity and multi-cell type isolation capability, while Percoll with cold mechanical dissociation better preserves native transcriptomic states. The optimal choice depends on specific research priorities: purity versus transcriptomic integrity. As microglia research continues to evolve, standardization of isolation protocols that minimize ex vivo confounds will be essential for generating comparable and biologically relevant data across studies.
The pursuit of high-purity microglia that accurately reflect their in vivo state is fundamental to advancing neuroimmunology and drug development. The isolation process itself can induce ex vivo activation, potentially confounding functional assays and leading to misinterpretation of a cell's true physiological role [35]. This guide provides an objective comparison of two predominant isolation techniques—magnetic bead isolation and Percoll gradient centrifugation—focusing on their performance in yielding microglia with preserved phenotype for key functional assays like phagocytosis and cytokine response.
The central challenge in microglial isolation lies in their inherent sensitivity; as the brain's resident macrophages, they respond rapidly to environmental changes, including the mechanical and enzymatic stresses of tissue dissociation [35]. Therefore, the choice of isolation method is critical, not merely for achieving cellular purity but for ensuring that the isolated cells are biologically relevant for downstream applications.
Technical Comparison of Isolation Methods
Core Principles and Workflows
The two methods operate on fundamentally different principles for separating microglia from a heterogeneous brain cell suspension.
Magnetic-Activated Cell Sorting (MACS): This method uses antibody-mediated capture. Magnetic beads are conjugated to antibodies targeting specific microglial surface markers, such as CD11b (integrin alpha M). When the cell suspension is passed through a magnetic column, labeled microglia are retained while other cells are washed away. The positive cells are then eluted after column removal [1] [39]. A key advantage is the ability to perform sequential isolation of microglia, astrocytes, and neurons from the same brain sample using different antibody cocktails [1] [39].
Percoll Gradient Centrifugation: This is a density-based separation technique. A density gradient is formed, typically using layers of different Percoll concentrations (e.g., 20%-65%). During centrifugation, cells migrate to the interface matching their own buoyant density, effectively separating microglia from myelin debris, neurons, and other glial cells [1] [11]. This method circumvents the use of expensive antibodies and enzymatic digestion, which can sometimes affect cell viability [1].
The following workflow diagrams illustrate the key steps and decision points for each isolation method.
Diagram 1: Magnetic-Activated Cell Sorting (MACS) workflow for microglia isolation, showing potential for tandem cell isolation.
Diagram 2: Percoll density gradient centrifugation workflow for microglia isolation.
Quantitative Performance Metrics
Direct comparative studies provide critical data on the yield, purity, and viability of microglia isolated via these methods. The following table summarizes key performance metrics from relevant research.
Table 1: Performance comparison of microglia isolation methods based on experimental data.
| Performance Metric | Magnetic Bead (MACS) | Percoll Gradient | Experimental Context & Notes |
|---|---|---|---|
| Cell Yield | ~1.5 million cells/brain [11] | Varies by protocol; one study found ~1 million cells/brain [11] | Yield is highly dependent on animal age, brain region, and specific protocol used. |
| Purity | High (>95% CD11b+ reported) [35] | High purity achievable [11] | Purity for both methods is sufficient for most functional assays. |
| Viability | High viability maintained [47] | Maintains cell viability by avoiding enzymatic steps in some protocols [1] | Gentle, non-enzymatic dissociation improves viability for both methods. |
| Relative Purity | High purity, similar to FACS [35] | High purity achievable [11] | A study comparing MACS and FACS found equivalent purity [35]. |
| Ex Vivo Activation | Activation occurs primarily during tissue dissociation, not the sort itself [35] | Potential for activation during prolonged centrifugation and dissociation [11] | Can be minimized in both methods with transcriptional/translational inhibitors or cold non-enzymatic dissociation [35]. |
Impact on Downstream Functional Assays
The ultimate test of an isolation method is how well the purified microglia perform in biologically relevant functional assays, mimicking their roles in the brain.
Phagocytosis Assay
Phagocytosis, the clearance of pathogens and cellular debris, is a critical microglial function. The integrity of this process post-isolation is a key indicator of preserved phenotype.
- Protocol Summary: Isolated microglia are plated and allowed to adhere. Fluorescent latex beads (e.g., 1-2 μm diameter) are then added to the culture medium. After incubation (typically 2 hours), the cells are thoroughly washed to remove non-phagocytosed beads. Phagocytosis can be quantified using flow cytometry (measuring the rightward shift in fluorescence intensity) or confocal microscopy to confirm internalization [3].
- Method Comparison: Studies on primary human microglia isolated with simple, high-purity methods have demonstrated that over 90% of cells are phagocytically active [3]. The key to preserving this function is minimizing isolation-induced activation and stress. While both methods can yield highly phagocytic cells, the gentler handling and avoidance of excessive mechanical stress during Percoll gradient preparation can be beneficial.
Cytokine Response Profiling
Microglia modulate the brain's immune environment through the release of cytokines and chemokines. Testing their response to inflammatory stimuli validates their functional competence.
- Protocol Summary: Isolated microglia are treated with various stimuli such as LPS (to trigger a pro-inflammatory response), IL-1β, or IFNγ. After a defined period (e.g., 6-24 hours), the cell culture supernatant is collected. The secretome is then analyzed using techniques like ELISA or multiplex bead arrays to quantify the concentration of specific cytokines like IL-6, IL-8, MCP-1, and IP-10 [3].
- Method Comparison: The stimulus-specific response is a hallmark of functional microglia. For instance, human microglia respond to IFNγ with a significant increase in IP-10 secretion and to LPS/IL-1β with elevated IL-6, IL-8, and MCP-1, while IFNγ does not induce these latter cytokines [3]. The isolation method must not pre-activate the cells. Research indicates that ex vivo activation signatures arise predominantly during the initial tissue dissociation, not during the subsequent MACS or Percoll sorting step itself [35]. Therefore, optimizing the dissociation protocol is paramount for both methods to obtain accurate cytokine response profiles.
The Scientist's Toolkit: Essential Reagents and Materials
Successful isolation and functional testing of microglia require a suite of specialized reagents.
Table 2: Key research reagent solutions for microglia isolation and functional assays.
| Reagent/Material | Function | Example Application |
|---|---|---|
| Anti-CD11b Magnetic Beads | Immunomagnetic label for microglial surface protein CD11b (ITGAM) for positive selection [1]. | MACS isolation of microglia from a single-cell suspension of brain tissue. |
| Percoll Solution | Silica colloidal suspension used to form density gradients for cell separation based on buoyant density [1] [11]. | Separation of microglia from myelin, debris, and other neural cells. |
| Cell Culture Media (DMEM/F12) | Nutrient medium for maintaining and growing isolated microglial cells in vitro [11] [5]. | Base medium for culturing microglia post-isolation, often supplemented with growth factors and serum. |
| Fluorescent Latex Beads | Particles used to measure the phagocytic capacity of microglia; internalization is quantified via flow cytometry or microscopy [3]. | Phagocytosis functional assays. |
| Inflammatory Stimuli (LPS, IFNγ) | Agents used to challenge microglia and trigger a defined immune response for functional characterization [5] [3]. | Cytokine response profiling assays. |
| Transcriptional/Translational Inhibitors | Cocktails used during tissue dissociation to prevent rapid ex vivo gene expression changes that distort the native microglial phenotype [35]. | Minimizing isolation-induced artifacts in transcriptomic and functional studies. |
Both magnetic bead isolation and Percoll gradient centrifugation are capable of yielding high-purity, functional microglia. The choice between them depends on research priorities.
- Choose Magnetic Bead (MACS) if: Your priority is high specificity and the ability to perform sequential isolation of multiple neural cell types from the same sample [1] [39]. It is also well-suited for standardizing protocols across labs.
- Choose Percoll Gradient if: Your priority is cost-effectiveness, avoiding antibody-based labeling, and potential sensitivity to enzymatic digestion [1] [11]. It is an excellent choice for studies where the budget is a constraint and high yields of viable cells are needed.
Crucially, both methods are susceptible to ex vivo activation confounds introduced during the initial tissue dissociation [35]. Therefore, irrespective of the chosen method, incorporating cold, non-enzymatic dissociation or using transcriptional/translational inhibitors is highly recommended to best preserve the in vivo phenotype. For drug development applications where human relevance is paramount, cross-validating key findings using primary human microglia or iPSC-derived microglia models is essential, as significant species-specific differences exist [5] [3].
This guide provides a systematic comparison of two primary methods for microglia isolation—immunomagnetic bead separation and Percoll density gradient centrifugation—to assist researchers in selecting the appropriate technique based on specific research goals. We evaluate these methods across critical parameters including cell purity, viability, yield, phenotypic preservation, and compatibility with downstream applications. Experimental data from controlled studies reveal that while both methods can successfully isolate microglia, significant differences in performance metrics make each technique particularly suitable for specific research scenarios. Magnetic bead separation demonstrates superior purity and phenotypic preservation, whereas Percoll gradients offer advantages in cell yield and technical accessibility.
Technical Specifications at a Glance
Table 1: Direct comparison of microglia isolation techniques across key performance metrics
| Performance Parameter | Magnetic Bead Separation | Percoll Gradient |
|---|---|---|
| Typical Purity | 90-97% [12] [10] | 85-95% [12] [11] |
| Cell Viability | >85% [10] | >85% [12] [10] |
| Cell Yield | Lower [11] [10] | Higher [12] [11] |
| Processing Speed | Faster (∼2.5 hours) [11] [10] | Slower (∼4 hours) [11] |
| Technical Difficulty | Moderate (requires specific antibodies) | Moderate (density gradient handling) |
| Specialized Equipment | Magnetic separator columns | High-speed centrifuge |
| Phenotype Preservation | Excellent (minimal activation) [12] | Good [12] |
| Species Compatibility | Mouse & human [23] | Primarily mouse [12] [11] |
| Myelin Removal | Requires pre-clearing step [12] | Integrated in the method [12] |
| Cost per Sample | Higher (antibodies, columns) | Lower (Percoll reagent only) |
Table 2: Downstream application compatibility by isolation method
| Downstream Application | Magnetic Bead Separation | Percoll Gradient |
|---|---|---|
| RNA Sequencing | Excellent (high purity critical) [12] | Good (with high purity preparation) |
| Flow Cytometry | Excellent [12] | Good [12] |
| Primary Cell Culture | Good [4] [11] | Good [11] |
| Phagocytosis Assays | Excellent [12] [3] | Excellent [5] |
| Cytokine Secretion Studies | Excellent (phenotype preserved) [12] [3] | Good (check activation status) |
| Transcriptomics | Excellent [12] | Moderate (myelin contamination risk) |
| Western Blot/ELISA | Excellent [12] | Excellent [12] |
Experimental Protocols in Practice
Magnetic-Activated Cell Sorting (MACS) Protocol
The immunomagnetic separation technique leverages antibody-conjugated magnetic beads targeting microglia-specific surface markers, most commonly CD11b [12] [23].
Step-by-Step Workflow:
- Tissue Dissociation: Brain tissue is enzymatically digested using a neural tissue dissociation kit (e.g., Miltenyi Biotec) for 35 minutes at 37°C [12].
- Myelin Removal: The cell suspension is processed through a myelin removal step using 30% Percoll, sucrose, or commercial myelin removal beads to prevent column clogging and ensure purity [12].
- Antibody Incubation: Cells are stained with PE-conjugated anti-CD11b antibodies for 10 minutes [12].
- Magnetic Labeling: Anti-PE magnetic beads are added and incubated for 15 minutes [12].
- Magnetic Separation: The cell suspension is passed through a magnetic MS column placed in a separator. CD11b+ cells are retained in the column while negative cells flow through [12].
- Elution: Pure microglia are eluted from the column by removing it from the magnetic field and flushing with buffer [12].
This method has been successfully adapted for both mouse and human brain tissue, including from aged models, demonstrating its versatility [4] [23].
Percoll Density Gradient Protocol
The Percoll method separates microglia based on their inherent buoyant density, which differs from other brain cells and myelin debris [12] [11].
Step-by-Step Workflow:
- Tissue Homogenization: Mechanically dissociated brain tissue is centrifuged to form a cell pellet [11].
- Gradient Preparation: A density gradient is prepared, typically using 30% Percoll solution [12].
- Centrifugation: The cell pellet is resuspended in the Percoll solution and centrifuged for 10 minutes at 700× g [12].
- Myelin Removal: Myelin forms a layer on top of the gradient due to its lower density, while microglia and other cells pellet at the bottom [12].
- Cell Collection: The supernatant containing myelin is carefully removed, and the pelleted cells are washed with buffer before plating or further processing [12].
This method effectively removes myelin contamination in a single step without requiring specialized antibodies, making it cost-effective for studies where ultimate purity is less critical than yield [12] [11].
Microglia Isolation Workflow Comparison
Research Reagent Solutions
Table 3: Essential reagents and materials for microglia isolation protocols
| Reagent/Material | Function | Example Product |
|---|---|---|
| CD11b Microbeads | Immunomagnetic labeling of microglia | Miltenyi Biotec [12] |
| Percoll | Density gradient medium for cell separation | GE Healthcare [12] |
| Neural Tissue Dissociation Kit | Enzymatic tissue digestion | Miltenyi Biotec [12] |
| M-CSF/GM-CSF | Microglia culture growth factors | R&D Systems [11] |
| Magnetic Separator & Columns | Magnetic cell separation | Miltenyi Biotec MS Columns [12] |
| Cell Strainers | Removal of tissue clumps | 40μm or 70μm nylon mesh [12] [5] |
| Anti-Myelin Beads | Myelin debris removal (MACS) | Miltenyi Biotec [12] |
| CD11b Antibody | Microglia identification | PE-conjugated anti-CD11b [12] |
Comparative Experimental Data Analysis
Purity and Viability Assessment
In a direct methodological comparison, both techniques demonstrated high viability exceeding 85%, with no significant difference in cell health immediately following isolation [10]. However, purity analysis revealed that magnetic bead separation consistently achieved higher purity levels (90-97%) compared to Percoll gradients [12] [10]. Flow cytometry confirmation of isolated microglia showed minimal contamination from astrocytes (GFAP+) or neurons (NeuN+) in MACS-isolated populations [12].
Percoll-isolated microglia occasionally showed slight myeloid cell contamination, which could impact downstream applications requiring extremely pure populations [10]. This purity advantage makes magnetic bead separation particularly valuable for transcriptomic studies and RNA sequencing, where even minor contamination can alter results [12].
Yield and Functional Preservation
Despite lower purity, Percoll gradients consistently yielded higher cell numbers per brain processed [12] [11]. One study comparing isolation protocols found modified Percoll methods provided superior yields compared to magnetic bead approaches [11]. This advantage makes Percoll preferable when cell quantity is the primary concern, such as for proteomic studies or establishing primary cultures.
Functional assessments confirmed that both methods preserve key microglial characteristics. Microglia isolated via magnetic beads maintained their in vivo activation state, with cells from LPS-treated mice showing pro-inflammatory phenotypes while control microglia remained quiescent [12]. Both isolation methods yielded microglia capable of phagocytosis and appropriate cytokine secretion in response to inflammatory stimuli [12] [5] [3].
Method Selection Guidelines
Application-Based Recommendations
Choose Magnetic Bead Separation When:
Choose Percoll Gradient When:
Special Considerations for Aging and Disease Models
Research using aged animals presents unique challenges, as aged microglia exhibit distinct gene expression profiles and higher baseline activation ("inflammaging") [4]. When studying aged microglia, magnetic bead separation may be advantageous due to its superior phenotypic preservation, ensuring that observed differences reflect biology rather than isolation artifacts [4] [12]. However, successful microglia culture from aged mice (up to 18 months) has been achieved using specialized media formulations regardless of isolation method [4].
For neurodegenerative disease research, where microglial dysfunction contributes to pathology, the preservation of native phenotype becomes particularly important. Magnetic bead separation has demonstrated effectiveness in isolating microglia with preserved phenotypes from neuroinflammatory models [12].
Conclusion
The choice between magnetic bead isolation and Percoll gradient centrifugation is not one of absolute superiority but of strategic alignment with research goals. Magnetic bead separation, leveraging antibodies like CD11b, typically offers higher specificity and purity, which is crucial for sequencing and specific phenotypic studies. In contrast, the Percoll method provides a gentler, cost-effective approach that can yield cells with high viability and potentially preserve a more native state, suitable for functional and long-term culture assays. Future directions should focus on standardizing these protocols across labs, developing even more specific markers to distinguish microglia from infiltrating macrophages, and integrating these isolation techniques with advanced human models, such as monocyte-derived microglia, to bridge the gap between rodent studies and human clinical applications in drug development.
References
- 1. Isolation of Primary Brain Cells: Challenges and Solutions [https://pmc.ncbi.nlm.nih.gov/articles/PMC12320896/]
- 2. iPSC-derived human microglia-like cells to study neurological ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5482419/]
- 3. Isolation of highly enriched primary human microglia for ... [https://www.nature.com/articles/srep19371]
- 4. Novel In Vitro Culture of Microglia from Aged Mice [https://pmc.ncbi.nlm.nih.gov/articles/PMC12365726/]
- 5. In vitro models of microglia: a comparative study [https://www.nature.com/articles/s41598-025-99867-z]
- 6. Protocol I: Isolation of adult microglia using magnetic sorting [https://bio-protocol.org/exchange/minidetail?id=19040132&type=30]
- 7. Cell Isolation Methods: Magnetic Bead Separation [https://www.akadeum.com/technology/magnetic-bead-cell-separation/?srsltid=AfmBOooe6iBtGkUndHGNxE-kUIVL6A3KlKHFxMPH9c8el_Me_GyE_d8N]
- 8. Magnetic Cell Separation | Cell Isolation Technology [https://www.stemcell.com/cell-separation/magnetic-cell-isolation]
- 9. Fractionation of cells and subcellular particles with Percoll [https://www.sciencedirect.com/science/article/abs/pii/S0165022X0000066X]
- 10. Methodological comparison of FACS and MACS isolation ... [https://www.sciencedirect.com/science/article/pii/S0022175920301186]
- 11. Microglia isolation from aging mice for cell culture [https://www.frontiersin.org/journals/cellular-neuroscience/articles/10.3389/fncel.2023.1082180/full]
- 12. Efficient isolation of live microglia with preserved phenotypes ... [https://jneuroinflammation.biomedcentral.com/articles/10.1186/1742-2094-9-147]
- 13. Development of a novel purification protocol to isolate and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9493764/]
- 14. Comparison of immunomagnetic beads coated with protein ... [https://www.sciencedirect.com/science/article/abs/pii/002217599390101C]
- 15. Characterization and Isolation of Mouse Primary Microglia by ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5931276/]
- 16. Continuous Percoll Gradient Centrifugation of Erythrocytes ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9028966/]
- 17. Percoll Gradient Centrifugation: What to consider when ... [https://labsup.net/blogs/blog/percoll-gradient-centrifugation-what-to-consider-when-isolating-cells-subcellular-particles-and-virus?srsltid=AfmBOooy4rnLqE4OZ2V1jOyKMfO0TUN8STptiZMd6qQYL_DmqxqLBuN3]
- 18. The influence of Percoll® density gradient centrifugation ... [https://bmcvetres.biomedcentral.com/articles/10.1186/s12917-022-03408-z]
- 19. Density Gradient Centrifugation - an overview [https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/density-gradient-centrifugation]
- 20. Validation of flow cytometry and magnetic bead-based ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4662612/]
- 21. Impact of microglia isolation and culture methodology on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11000335/]
- 22. Microglia isolation from adult mouse brain - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC4145600/]
- 23. Protocol Isolation of Microglia from Mouse or Human Tissue [https://www.sciencedirect.com/science/article/pii/S2666166720300228]
- 24. Efficient isolation of live microglia with preserved phenotypes ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3418565/]
- 25. Human CD11b Magnetic Beads MS008 [https://www.ptglab.com/products/Human-CD11b-Magnetic-Beads-MS008.htm?srsltid=AfmBOor_C9B6AwVwQjcvHdMFbtISXI1hsYSnYfcb18-IGw3a14o1qYOr]
- 26. BD IMag™ Anti-CD11b Magnetic Particles - DM [https://www.bdbiosciences.com/en-us/products/reagents/cell-preparation-separation-reagents/magnetic-cell-separation/anti-cd11b-magnetic-particles-dm.558013]
- 27. Purification in the Real World: 5 Uses You'll Actually... Magnetic Bead [https://www.linkedin.com/pulse/magnetic-bead-purification-real-world-5-uses-youll-yqckc/]
- 28. EasySep™ Mouse CD11b Positive Selection Kit II [https://www.stemcell.com/products/easysep-mouse-cd11b-positive-selection-kit-ii.html]
- 29. Critical factors for flow cytometry analysis of the brain - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12220843/]
- 30. Proposed practical protocol for flow cytometry analysis of ... [https://www.frontiersin.org/journals/cellular-neuroscience/articles/10.3389/fncel.2022.1017976/full]
- 31. Dissociation of neonatal and adult mice brain for ... [https://www.sciencedirect.com/science/article/pii/S2451830120300017]
- 32. Isolation of Microglia from Mouse or Human Tissue - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7416840/]
- 33. Protocol to sequentially isolate mouse oligodendrocytes ... [https://www.sciencedirect.com/science/article/pii/S2666166725005453]
- 34. Cell isolation and separation: Essential techniques [https://www.abcam.com/en-us/knowledge-center/cell-biology/cell-isolation-and-separation]
- 35. Minimizing the Ex Vivo Confounds of Cell-Isolation ... [https://www.eneuro.org/content/9/2/ENEURO.0348-21.2022]
- 36. Video: Rapid and Refined CD11b Magnetic of Primary... Isolation [https://www.jove.com/v/55364/rapid-refined-cd11b-magnetic-isolation-primary-microglia-with]
- 37. of Isolation and Analysis of Protein Expression by Flow... Microglia [https://bio-protocol.org/en/bpdetail?id=4091&type=0]
- 38. A new method for obtaining bankable and expandable adult ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8680120/]
- 39. Protocol for the isolation and culture of microglia ... [https://www.sciencedirect.com/science/article/pii/S2666166723007712]
- 40. Using mechanical homogenization to isolate microglia from ... [https://www.sciencedirect.com/science/article/pii/S2666166722005500]
- 41. Lymphoid gene expression supports neuroprotective ... [https://www.nature.com/articles/s41586-025-09662-z]
- 42. Quantitative Assessment of Immune Cells in the Injured Spinal Cord... [https://www.jove.com/t/2698/quantitative-assessment-immune-cells-injured-spinal-cord-tissue-flow]
- 43. Simplified quantitative analysis of spinal cord cells from... [https://pubmed.ncbi.nlm.nih.gov/15914195/]
- 44. Isolation of Primary Brain Cells: Challenges and Solutions [https://www.fortunejournals.com/articles/isolation-of-primary-brain-cells-challenges-and-solutions.html]
- 45. Isolation and Phenotyping of Adult Mouse Microglial Cells | SpringerLink [https://link.springer.com/protocol/10.1007/978-1-4939-7837-3_7]
- 46. Single-cell transcriptomic and genomic changes in the ... [https://www.nature.com/articles/s41586-025-09435-8]
- 47. Comparing the Efficiency of T Cell Isolation Techniques [https://www.akadeum.com/blog/efficient-t-cell-isolation-techniques-comparison/?srsltid=AfmBOooT0BGWkVtl3lpNxImRIKfsy6TCN6vyHnN_kEkecV2sdPU1wrlQ]